Clear Sky Science · en
Efficacy of phytochemicals derived from Artocarpus heterophyllus (Jackfruit) as inhibitors against NS2B/NS3 protease of dengue virus: an in-silico investigation
Why Jackfruit Matters for a Mosquito-Borne Disease
Dengue fever, spread by mosquitoes, sickens millions of people every year and can sometimes turn deadly. Yet doctors still have no widely available pill that directly stops the virus once someone is infected. This study explores an unusual source of potential treatments: natural chemicals found in jackfruit, a common tropical tree. Using powerful computer simulations, the researchers asked whether any of these plant compounds might latch onto a key viral part and slow the virus down.
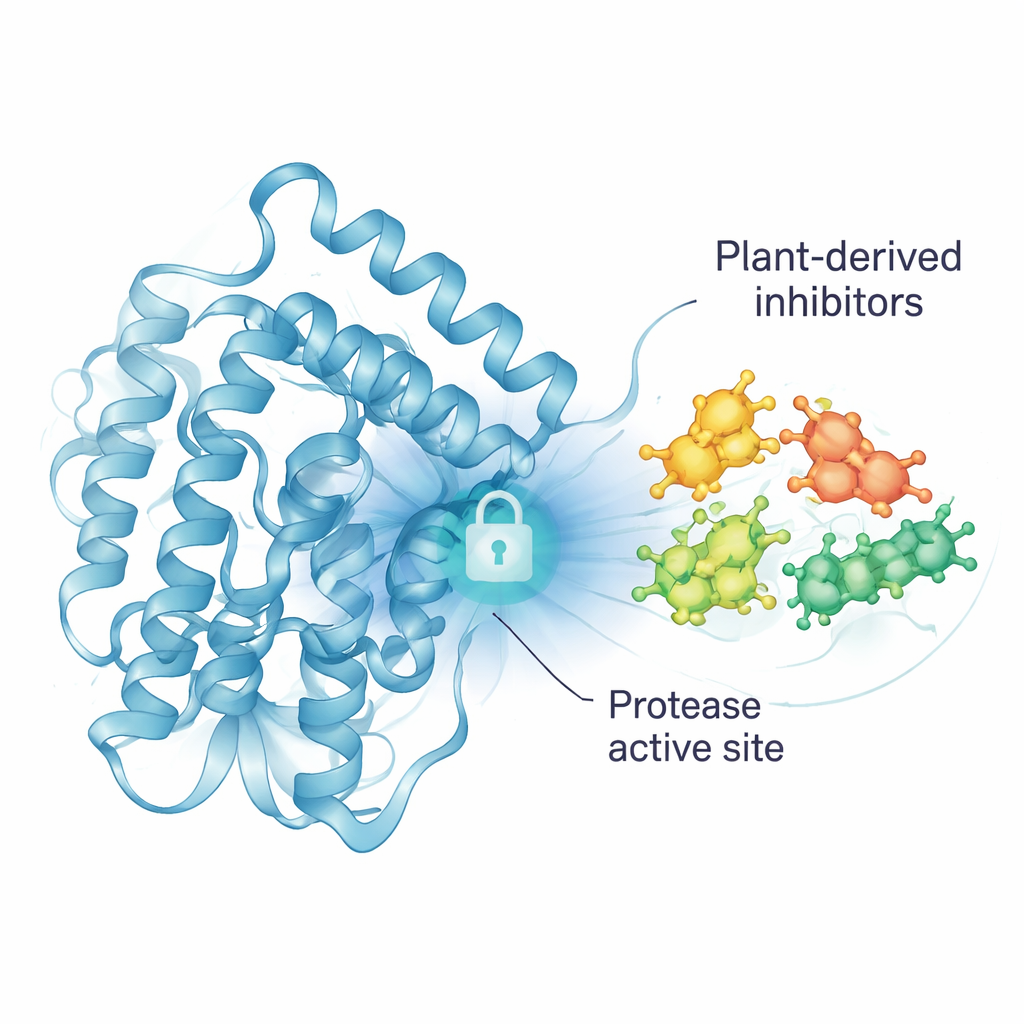
A Viral “Scissor” as a Key Weak Point
To multiply inside the body, the dengue virus relies on a molecular “scissor” called the NS2B/NS3 protease. This tiny machine cuts a long viral protein chain into smaller, working pieces. If the scissor is jammed, the virus cannot properly assemble itself, and infection should stall. Because of this, scientists worldwide see the protease as a prime target for antiviral drugs. However, past attempts to design such drugs have often failed due to poor strength, side effects, or lack of success in patients, so new types of molecules are still urgently needed.
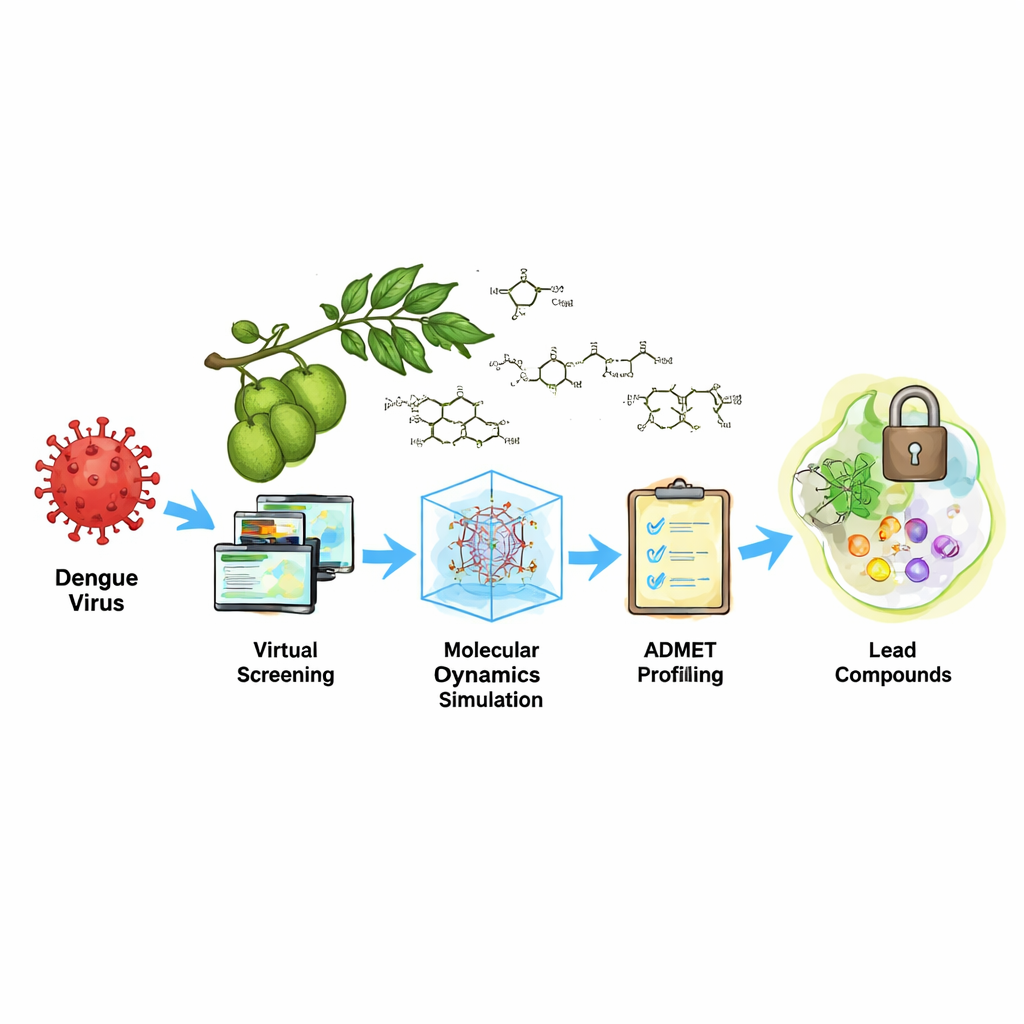
Jackfruit Chemistry Put to the Test
Jackfruit has a long history in traditional medicine and is packed with diverse plant chemicals. The team gathered 47 known jackfruit compounds from scientific databases and prepared them for “virtual screening.” In this process, computer programs estimate how well each compound might fit into the protease’s active site—the cutting groove of the viral scissor. The researchers used several layers of docking calculations to predict how tightly each molecule could bind and how well it matched the shape and charge of the target. They then applied a more detailed energy calculation method, known as MM-GBSA, to refine their list of candidates and focus on those with the strongest predicted grip.
Three Standout Molecules from the Crowd
From the original 47 compounds, three jackfruit molecules rose to the top: Oxidihydroartocarpesin, Cyanomaclurin, and Dihydromorin. All three were predicted to nestle into the protease’s active groove and form multiple noncovalent contacts with the “catalytic triad,” a trio of amino acids (His51, Asp75, Ser135) that does the actual cutting. These contacts included hydrogen bonds and hydrophobic interactions, which together help hold the compounds in place. In the energy calculations, the three molecules showed more favorable binding than many other tested compounds and performed similarly to a known reference inhibitor, suggesting they could interfere with the protease’s function.
Simulating the Viral Machine in Motion
Proteins are not statues; they move and flex in water and inside cells. To see how stable the jackfruit compounds would be over time, the researchers ran long molecular dynamics simulations, effectively watching the protease and each candidate molecule interact over tens of nanoseconds in a virtual environment. When no compound was bound, the protease shifted and wobbled more, especially around its active site. When the jackfruit molecules were bound, the overall structure became more compact and steady. Motion near the key catalytic residues decreased, and the protein surface exposed to water shrank slightly. These changes suggest that the compounds help “lock” the protease into a less flexible shape that is less capable of cutting its viral targets.
Safety Hints and the Road Ahead
The team also used online tools to estimate how the three compounds might behave in the human body—whether they could be absorbed, whether they might damage the liver, or whether they might cause genetic harm. The early signs were mixed: the molecules generally fit many criteria for drug-like behavior and did not raise alarms for liver damage or certain types of toxicity. However, the predictions hinted at a possible risk of cancer-like effects that would need to be checked carefully in laboratory and animal studies. Because of this, the authors view these compounds as starting points, or “lead” structures, rather than ready-made drugs.
What This Means for Future Dengue Treatments
For non-specialists, the main message is that everyday plants such as jackfruit may harbor promising blueprints for future antiviral drugs. This study did not test the compounds in cells or animals, so it does not offer a cure for dengue today. Instead, it uses advanced computer methods to narrow a large list of natural molecules down to a few that are most likely to block a crucial viral machine. By pointing drug designers toward Cyanomaclurin, Oxidihydroartocarpesin, and Dihydromorin, and by revealing how these compounds may stiffen and disable the dengue protease, the work helps chart a more focused path toward medicines that could one day turn a dangerous infection into a far more manageable illness.
Citation: Uddin, M.A.R., Paul, A.C., Islam, M.S. et al. Efficacy of phytochemicals derived from Artocarpus heterophyllus (Jackfruit) as inhibitors against NS2B/NS3 protease of dengue virus: an in-silico investigation. Sci Rep 16, 7543 (2026). https://doi.org/10.1038/s41598-026-38726-x
Keywords: dengue virus, jackfruit phytochemicals, protease inhibitors, virtual screening, antiviral drug discovery