Clear Sky Science · en
Effect of sonication and protease inhibitors on Elisa quantification of selected proteins in bovine udder tissue homogenates
Why tiny milk proteins matter
Behind each glass of milk lies a complex factory inside the cow’s udder, where thousands of proteins work together to produce safe, nutritious milk. Scientists often measure these proteins to monitor udder health and understand diseases like mastitis, a costly infection in dairy herds. This study asks a deceptively simple question with big practical consequences: does the way we prepare tissue samples before a common lab test, called ELISA, quietly distort the results we trust to make decisions?
How lab handling can change what we see
To measure proteins, researchers first have to break open tissue and release its contents. In this work, scientists studied pieces of udder tissue from 22 cows, half healthy and half infected with a type of bacteria that can cause mastitis. They then measured three important proteins linked to milk production and udder health: alpha-casein (a major milk protein), lactoferrin (an immune-related protein), and alkaline phosphatase (an enzyme tied to tissue activity). All measurements were done with sandwich ELISA, a widely used method for detecting very small amounts of specific proteins. The twist was that the same tissue was processed four different ways before testing, to see how much the preparation alone could change the final numbers.
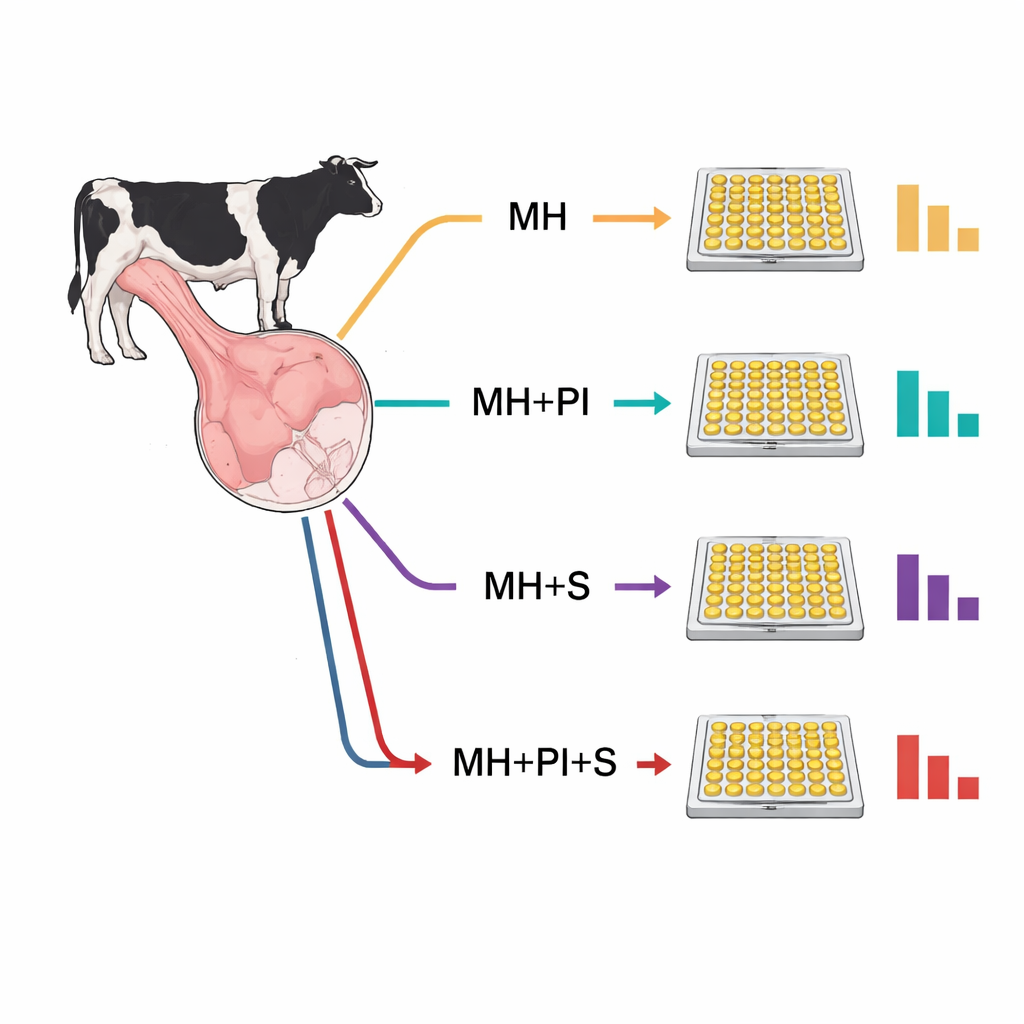
Four ways to treat the same tissue
The team compared a basic mechanical homogenization step—physically grinding the tissue in a liquid buffer—to three more elaborate versions. One added a cocktail of protease inhibitors, chemicals meant to block natural protein-cutting enzymes released when cells are broken. Another added sonication, bursts of high-frequency sound waves that further disrupt cells. The fourth combined both protease inhibitors and sonication. In principle, these extra steps should either protect or better release the proteins of interest. In practice, they produced the opposite effect: every added step led to lower measured protein levels compared with homogenization alone, and the combination of inhibitors plus sonication produced the lowest readings for all three proteins.
Some proteins shrink more than others
The reductions were not equal across proteins. Alpha-casein was the most resilient, showing the smallest drop when extra processing steps were used. Lactoferrin and alkaline phosphatase, by contrast, fell much more sharply, especially when sonication was involved. This pattern suggests that certain proteins are more vulnerable to physical disruption, heat, or chemical interference, even when protocols are carefully cooled and standardized. The cows’ health status also played a role, but only for alpha-casein: udders infected with coagulase-positive staphylococci showed higher levels of this milk protein, while lactoferrin and alkaline phosphatase did not differ clearly between healthy and infected animals. Crucially, however, the direction and size of the processing-related drops were similar in both healthy and infected groups.
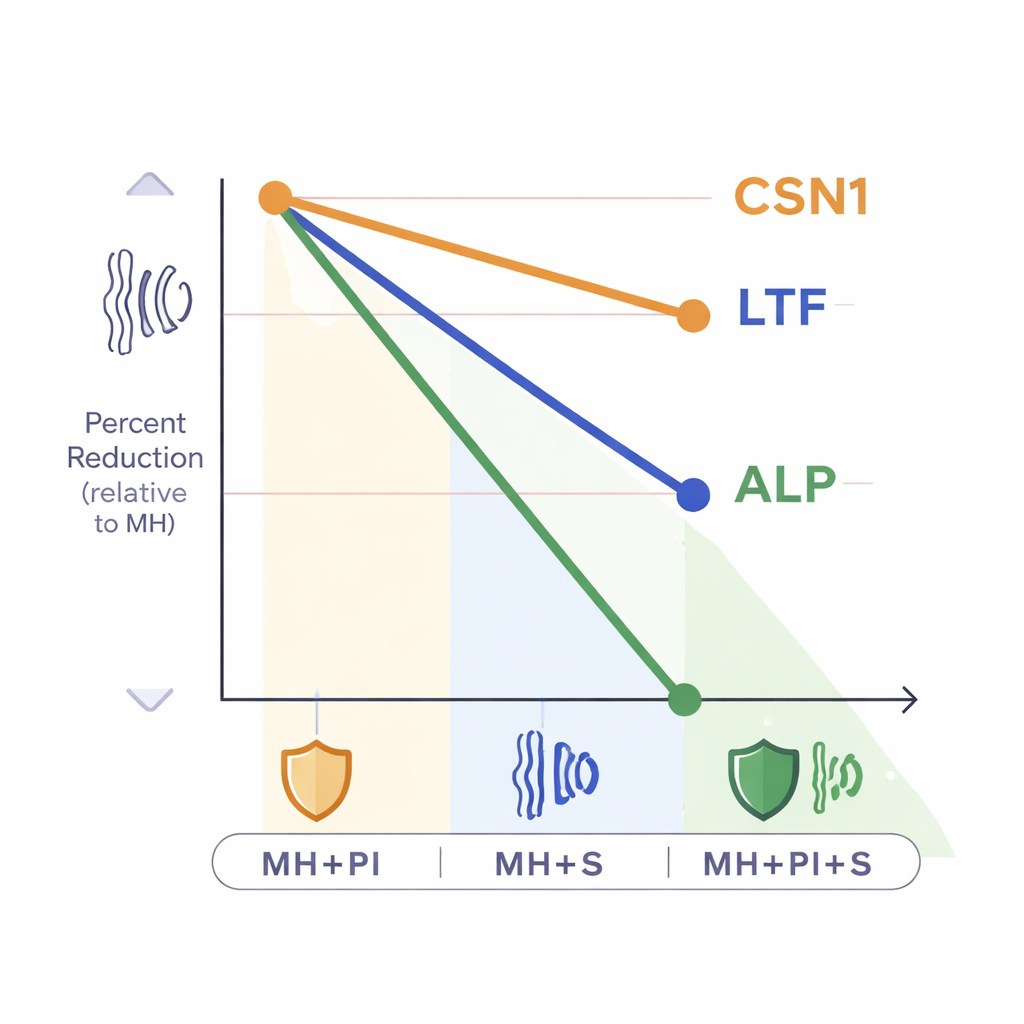
When protection backfires
Why would steps designed to help—like protease inhibitors and sonication—lead to lower measured protein levels? The study did not dissect the exact molecular reasons, but it points to several possibilities. Sonication can generate heat and strong physical forces that partially damage proteins or alter the specific parts recognized by ELISA antibodies. Protease inhibitors, while blocking natural enzymes, may themselves interfere with the assay by binding nonspecifically or affecting detection chemistry. Whatever the mechanism, the message is clear: these widely recommended add-ons can systematically depress ELISA readings, and they do so to different degrees for different proteins.
What this means for milk research and diagnostics
For farmers, veterinarians, and food safety experts, ELISA results guide decisions about animal health, milk quality, and processing safety. This study shows that simply choosing a more complex preparation protocol can make target proteins appear markedly scarcer than they really are. In bovine udder tissue, the plainest method—mechanical homogenization alone—actually yielded the highest and most comparable protein concentrations. For non-specialists, the takeaway is straightforward: before interpreting protein numbers as a sign of disease, milk quality, or treatment success, researchers must carefully standardize how samples are handled. Otherwise, differences in test results may reflect the roughness of the lab’s preparation steps more than the true biology inside the cow.
Citation: Szprynca, A., Czopowicz, M., Zalewska, M. et al. Effect of sonication and protease inhibitors on Elisa quantification of selected proteins in bovine udder tissue homogenates. Sci Rep 16, 7366 (2026). https://doi.org/10.1038/s41598-026-38653-x
Keywords: ELISA sample preparation, bovine mastitis, udder tissue proteins, sonication effects, protease inhibitors