Clear Sky Science · en
Efficacy of artemether lumefantrine vs chloroquine for the treatment of Plasmodium Vivax infection in Pakistan
Why this malaria study matters now
Pakistan has seen a dramatic surge in malaria, especially infections caused by the Plasmodium vivax parasite, which can hide in the liver and come back months later. Doctors there often have to choose between different drug combinations without clear, local evidence on which ones best prevent the disease from returning. This study follows hundreds of adult patients in Karachi to compare two widely used front-line drugs—with and without an add‑on medicine designed to wipe out hidden parasites—to see which options keep people malaria‑free in the months after treatment.
Understanding a stubborn kind of malaria
Not all malaria works the same way. Plasmodium vivax often circulates in lower numbers in the blood, making it harder to spot on lab tests, and it can linger silently in the liver as “dormant” forms that reactivate later. A person who appears cured can suddenly fall ill again weeks or even years after a mosquito bite. In Pakistan, where recent floods and damaged health systems have helped malaria explode from about half a million reported cases to more than four million, this pattern of quiet relapse makes control especially difficult. Missed diagnoses and incomplete treatment can turn treated patients into continuing sources of infection for their families and communities.
The treatments doctors are weighing
Pakistan’s treatment guideline for P. vivax calls for three days of chloroquine to clear parasites from the blood, followed by 14 days of primaquine to kill the dormant liver forms and reduce the chance of relapse. Another strong option for blood‑stage malaria is the artemisinin‑based combination artemether–lumefantrine, commonly used when the precise malaria species is unknown or when both P. vivax and P. falciparum may be present. However, shortages of chloroquine and primaquine, together with concern about drug resistance, have pushed many doctors to rely more heavily on artemether–lumefantrine alone. Until now, there has been limited long‑term, real‑world data from Pakistan comparing these approaches for P. vivax.
How the study was carried out
Researchers at a large hospital in Karachi followed 354 adults with confirmed P. vivax malaria from late 2023 to mid‑2024. Patients did not receive treatments at random; instead, doctors chose one of four common regimens based on routine practice: chloroquine alone, artemether–lumefantrine alone, chloroquine plus primaquine, or artemether–lumefantrine plus primaquine. All patients were examined repeatedly during the first month and then monthly for up to six months. At each visit, staff checked symptoms, did blood tests to look for returning parasites, and monitored simple safety measures such as hemoglobin levels and common side effects like headache or nausea.
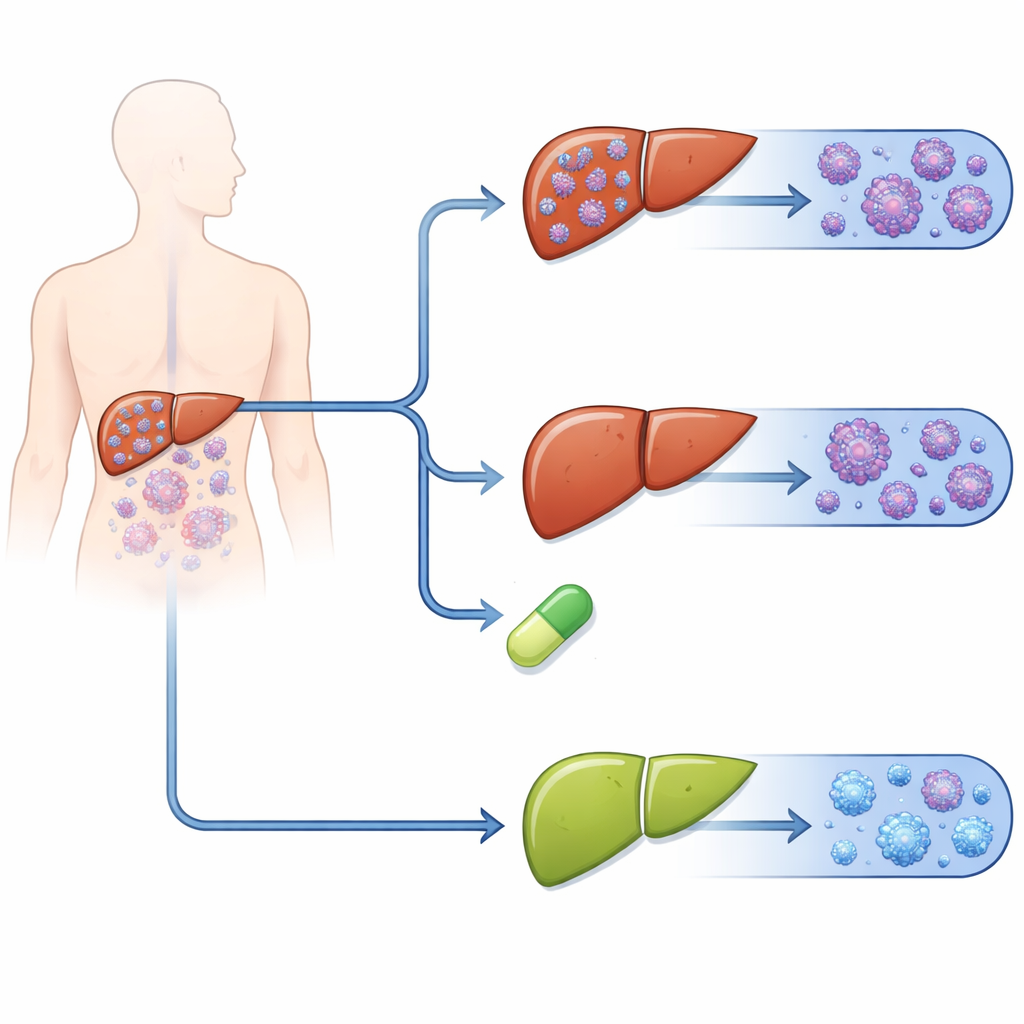
What happened over six months
Across the first 28 days, malaria returned most often in people who received artemether–lumefantrine alone and least often in those who also received primaquine. By six months, about one‑third of patients on artemether–lumefantrine alone had another P. vivax infection, compared with about one in six on chloroquine alone. In striking contrast, recurrences were rare among patients who took primaquine: just over 1% with chloroquine plus primaquine and about 6% with artemether–lumefantrine plus primaquine. When the authors analyzed how quickly recurrences appeared, people on artemether–lumefantrine alone were more than twice as likely to experience a new P. vivax episode as those on chloroquine alone. Adding primaquine substantially lowered the likelihood of a repeat infection, no matter which main drug was used to clear parasites from the blood.
Safety, limits, and what remains uncertain
The treatments were generally well tolerated. No serious drug‑related events were reported, and common complaints such as headache, vomiting, and abdominal pain occurred at similar rates in all groups. Before giving primaquine, the team screened patients for a genetic enzyme deficiency that can make some people vulnerable to red blood cell damage from this drug, and anyone at higher risk was excluded. Still, the study has important caveats: patients were not randomly assigned to treatments, which means hidden differences between groups may have influenced who relapsed; genetic tests were not done to separate true relapses from new infections; and drug levels were not measured, so the impact of missed doses or incomplete courses remains unclear.
What this means for patients and policy
For lay readers, the core message is this: in this Pakistani hospital, the long‑term return of P. vivax malaria was less common after chloroquine than after artemether–lumefantrine when each was used alone, and it was far less common when either drug was paired with the liver‑targeting medicine primaquine. The findings do not prove that chloroquine always works better, nor do they rule out the possibility of emerging resistance. But they strongly reinforce that tackling the hidden liver stages of P. vivax is crucial if the goal is to keep patients from getting sick again. As Pakistan considers newer one‑dose options to replace a two‑week primaquine course, this study underscores that any future strategy must both fit real‑world conditions and reliably clear the parasite’s lingering forms to prevent a revolving door of recurrent malaria.
Citation: Khan, S., Muqtadir, J., Abbas, S.A. et al. Efficacy of artemether lumefantrine vs chloroquine for the treatment of Plasmodium Vivax infection in Pakistan. Sci Rep 16, 7978 (2026). https://doi.org/10.1038/s41598-026-38713-2
Keywords: Plasmodium vivax, malaria treatment, Pakistan, primaquine, chloroquine vs artemether-lumefantrine