Clear Sky Science · en
Study on the differential expression of disulfidptosis-related genes and their association with immune regulation in patients with diabetic retinopathy
Why this eye disease matters to all of us
As diabetes becomes more common worldwide, one of its most feared complications is diabetic retinopathy, a condition that damages the light‑sensing tissue at the back of the eye and can lead to irreversible blindness. Doctors still struggle to predict which patients will lose vision and how best to protect them. This study explores a newly recognized form of cell death, called disulfidptosis, and asks whether related genes in the blood can flag early damage in the eye and reveal new ways to prevent sight loss.
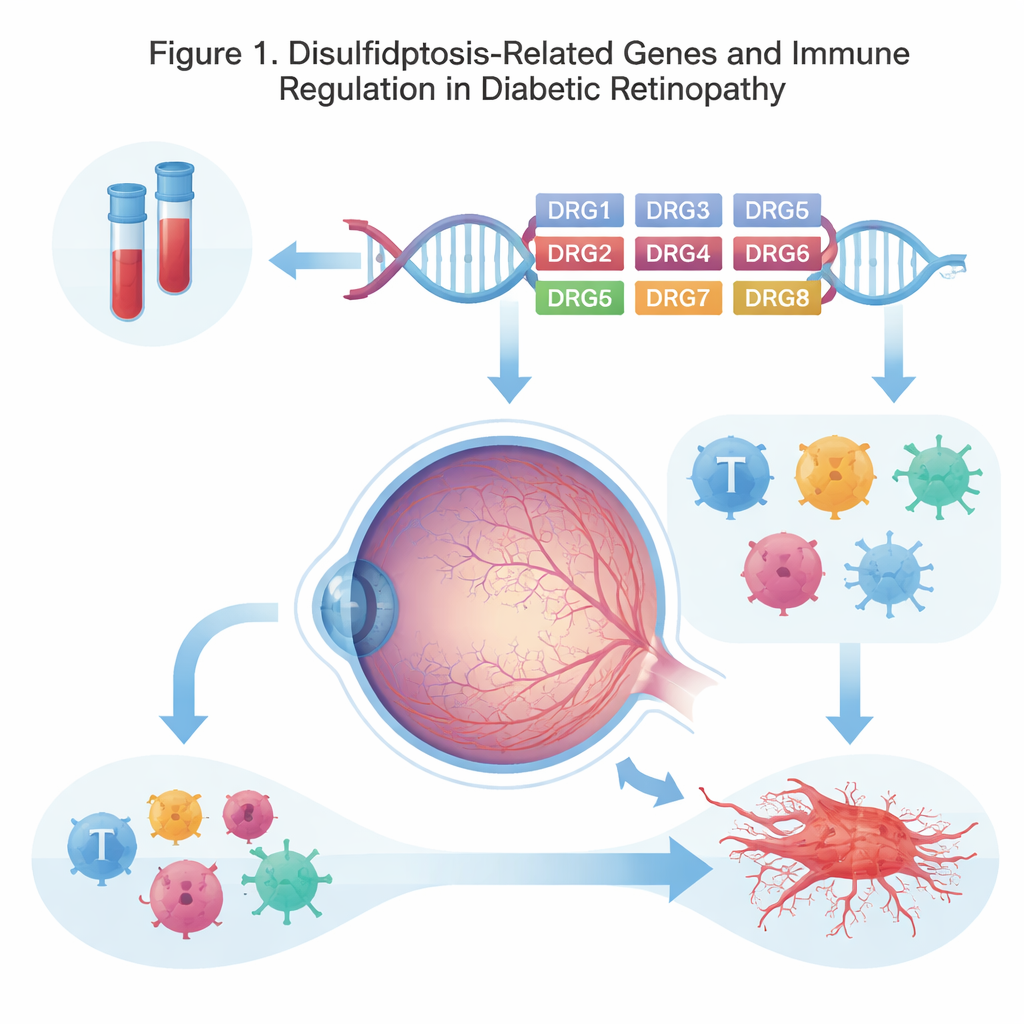
A new kind of cell breakdown
Our cells constantly repair and recycle themselves, and many diseases arise when these built‑in death programs go wrong. Researchers recently described disulfidptosis, a process in which certain chemical bonds inside structural proteins build up abnormally, destabilizing the cell’s inner scaffolding and leading to its collapse. Because diabetic retinopathy involves long‑term high blood sugar, oxidative stress, and fragile blood vessels in the retina, the authors suspected that disulfidptosis‑related genes might be involved in the slow damage that robs people of vision.
Hunting for warning signals in the blood
The team turned to a large public database of blood samples from people with and without diabetic retinopathy. They focused on 16 genes previously linked to disulfidptosis and compared how strongly these genes were switched on in patients with eye disease versus healthy volunteers. Eight genes stood out as consistently different, with several showing slightly higher activity and two showing lower activity in affected patients. The researchers then collected fresh blood samples from their own hospital patients and confirmed that the same genes, especially two called FLNB and GYS1, followed the same up‑or‑down pattern. Even though each gene changed only modestly, the changes were steady enough across many people to be meaningful.
How blood cells and platelets may damage the eye
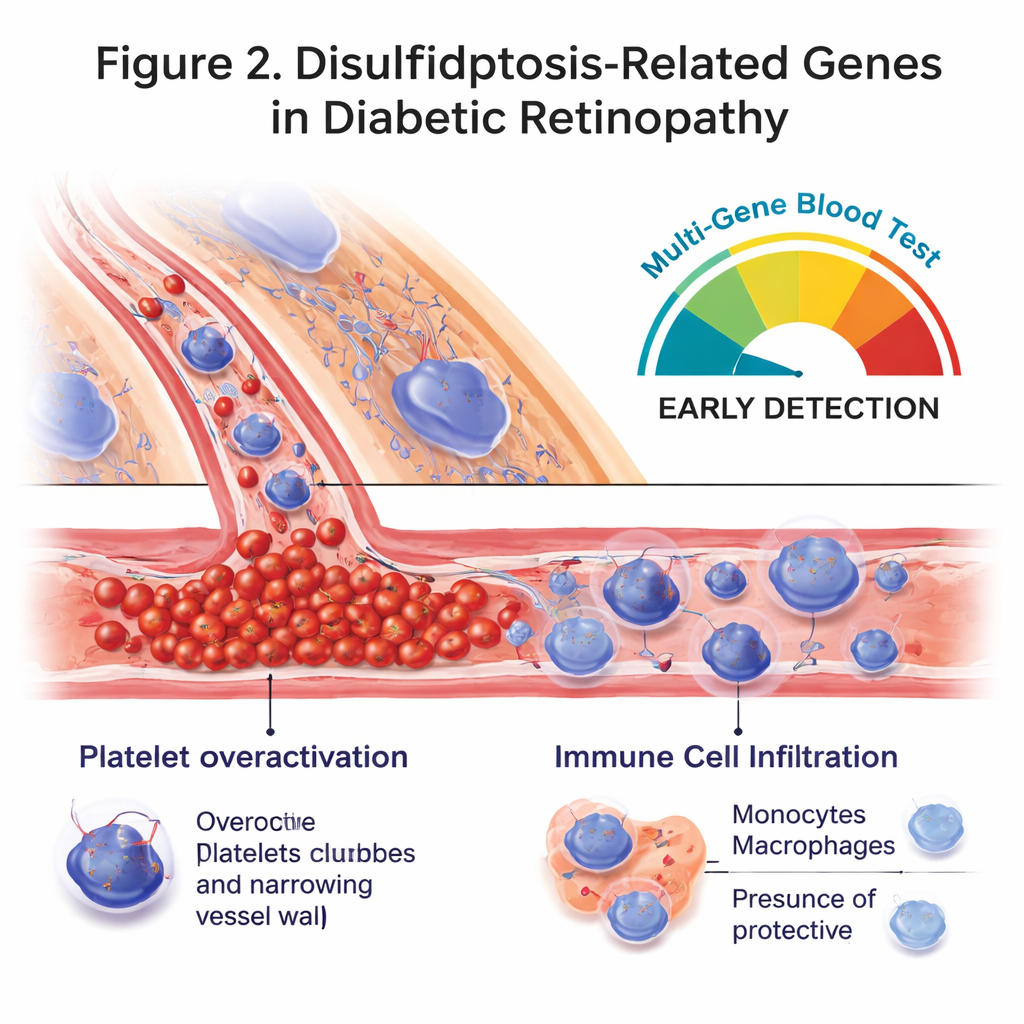
Next, the scientists asked what these genes might be doing. Using standard bioinformatics tools, they found that the altered genes were enriched in pathways related to blood clotting, platelet activation, and maintenance of normal circulation. These processes are central to diabetic retinopathy, where tiny capillaries in the retina become blocked, leaky, and then sprout fragile new vessels. Another group of pathways, including Rap1 and MAPK signaling, are known to control how tightly blood vessel cells stick together and how they respond to inflammatory stress. The study suggests that subtle shifts in disulfidptosis‑related genes could push platelets and vessel cells toward harmful behavior, contributing to blocked or weakened retinal vessels over time.
Immune cells and a blood‑based risk score
The authors also examined immune cell patterns in the same blood samples using computational methods. People with diabetic retinopathy had more circulating monocytes and early‑stage macrophages—cells that can amplify inflammation—and fewer activated memory T cells, which usually help coordinate a balanced immune response. Several of the disulfidptosis‑related genes were moderately linked to these immune shifts, hinting that they may help steer how the immune system behaves in diabetes. Building on these connections, the team combined all eight key genes into a single risk score using a statistical model. This multi‑gene “signature” distinguished patients with eye disease from healthy controls with fairly high accuracy, outperforming any single gene alone and offering a potential blueprint for a future blood test.
What this could mean for people with diabetes
For now, the work is mainly a proof of concept. The model was built on one public dataset and a relatively small hospital group, and the study did not directly show disulfidptosis happening in eye tissue. Still, it is the first to link this new cell‑death pathway to diabetic retinopathy and to propose a set of blood‑based genes connected to both blood vessel injury and immune imbalance. If confirmed in larger and more diverse groups of patients, these gene patterns could become part of a low‑cost blood test to identify individuals at high risk of vision loss while their eyes still appear healthy, opening the door to earlier monitoring and more personalized prevention strategies.
Citation: Hao, Y., Zhang, XX., Wang, XY. et al. Study on the differential expression of disulfidptosis-related genes and their association with immune regulation in patients with diabetic retinopathy. Sci Rep 16, 7654 (2026). https://doi.org/10.1038/s41598-026-38671-9
Keywords: diabetic retinopathy, disulfidptosis, biomarkers, immune cells, gene risk model