Clear Sky Science · en
Chemical synthesis of novel aminopyrimidin-4-yl-1H-pyrazole derivatives as spleen tyrosine kinase (SYK) inhibitors
Why blocking a single enzyme could help fight cancer and inflammation
Our immune system relies on fast internal messaging to tell cells when to attack invaders and when to stand down. When these signals go wrong, they can fuel autoimmune diseases and cancer. This study explores a promising way to quiet those faulty signals by designing small drug-like molecules that selectively block a key enzyme, spleen tyrosine kinase (SYK), which sits near the heart of many immune reactions.
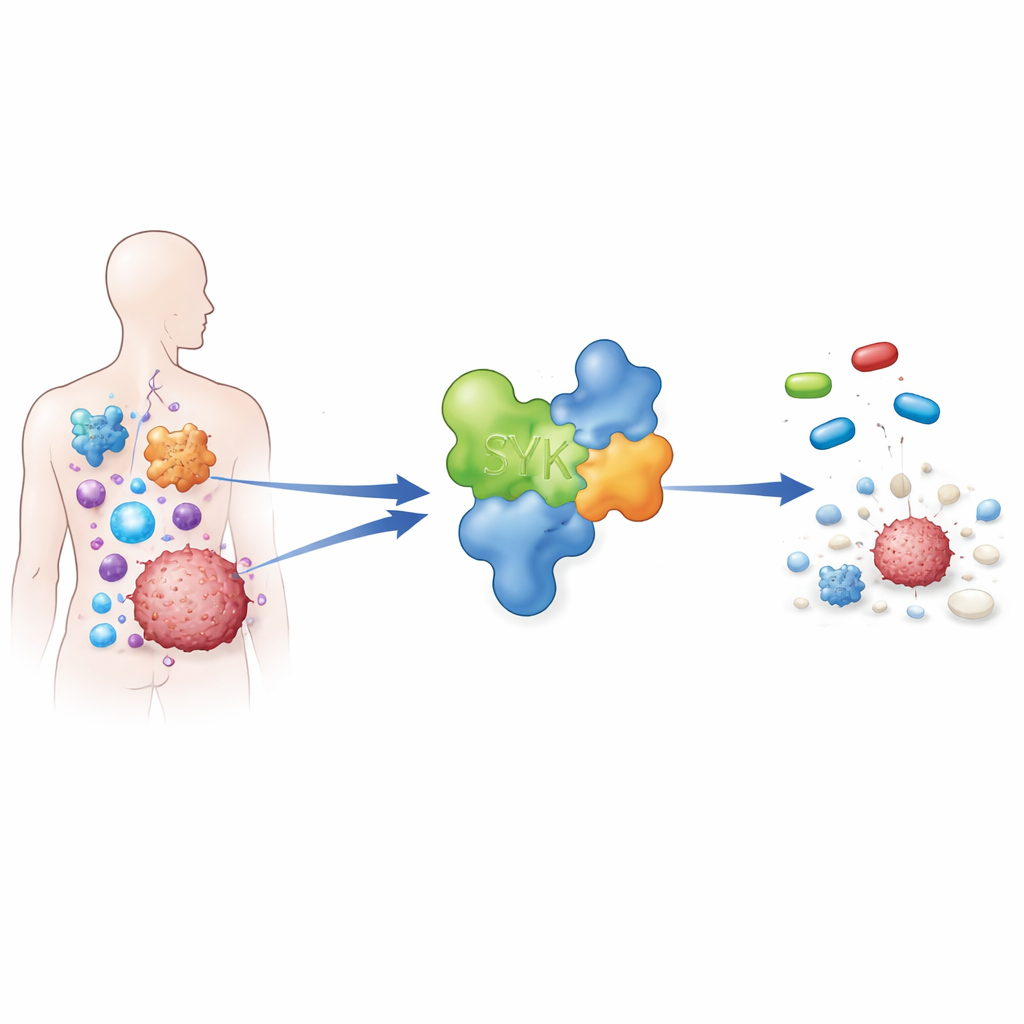
A signal switch at the core of many diseases
SYK acts like a master switch inside certain immune cells. When activated, it helps drive inflammation, antibody production, and cell growth. In healthy settings this is useful for fighting infections. But in conditions such as rheumatoid arthritis, asthma, and several blood and solid cancers, SYK activity can become excessive, keeping immune cells switched on and supporting tumor survival. Because of this central role, researchers worldwide are trying to develop SYK-blocking medicines that are powerful enough to work, yet precise enough to avoid hitting many other enzymes and causing side effects.
Building a family of tailored small molecules
The authors created a new series of related molecules built around a common chemical scaffold called aminopyrimidin-4-yl-1H-pyrazole. Starting from simple building blocks, they stitched these pieces together step by step and then modified one portion of the structure to produce nine final candidates. Each molecule was carefully checked using standard analytical methods to confirm that its structure and purity were as intended. This modular approach allowed the team to probe how subtle changes in shape and flexibility affect the way these compounds interact with SYK.
Watching how the compounds grip their target
Before heading to the lab bench, the researchers used computer simulations to see how tightly each molecule might latch onto SYK. Docking calculations estimated how well the compounds fit into the enzyme’s binding pocket, highlighting one candidate, labeled 44, as especially promising. More detailed molecular dynamics simulations, in which the protein–drug complex is followed over hundreds of nanoseconds, showed that 44 forms a particularly stable partnership with SYK. The protein remained compact, key contacts stayed in place, and the system settled into a single, low-energy shape with many strong hydrogen bonds, especially involving amino acids such as tryptophan and serine that help cradle the molecule.
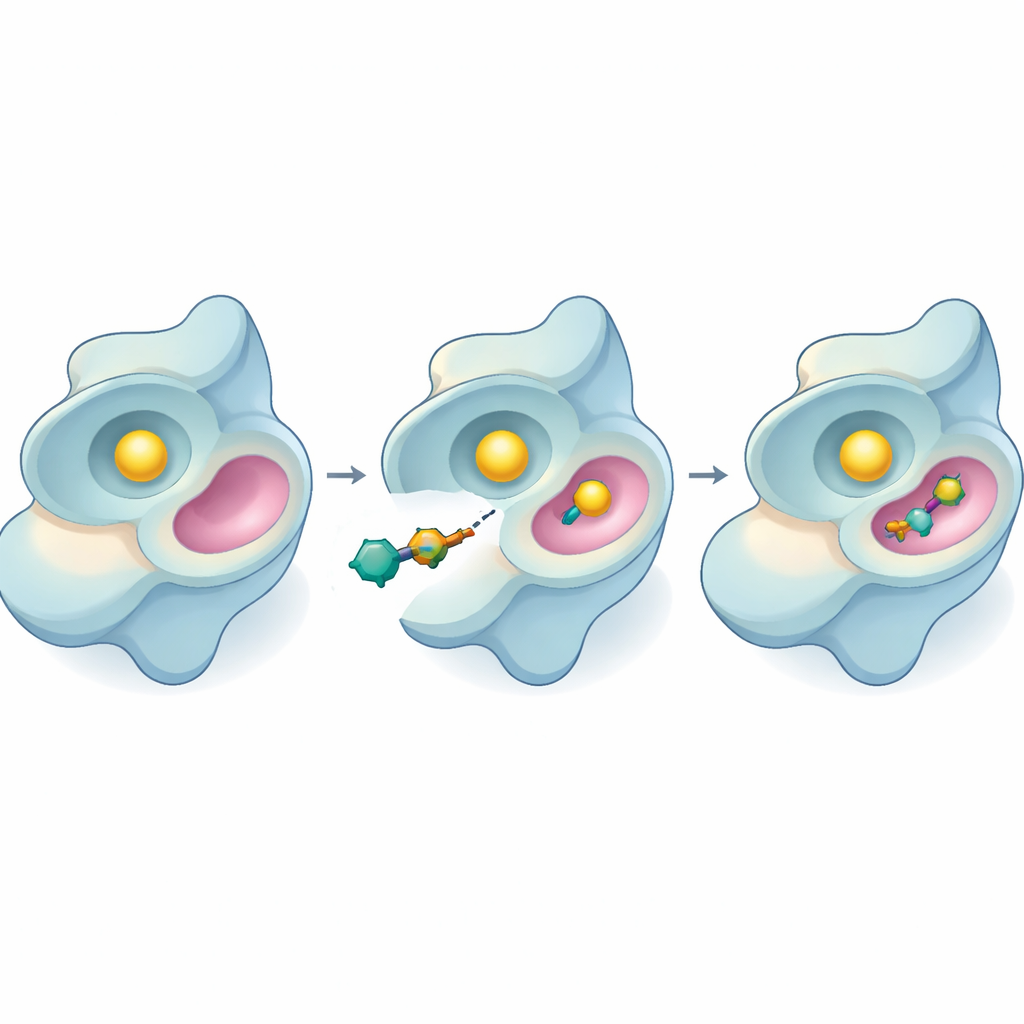
Putting the new inhibitors to the test
The team then moved to laboratory experiments using a light-based assay that measures how actively SYK consumes its fuel molecule, ATP. Several of the new compounds slowed the enzyme, but 44 stood out again. It inhibited SYK at very low concentrations and became more effective the longer it stayed with the enzyme. By varying the amount of substrate present, the researchers could infer how 44 works: rather than competing directly with ATP at the main active site, it appears to bind at a different, allosteric site and change the enzyme’s behavior from a distance. This is important because allosteric inhibitors often maintain their effect even when ATP levels are high or when mutations alter the primary site.
What this could mean for future treatments
Taken together, the computer models and lab measurements paint a consistent picture: molecule 44 binds SYK strongly, holds the enzyme in a stable, less flexible state, and shuts down its activity through an indirect, non-competitive mechanism. For non-specialists, this means the researchers have identified a finely tuned chemical key that fits a lock critical to many immune-driven diseases, without simply jamming the main opening. While much work remains—especially tests in cells, animals, and broader panels of related enzymes—this scaffold offers a promising starting point for future drugs aimed at taming harmful inflammation and slowing certain cancers by targeting SYK.
Citation: Rajasheker, K.V., Pallavi, M.S., Singh, P. et al. Chemical synthesis of novel aminopyrimidin-4-yl-1H-pyrazole derivatives as spleen tyrosine kinase (SYK) inhibitors. Sci Rep 16, 8323 (2026). https://doi.org/10.1038/s41598-026-38719-w
Keywords: spleen tyrosine kinase, kinase inhibitors, cancer therapeutics, autoimmune disease, allosteric drugs