Clear Sky Science · en
Study on biomarkers of homocysteine-induced transformation of vascular smooth muscle cells into foam cells
Why clogged arteries start with tiny cell changes
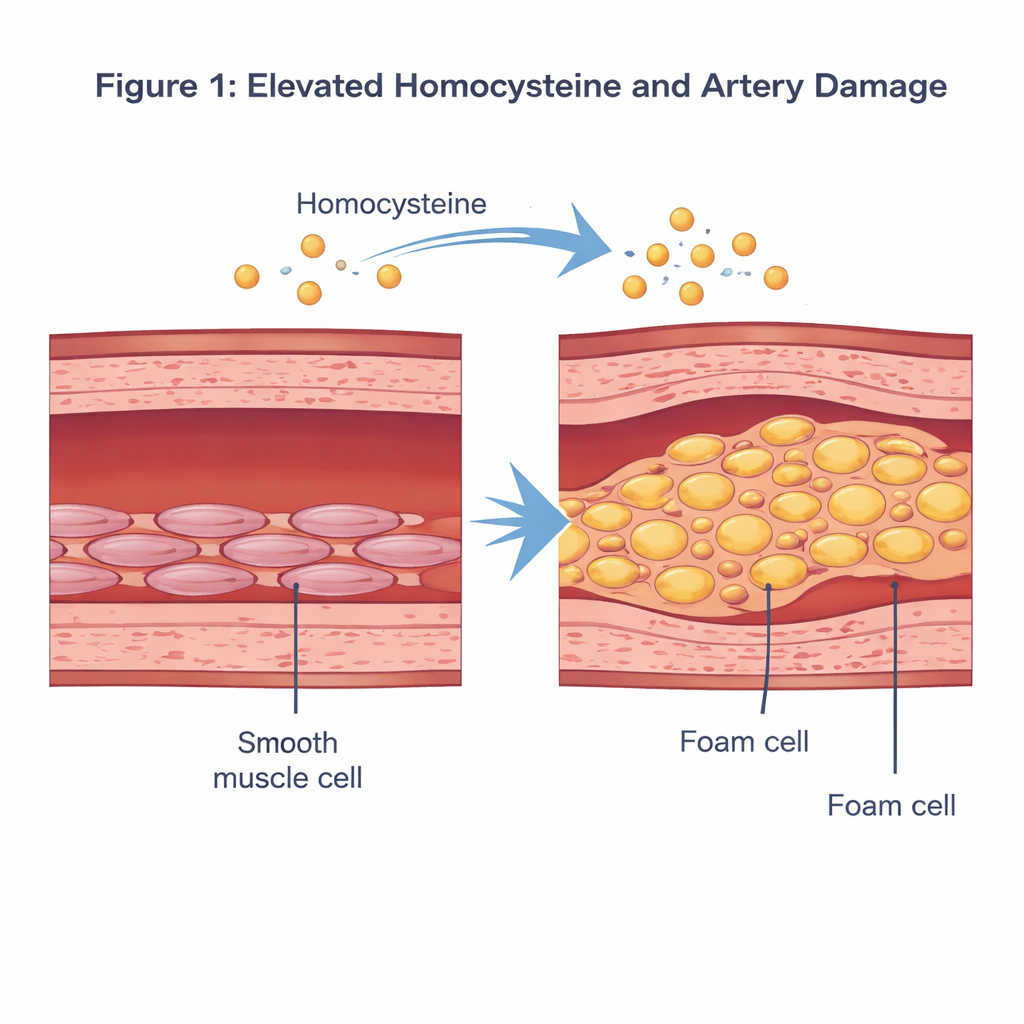
Atherosclerosis—the slow clogging and hardening of arteries—is a leading cause of heart attacks and strokes. We usually hear about cholesterol and diet, but less about how individual cells in the vessel wall change as disease develops. This study looks closely at one such trigger, a blood molecule called homocysteine, and shows how it can push normally well‑behaved muscle cells in arteries to hoard fat and turn into so‑called foam cells, the building blocks of dangerous plaques.
Amino acid troublemaker in the bloodstream
Homocysteine is a sulfur‑containing amino acid produced when our bodies process the dietary nutrient methionine. Under normal conditions, its level in blood is low. When it rises above about 15 micromoles per liter—a condition called hyperhomocysteinemia—epidemiological studies have linked it to a higher risk of cardiovascular disease. Earlier research suggested that excess homocysteine can damage the inner lining of arteries, stir up inflammation, and alter the behavior of vascular smooth muscle cells, which sit in the middle layer of the vessel wall and help regulate blood pressure by contracting and relaxing.
How artery muscle cells turn into fat‑laden foam cells
The researchers focused on how homocysteine reshapes these smooth muscle cells. In lab dishes, they exposed human vascular smooth muscle cells to a dose of homocysteine meant to mimic a disease‑like state and compared them with untreated cells. Using standard stains and biochemical tests, they saw that homocysteine‑treated cells filled up with red‑colored fat droplets, and their cholesterol and triglyceride levels rose sharply. At the same time, protein markers of their usual “contractile” identity fell, while markers of a more synthetic, plaque‑forming state increased. Taken together, these changes show that homocysteine can drive smooth muscle cells away from their normal role and toward a foam cell‑like, fat‑loaded phenotype that contributes directly to plaque formation.
Hunting for molecular fingerprints inside the cells
To understand which proteins change during this transformation, the team turned to modern proteomics, a technology that can measure thousands of proteins at once. They compared protein levels in homocysteine‑treated cells versus controls and reliably quantified nearly 4,800 proteins. Among these, 54 stood out as significantly altered: 13 increased and 41 decreased. Many of the shifting proteins are involved in lipid handling, cell survival, oxidative stress, and the structural remodeling of cells. The authors then used bioinformatics tools to group these proteins into functional pathways and to map how they interact with one another, highlighting networks linked to cholesterol metabolism and stress responses in the vessel wall.
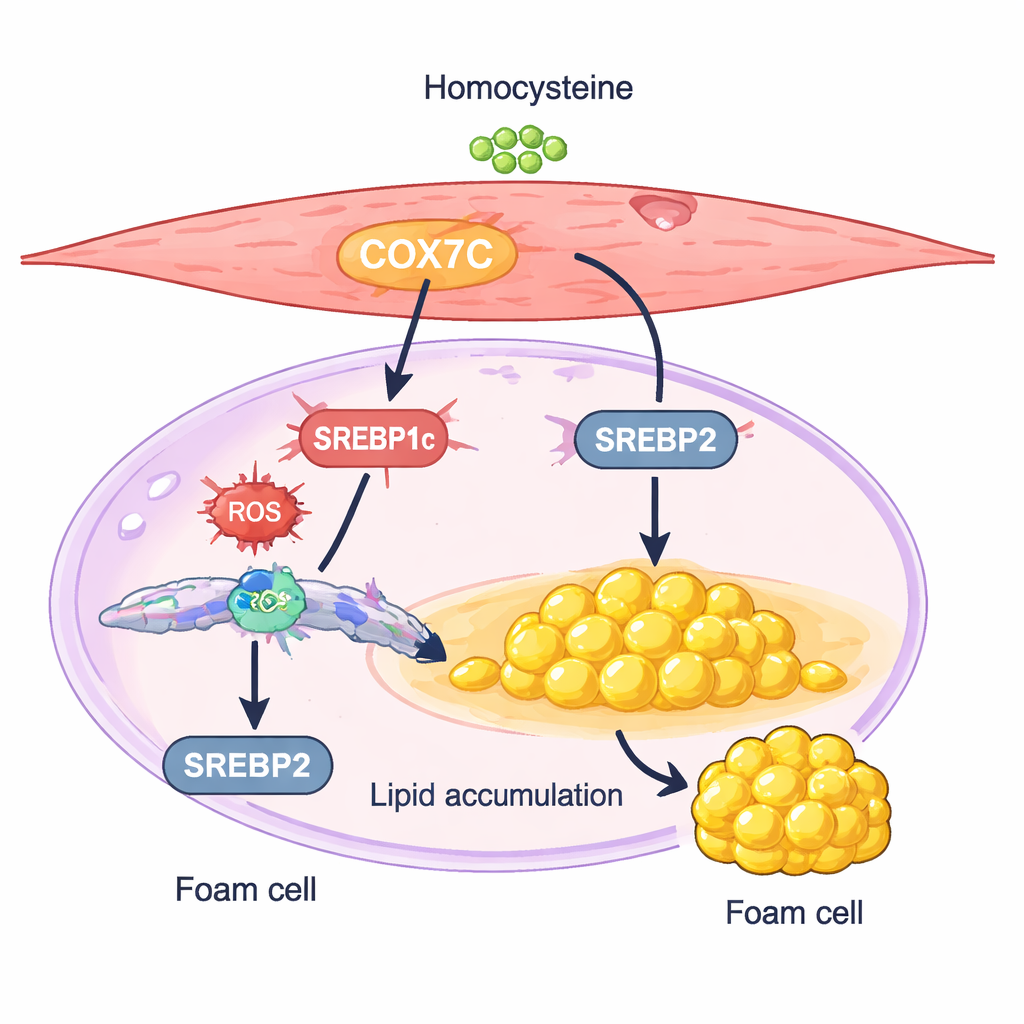
A mitochondrial switch that pushes cells to store fat
One protein, called COX7C, emerged as a particularly interesting suspect. COX7C is a component of the cell’s power plants, the mitochondria, where it helps manage energy production. In the homocysteine‑treated smooth muscle cells, COX7C levels were higher than in controls. The researchers showed that boosting COX7C further made the cells accumulate even more fat and express higher levels of two master regulators of lipid production, SREBP1c and SREBP2. Silencing COX7C had the opposite effect: it blunted SREBP activity, reduced cholesterol and triglyceride build‑up, and lessened foam cell‑like changes. These results support a model in which homocysteine ramps up COX7C in mitochondria, increases cellular stress signals, and in turn activates SREBP‑driven fat synthesis, tipping smooth muscle cells into a lipid‑hoarding state.
What this means for heart health and future therapies
This work does not yet prove what happens in human arteries, and it was done in cultured cells rather than in patients. Still, it offers a detailed look at how elevated homocysteine may help convert helpful artery muscle cells into harmful foam cells, and it identifies COX7C and related proteins as potential biomarkers or drug targets. For a layperson, the key message is that beyond “good” and “bad” cholesterol, small shifts in blood chemistry—like excess homocysteine—can reprogram the vessel wall from within. By pinpointing molecular players such as COX7C and the SREBP pathway, this study lays groundwork for future strategies aimed at stopping plaques before they form, by keeping arterial cells from becoming miniature fat‑storage depots.
Citation: Wang, X., Ma, X., Zhang, X. et al. Study on biomarkers of homocysteine-induced transformation of vascular smooth muscle cells into foam cells. Sci Rep 16, 7411 (2026). https://doi.org/10.1038/s41598-026-38763-6
Keywords: atherosclerosis, homocysteine, foam cells, vascular smooth muscle cells, COX7C