Clear Sky Science · en
Association of EGFR and EGF gene polymorphisms with cervical cancer in a case–control study and cross-cancer meta-analysis
Why tiny changes in our DNA matter
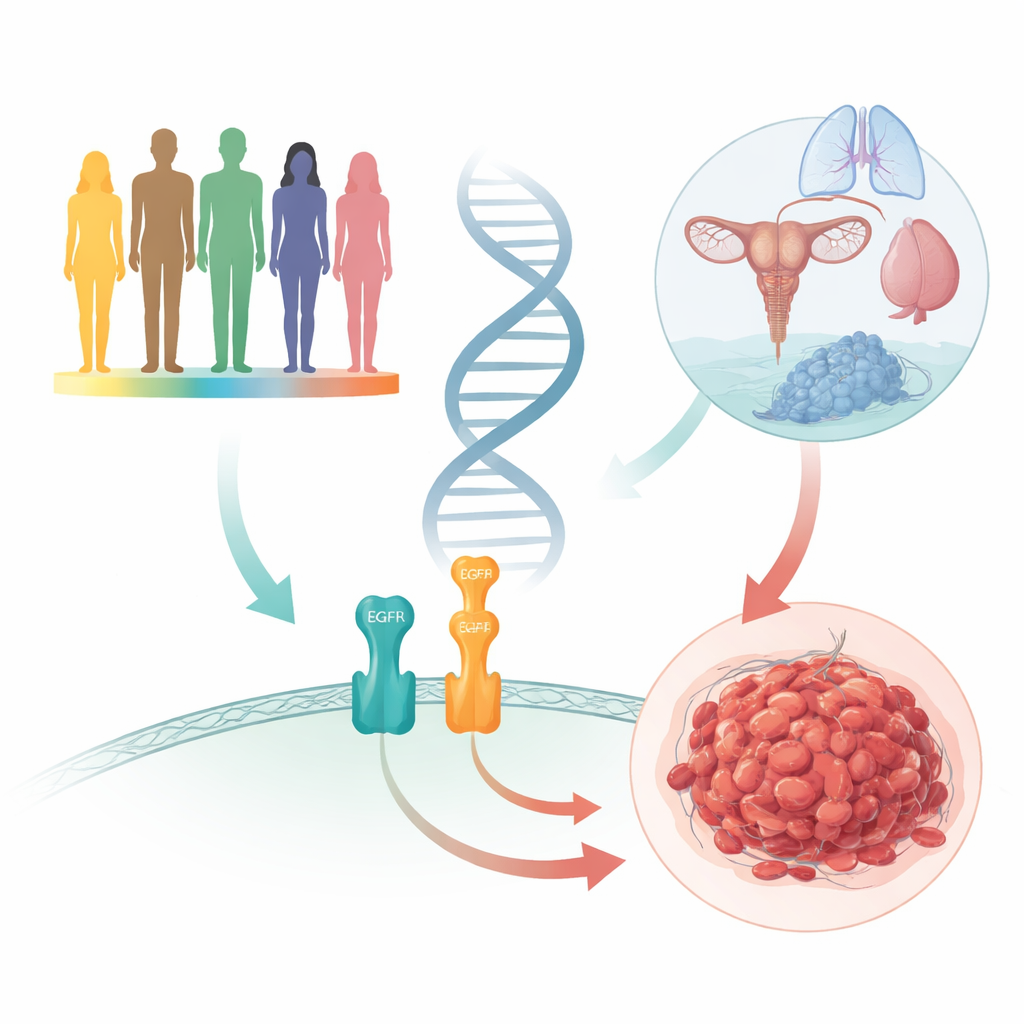
Cervical cancer remains a major threat to women’s health worldwide, even in the era of HPV vaccines and screening. Yet not everyone exposed to the same virus or environment develops cancer, suggesting that inherited differences help tip the balance. This study examines how small DNA variations in two closely linked players in cell growth—epidermal growth factor (EGF) and its receptor (EGFR)—shape the risk of cervical cancer and other tumors, especially in Asian populations.
From growth signals to cancer risk
EGF and EGFR form a kind of cellular on–off switch for growth. When EGF binds to EGFR on the surface of a cell, it can trigger chains of signals that tell cells to divide, repair damage, or migrate. In many cancers, this switch gets stuck in the "on" position. The authors focused on tiny DNA differences called single nucleotide polymorphisms, or SNPs, in the genes that encode EGF and EGFR. These SNPs do not rewrite the whole gene; instead, they subtly alter how much protein is made, how stable it is, or how strongly it signals—changes that, over a lifetime, may influence whether a cell becomes malignant.
A focused look at Chinese women
The team first carried out a case–control study in nearly two thousand Chinese women. They compared nine SNPs in the EGF and EGFR genes between 868 patients with cervical cancer and 1,066 healthy controls. The cancer cases included both common squamous tumors and less common gland-type tumors, and they spanned early to advanced stages of disease. After careful statistical corrections to avoid false positives, none of the individual SNPs showed a clear difference between women with cervical cancer and those without. Nor did these DNA changes line up with tumor type or clinical stage, suggesting that, on their own, these variants are not major drivers of cervical cancer in this population.
When gene variants act together
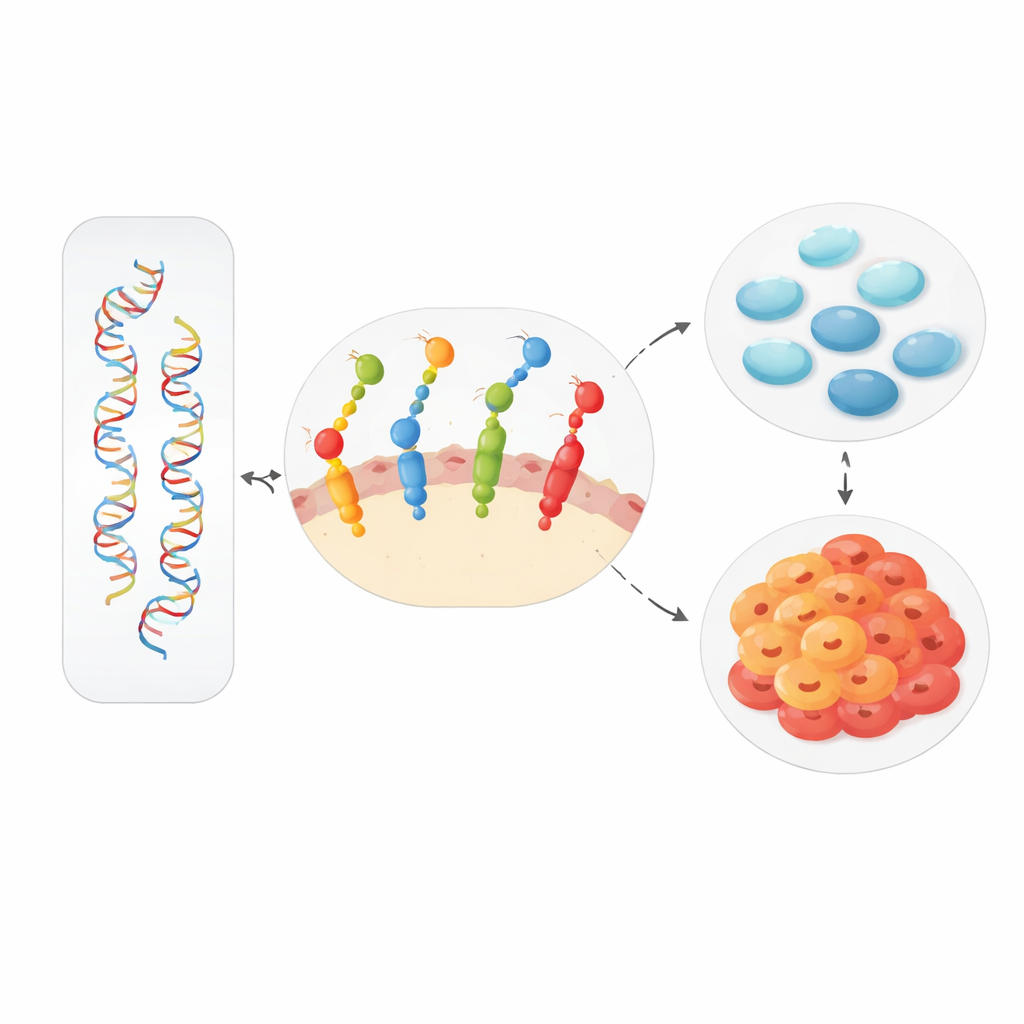
Although single SNPs looked unremarkable, the picture changed when combinations were examined. Using a method designed to detect interactions between multiple genetic factors, the researchers found evidence that a specific pair of variants—one in EGFR (rs1050171) and one in EGF (rs2237051)—was linked to cervical cancer risk. Laboratory and public gene-expression data suggest a plausible explanation: one variant tends to increase the amount of EGFR, while the other can reduce the stability of EGF. Together, they may shift how long and how strongly growth signals last, subtly rewiring the balance between normal repair and runaway cell division in cervical tissue.
Clues from many cancers around the world
To see whether these genetic patterns were unique to cervical cancer or part of a broader theme, the authors combined their data with results from dozens of previous studies across many cancer types. This meta-analysis pulled in more than 10,000 patients and 14,000 controls for some SNPs, spanning populations from Asia, Africa, Europe, and the Americas. Several EGF and EGFR variants—especially those known as rs2237051, rs3733625, and rs4444903—repeatedly tracked with overall cancer risk, but the direction and strength of the effect often depended on ancestry. A change that seemed to protect against cancer in one population could increase risk in another, highlighting how genetic background and environment interact in complex ways.
Shared genetic patterns across tumors
Looking across cancers, the researchers again used interaction analysis to ask whether combinations of these nine SNPs worked together in a consistent way. They found that multi-SNP models—especially one built from eight of the variants—were better at distinguishing cancer patients from controls than any single SNP alone. This suggests that EGF and EGFR variants may not act in isolation, but as part of a flexible network that can be pushed toward malignancy in multiple organs. The same pairs of interacting variants that appeared in cervical cancer also emerged in this cross-cancer view, hinting at underlying pathways that are shared across different tumor types.
What this means for patients and prevention
For the individual woman, these findings do not yet translate into a simple genetic test for cervical cancer risk. In this Chinese cohort, no single EGF or EGFR variant was a strong predictor of disease, and the combined effects remain modest. However, the work does underscore that inherited differences in growth-factor signaling can shape cancer susceptibility, and that these effects depend heavily on ancestry and on how variants interact. As larger and more diverse studies accumulate, such interaction patterns could one day refine risk models, inform targeted screening, or guide the use of drugs that block EGFR signaling. For now, HPV vaccination and regular screening remain the most powerful tools, while genetics offers a deeper glimpse into why some people are more vulnerable than others.
Citation: Shi, Y., Yao, Y., He, F. et al. Association of EGFR and EGF gene polymorphisms with cervical cancer in a case–control study and cross-cancer meta-analysis. Sci Rep 16, 9051 (2026). https://doi.org/10.1038/s41598-026-38712-3
Keywords: cervical cancer genetics, EGFR, EGF, cancer risk variants, Asian populations