Clear Sky Science · en
Validation of chemoresistance phenotypes in pleural mesothelioma across 2D, 3D, and in vivo models
Why some cancers shrug off chemotherapy
For people diagnosed with pleural mesothelioma—a cancer linked to asbestos exposure—chemotherapy is often the main treatment option. Yet many tumors barely respond, or they shrink at first and then quickly rebound. This study asks a deceptively simple question with big consequences: are the lab models researchers use to test drugs realistic enough to predict what actually happens inside a patient’s body?
From flat cell layers to tiny tumor balls
Most cancer drugs are first tested on cells grown in flat sheets on plastic dishes. These two-dimensional (2D) cultures are convenient, but they don’t resemble real tumors, which are three-dimensional (3D) masses of tightly packed cells surrounded by a complex neighborhood of blood vessels, scaffolding proteins, and immune cells. The researchers built a 3D “spheroid” model of mesothelioma, where cancer cells cluster into compact mini-tumors. They compared how mesothelioma cells of all major subtypes—epithelioid, biphasic, and the highly aggressive sarcomatoid—responded to standard chemotherapy when grown either as flat 2D layers or as 3D spheroids.
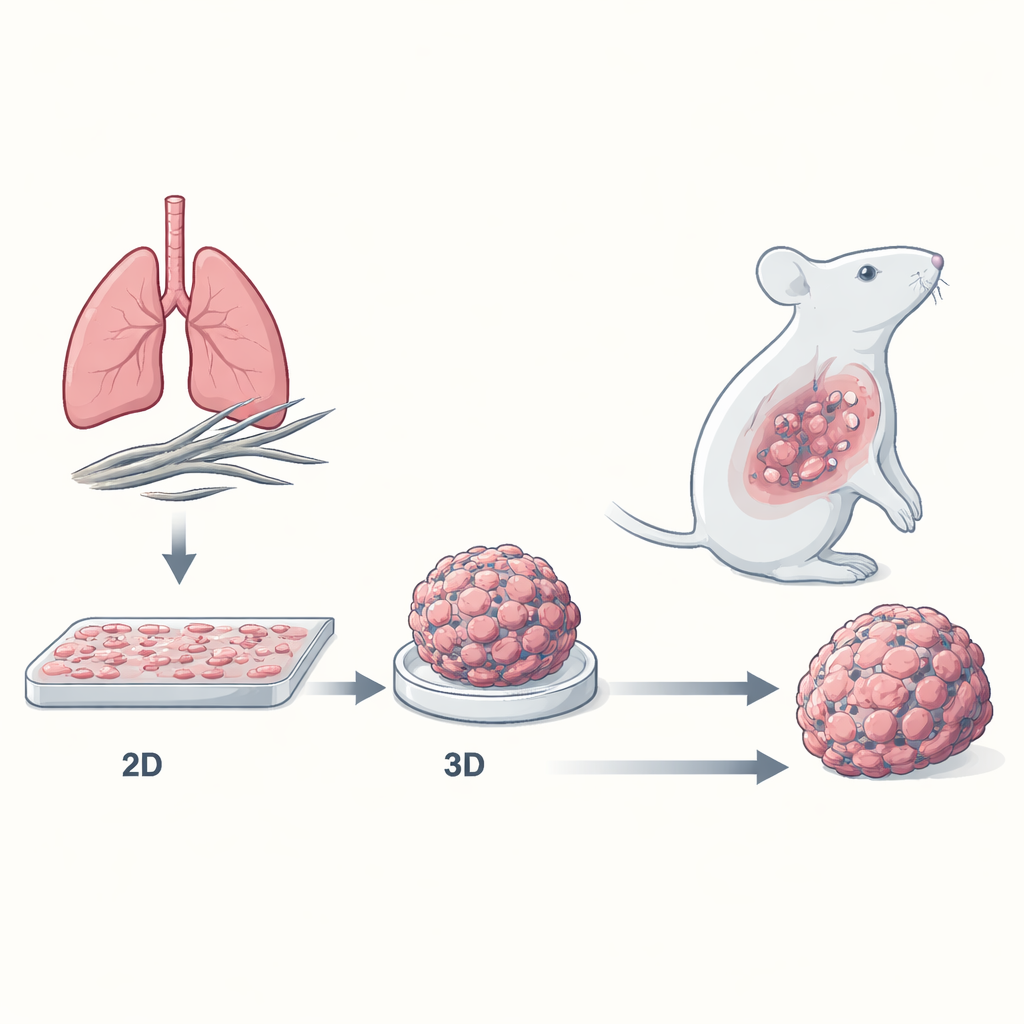
3D mini-tumors are far harder to kill
When exposed to the commonly used cisplatin–pemetrexed combination, mesothelioma cells grown as 2D layers were readily damaged: their growth slowed, many entered a halted “S phase” of the cell cycle, and large numbers underwent programmed cell death (apoptosis). In contrast, 3D spheroids barely shrank and required much higher drug doses to achieve similar effects. The sarcomatoid subtype, known in patients to be the hardest to treat, was also the most drug-resistant in the 3D model—mirroring real-world clinical behavior. Detailed measurements showed that, after treatment, 2D cells lost viability and progressed toward cell death, whereas cells in spheroids largely stayed alive, with only modest increases in early-stage apoptosis.
A quieter metabolism and survival-friendly signals
The team probed how energy use differed between the two models using a metabolic “stress test.” Chemotherapy pushed 2D cells from an active, energy-hungry state into a quieter mode, with sharp drops in mitochondrial oxygen consumption—evidence that the treatment was disrupting their internal power plants. By contrast, 3D spheroids already existed in a low-energy, oxygen-poor state that barely changed with treatment, similar to the stressed, hypoxic conditions seen inside real tumors. The researchers also measured small regulatory molecules called microRNAs and found subtype-specific patterns linked to drug resistance. Non-epithelioid spheroids, in particular, boosted microRNAs previously tied to poor prognosis and reduced cell death in other cancers. In tumors grown in mice from 3D spheroids, proteins involved in powerful survival pathways—PI3K/AKT and VEGF/Notch—were ramped up, further protecting cancer cells from dying.
Mouse tumors confirm what the dish predicts
To see whether these differences mattered in living organisms, the scientists implanted mesothelioma cells into mice either as single cells (mimicking 2D culture) or as pre-formed 3D spheroids. Tumors that started as spheroids grew larger and responded less to chemotherapy than those that began as single cells. Under the microscope, 3D-derived tumors showed dense bands of collagen and fibrous tissue, with more organized cell nests and less dead tissue. This fibrotic shell likely acts as a physical and biochemical barrier, limiting drug penetration and reinforcing survival signals—conditions that closely resemble stubborn human tumors.
What this means for future treatment
For a layperson, the central message is that how we grow cancer cells in the lab can make or break the realism of drug testing. Tiny 3D tumor spheroids recreate key features of mesothelioma that flat cell layers miss: oxygen-poor cores, stressed but hard-to-kill cells, protective scar-like tissue, and activation of survival pathways that block cell death. Because these 3D models behave much more like real tumors in patients and in mice, they offer a stronger platform for discovering drugs and for testing combinations that target both the cancer cells and their protective surroundings. In the long run, using such realistic models could help researchers identify treatments that stand a better chance of working in the clinic, not just in the lab.
Citation: Shi, H., Selvamani, S.P., Zelei, R. et al. Validation of chemoresistance phenotypes in pleural mesothelioma across 2D, 3D, and in vivo models. Sci Rep 16, 8396 (2026). https://doi.org/10.1038/s41598-026-38692-4
Keywords: pleural mesothelioma, chemotherapy resistance, 3D tumor spheroids, tumor microenvironment, cancer drug testing models