Clear Sky Science · en
Photoinduced proton transfer in differently structured water: an EPR approach to solving a classic problem
Why the Hidden Journeys of Protons Matter
Every time you breathe, think, or convert sunlight into useful energy in a solar cell, tiny positively charged particles called protons are on the move. How these protons travel through water is a centuries‑old puzzle, first posed by the scientist Theodor Grotthuss in 1806. This new study uses a clever combination of light and magnetic measurements to watch protons move in water with different structures, from salty and crowded solutions to icy, glassy mixtures and even inside a membrane protein. The work offers a fresh way to test classic ideas about proton motion and to probe environments that are hard to study by other methods.
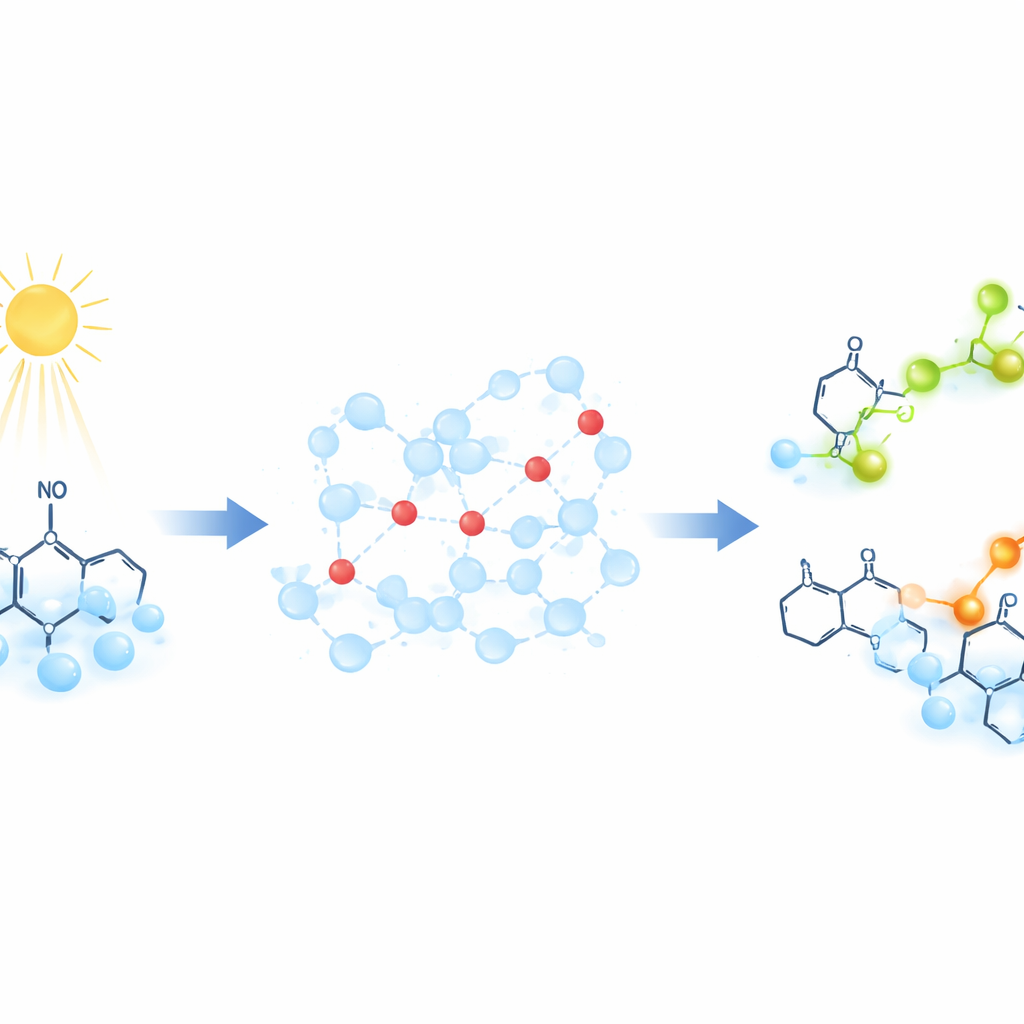
A New Way to Watch Protons Move
Instead of following protons directly, the researchers use special “probe” molecules that change their magnetic signature when they pick up a proton. These probes are stable organic radicals whose unpaired electron produces a well‑defined signal in electron paramagnetic resonance (EPR), a technique that detects how unpaired electrons respond to a magnetic field. To launch proton motion on demand, the team shines light on a compound called 2‑nitrobenzaldehyde. This light‑driven reaction releases a burst of protons in less than a billionth of a second, suddenly lowering the pH of the solution. As protons spread through the water and bind to the probes, the EPR signal shifts in a time‑dependent way, allowing the scientists to track how fast proton transfer occurs.
How Water’s Neighbors Slow or Speed the Flow
Water rarely exists alone; it is usually crowded with dissolved salts or other molecules that subtly rearrange its network of hydrogen bonds. The team tested how such additives affect proton motion by comparing pure water to solutions containing potassium chloride, urea, or guanidinium hydrochloride, all at high concentrations. By fitting the EPR changes to simple kinetic models, they extracted apparent rates for proton transfer. They found that in 6 molar guanidinium hydrochloride, proton transfer was about 40 times slower than in pure water, whereas 8 molar urea caused only a modest slowdown, and potassium chloride produced intermediate effects. Because the rate at which protons react with the probes is limited by how they move through the hydrogen‑bond network, these differences point to substantial changes in how water is structured around the added ions and molecules.
Proton Motion in Glassy Ice and Inside Proteins
The method is not limited to ordinary liquid water. The authors also studied a water–glycerol mixture cooled to 160 kelvin, where it forms a rigid glass. Free diffusion of molecules is essentially frozen, yet the EPR signal still showed that the probes were being protonated after light exposure. This suggests that proton transfer can proceed without classical diffusion, possibly through quantum tunneling along a chain of hydrogen bonds, echoing modern refinements of the Grotthuss idea. To explore biological relevance, the team attached a pH‑sensitive radical to a specific site on the membrane protein bacteriorhodopsin, at the boundary between the protein and surrounding water. When they triggered proton release with light, they observed a time‑dependent EPR response from this labeled site, demonstrating that their approach can monitor proton delivery to a defined position on a protein surface.
Connecting Structure, Water, and Proton Highways
To better understand why the protein‑bound probe behaved much like one in bulk water, the researchers ran computer simulations of bacteriorhodopsin in a membrane and of a small peptide in solution. They calculated how water molecules surround the nitroxyl group of the spin label and found nearly identical local hydration patterns in both cases, with only mild shielding by the membrane environment. This means that, at least for the studied site, protons have access through a water layer that closely resembles ordinary liquid water. The authors also show that their probes can be used in time‑resolved EPR experiments, opening the door to tracking both proton and electron motion together in reactions where the two are tightly coupled.
What This Work Tells Us in Simple Terms
In essence, this study turns light into a sharp starting pistol for proton motion and then uses sensitive magnetic probes to watch how quickly those protons reach different targets. By comparing simple salt solutions, dense denaturants, glassy mixtures, and a membrane protein, the researchers demonstrate that proton flow is strongly shaped by how water molecules are arranged and by the surrounding environment. Their approach not only supports the idea that protons hop along networks of hydrogen bonds, but also reveals that this hopping can persist even when ordinary molecular motion is nearly frozen. This new toolkit could help scientists dissect how protons move in enzymes, energy‑conversion systems, and novel materials, bringing us closer to controlling these tiny but powerful charge carriers.
Citation: Barbon, A., Savitsky, A., Grigoriev, I. et al. Photoinduced proton transfer in differently structured water: an EPR approach to solving a classic problem. Sci Rep 16, 7983 (2026). https://doi.org/10.1038/s41598-026-38650-0
Keywords: proton transfer, water structure, electron paramagnetic resonance, hydrogen bond network, bacteriorhodopsin