Clear Sky Science · en
Two-component dynamics in supercritical $$\text {CO}_2$$ from inelastic X-ray scattering
Why this strange state of matter matters
Most of us think of carbon dioxide as a simple gas in the air or a liquid held under pressure, but when CO2 is pushed beyond its usual boiling and condensation limits it enters a "supercritical" state with properties unlike any familiar fluid. This exotic form of matter is already used for decaffeinating coffee, making polymers and potentially storing captured carbon underground. Yet, at the microscopic level, scientists still struggle to explain how molecules move and interact in this regime. This study reveals that in supercritical carbon dioxide, the fluid behaves as if it has two intertwined personalities at once—one gas-like and one liquid-like—and ties that split behavior to tiny, ever-changing clusters of molecules.
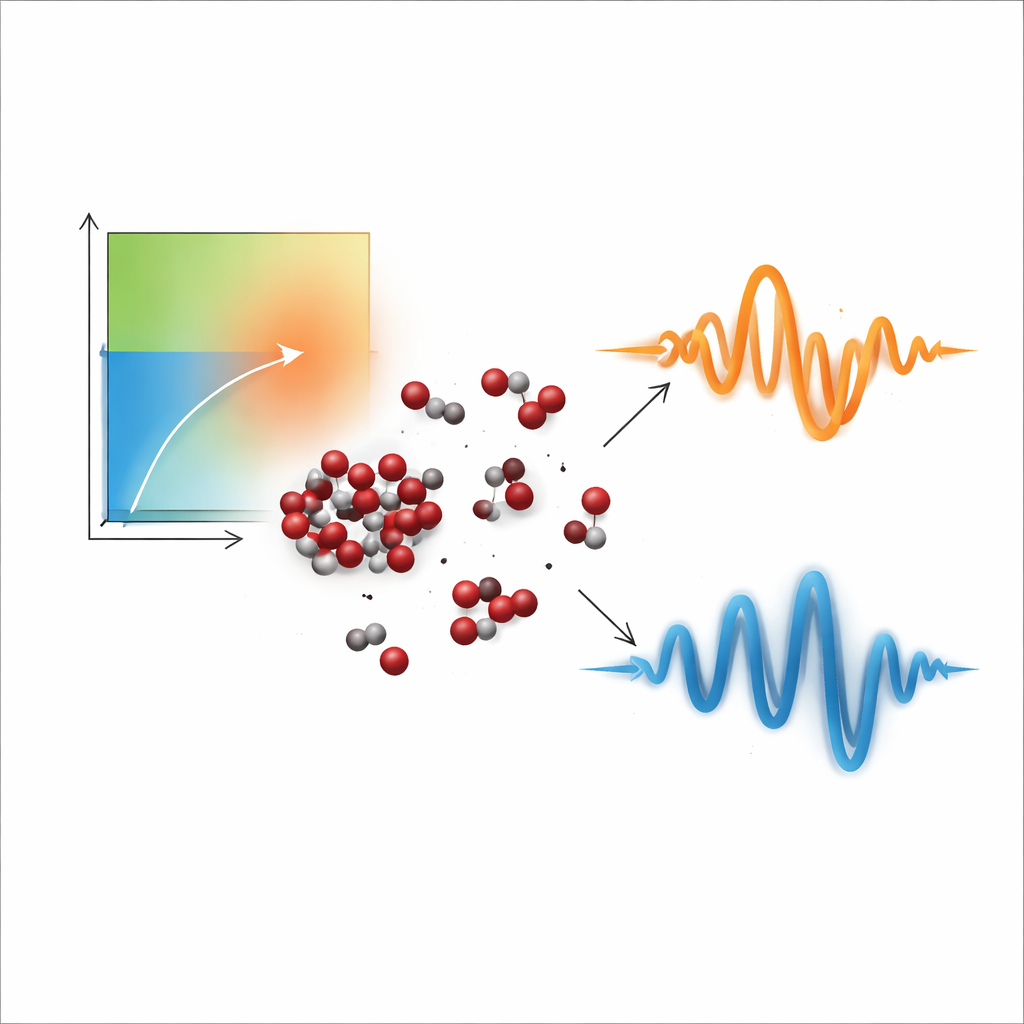
A fluid that is neither liquid nor gas
Above a certain pressure and temperature, a substance crosses its critical point and becomes a supercritical fluid. In this regime, there is no sharp boundary between liquid and gas, but scientists still distinguish more "liquid-like" and more "gas-like" regions of the phase diagram using markers such as the Widom line, where many fluid properties show strong variations. Supercritical carbon dioxide is especially important for technologies such as underground carbon storage, where CO2 may remain supercritical for long periods. Earlier X-ray and neutron experiments had hinted that even in this seemingly uniform state, the fluid contains microscopic patches of higher density—clusters where molecules briefly huddle together—raising the question of how these hidden structures influence how the fluid flows and vibrates.
Listening to molecular motion with X-rays
To probe this hidden world, the researchers used inelastic X-ray scattering, a technique that fires highly energetic X-rays through supercritical CO2 and measures how much energy and momentum the X-rays gain or lose. These tiny shifts encode how density waves and vibrations travel through the fluid on nanometer length scales and trillionth-of-a-second time scales. Experiments at a synchrotron facility scanned a range of temperatures and pressures that cross from liquid-like to gas-like conditions around the Widom line. In parallel, large-scale molecular dynamics simulations of thousands of CO2 molecules reproduced the same conditions, allowing the team to compare measured spectra with computed ones and to see directly how the molecules move.
Two intertwined voices in one fluid
By analyzing the spectra in terms of the current correlation function—a measure of how momentum propagates through the fluid—the team found clear evidence that supercritical CO2 does not vibrate with a single acoustic mode, as a simple liquid would. Instead, it shows two distinct components: a low-frequency part that behaves like sound in a dilute gas and a higher-frequency part that resembles sound in a dense liquid. As temperature rises and the fluid becomes more gas-like, the high-frequency contribution fades while the low-frequency one grows stronger, with a rapid crossover near the Widom line. Using a model-free mathematical technique called non-negative matrix factorization, the authors separated these overlapping contributions and mapped how each one changes with wavelength and thermodynamic conditions.
Clusters as the source of the split behavior
The key question is what microscopic feature gives rise to this dual personality. The simulations allowed the researchers to identify and track molecular clusters, defined as groups of CO2 molecules that are temporarily bound together by their combined kinetic and potential energies. They found that the fraction of molecules inside such clusters is linearly related to the strength of the high-frequency component, while molecules that spend more time unbound contribute mainly to the low-frequency part. Trajectory analysis showed that molecules residing longer in clusters experience more frequent collisions and stronger momentum fluctuations, leading to faster vibrational responses. In contrast, isolated molecules travel farther between encounters, producing slower, more gas-like vibrations. This direct link between cluster residence time, collision rate and vibrational frequency provides a physical picture of how the two components emerge.
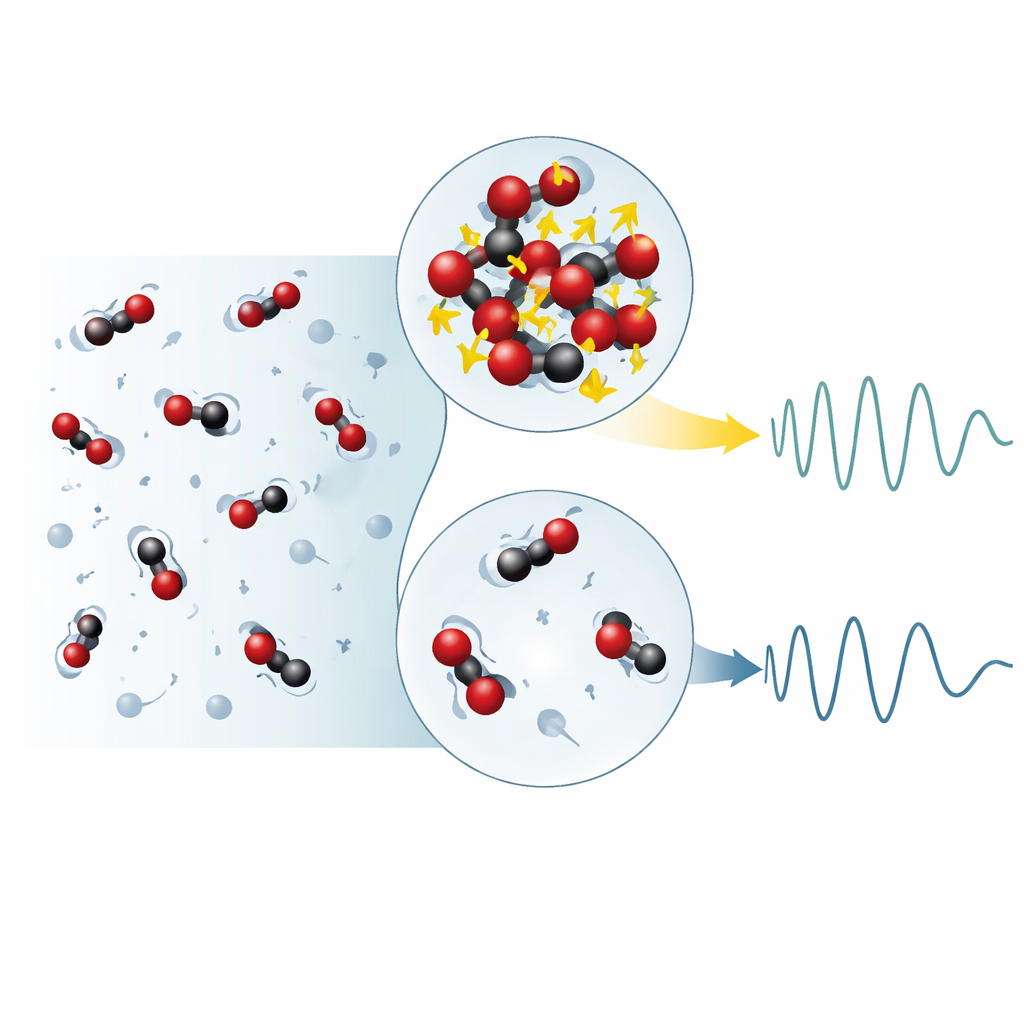
What this means for real-world fluids
The authors conclude that the two-component dynamics in supercritical CO2 arise from the coexistence of clustered and unbound molecules and their distinct patterns of motion. Because such clusters are a generic feature of supercritical fluids, this mechanism likely applies broadly, not just to CO2 but also to other substances, including water, that show similar dual acoustic behavior. Understanding how nanoscale structure controls vibrational and transport properties can improve models of industrial processes that rely on supercritical fluids and inform strategies for long-term carbon storage underground. More broadly, the work illustrates how even a seemingly simple fluid can hide rich and surprising behavior when pushed into extreme conditions.
Citation: Majumdar, A., Sun, P., Singleton, M. et al. Two-component dynamics in supercritical \(\text {CO}_2\) from inelastic X-ray scattering. Sci Rep 16, 8359 (2026). https://doi.org/10.1038/s41598-026-38697-z
Keywords: supercritical fluids, carbon dioxide, molecular clusters, X-ray scattering, fluid dynamics