Clear Sky Science · en
Immune checkpoint inhibition increases antigen-specific T cell response in head and neck cancer
Turning the Body’s Defenses Against Head and Neck Cancer
Head and neck cancers remain difficult to treat, and even powerful new immunotherapies only help a fraction of patients. This study explores a promising idea: teaching immune cells to better recognize cancer-specific signals, then freeing those cells from molecular “brakes” so they can attack tumors more effectively. For people living with, or at risk for, head and neck cancer, this work points toward future vaccines and drug combinations that could make current treatments more potent and longer-lasting.
Why Current Immunotherapy Helps So Few
Modern cancer immunotherapy often targets proteins called immune checkpoints, which act like brakes on T cells, the white blood cells that can destroy tumors. In head and neck squamous cell carcinoma (HNSCC), drugs that block the checkpoint PD-1—such as Nivolumab or Pembrolizumab—are already in use. Yet only about 20% of patients benefit, and many cancers eventually progress. The authors set out to understand, in a controlled lab setting, how blocking PD-1 and related checkpoints affects T cells that are primed to recognize tumor-specific markers, and whether combining multiple checkpoint drugs adds anything beyond PD-1 alone.
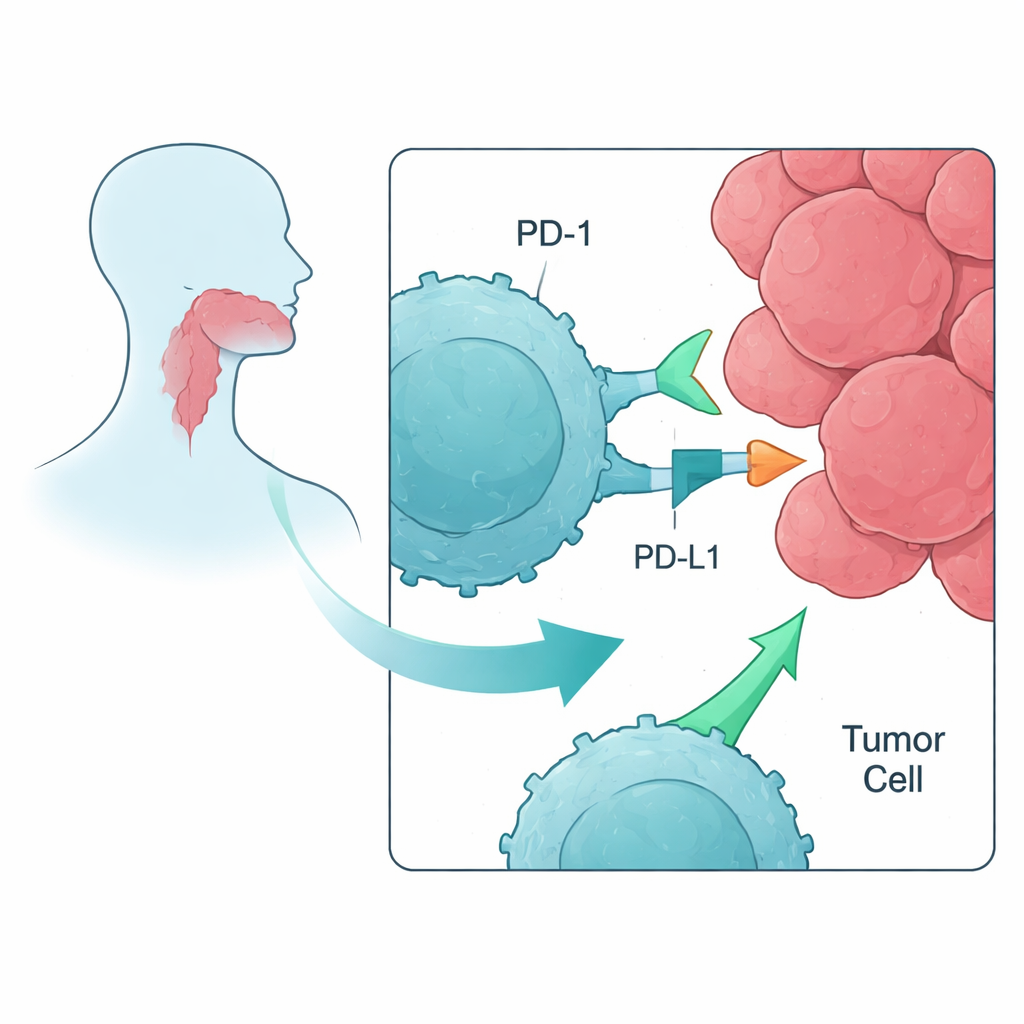
Training Immune Cells to See Cancer Targets
The researchers began with blood cells from healthy volunteers rather than from patients, to model how a less damaged immune system might respond early in disease. They isolated peripheral blood mononuclear cells, which include T cells, and exposed them to short protein fragments—peptides—taken from tumor-associated antigens commonly found in head and neck cancers. Three such targets proved especially good at waking up T cells: MAGE, NY-ESO-1, and PRAME. Over about a week in mixed lymphocyte–peptide cultures, these peptides acted like mini-vaccines, expanding rare T cells that could specifically recognize each cancer-related marker.
Releasing the Brakes on Trained T Cells
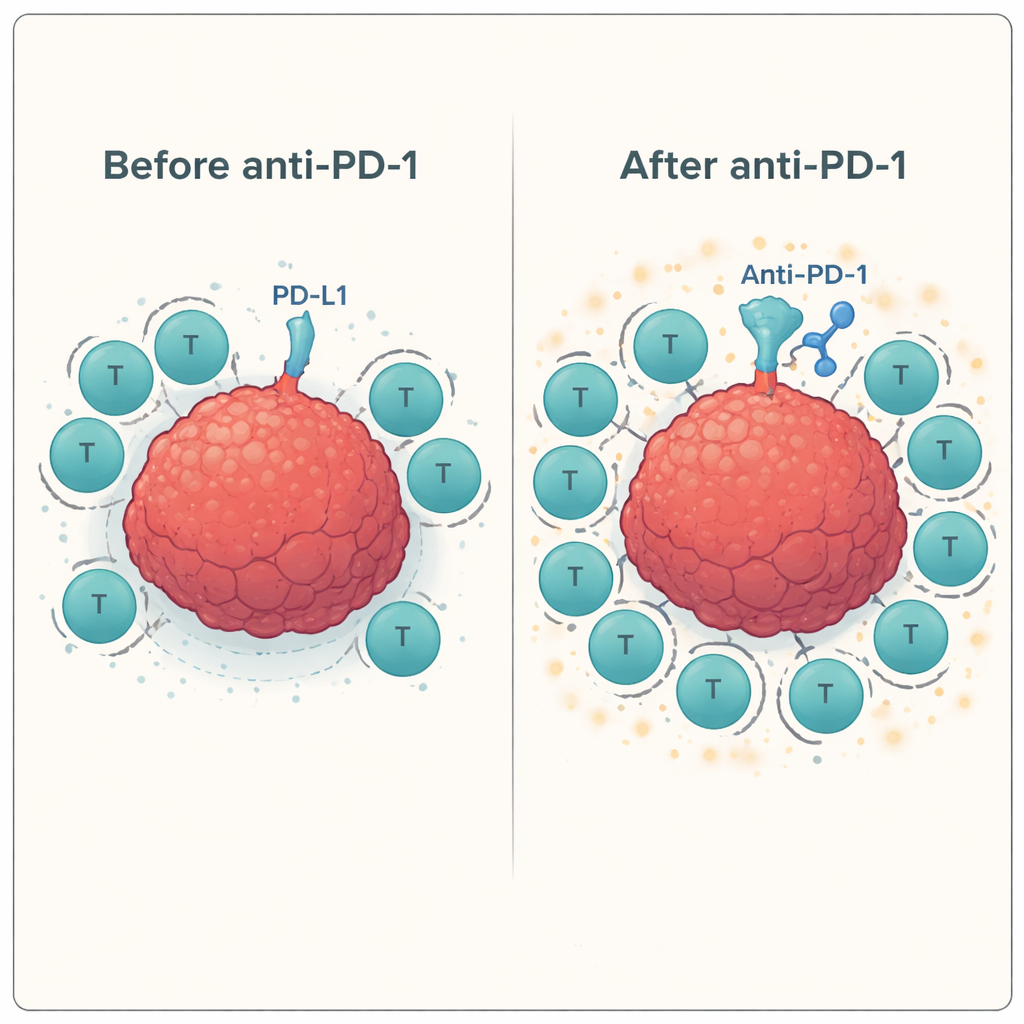
Next, the team tested how these trained T cells behaved when confronted with actual head and neck cancer cells in the presence or absence of checkpoint-blocking antibodies. They used a standard lab assay (ELISPOT) to measure the release of interferon-gamma and granzyme B—signals that T cells are activated and capable of killing. Across multiple donors, adding the anti-PD-1 antibody consistently boosted the response of antigen-specific T cells against the tumor cell line, often increasing activity by 60–100% or more compared with peptide stimulation alone. In contrast, blocking other checkpoints, such as LAG-3 or TIM-3, either alone or in combination with PD-1 blockade, produced little to no added benefit in this system.
Mapping the Immune Landscape in Detail
To understand why some combinations worked and others did not, the researchers used flow cytometry, a technique that counts and characterizes individual cells. They showed that both T cells and tumor cells express PD-1 and its ligands PD-L1 and PD-L2 at meaningful levels, creating multiple contact points where the PD-1 brake can be engaged—and therefore blocked by drugs. Markers tied to LAG-3 and TIM-3 were also present, but the tumor cell line expressed only a subset of their partner molecules, which may help explain the limited impact of targeting those checkpoints. They also observed a trend toward increased “go signals” (co-stimulatory molecules such as CD137 and GITR) on T cells after peptide stimulation and PD-1 blockade, suggesting that once the main brake is released, the accelerator can press down more firmly.
What This Could Mean for Future Patients
Because these experiments were done outside the body, they cannot fully mimic the complex tumor environment or the often-weakened immune systems of patients who have undergone chemotherapy and radiation. Even so, the findings offer a clear message in plain terms: when T cells are first taught to recognize specific cancer flags (through a peptide vaccine) and then relieved of the PD-1 brake, they become much better at attacking head and neck tumor cells. Adding more checkpoint drugs on top of PD-1 did not help in this setting. The authors conclude that a focused strategy—vaccinating patients with tumor-associated peptides such as MAGE, NY-ESO-1, or PRAME, combined with PD-1 blockade—deserves testing in clinical trials as a way to turn more patients’ immune systems into effective cancer fighters.
Citation: Schuler, P.J., Oliveri, F., Puntigam, L. et al. Immune checkpoint inhibition increases antigen-specific T cell response in head and neck cancer. Sci Rep 16, 5583 (2026). https://doi.org/10.1038/s41598-026-38740-z
Keywords: head and neck cancer, immune checkpoint therapy, PD-1 blockade, cancer vaccines, tumor antigens