Clear Sky Science · en
DNMT3A p.R882C driven proliferation and anti-apoptotic effects in pancreatic cancer cells
Why this research matters for pancreatic cancer
Pancreatic cancer is notorious for being discovered late, spreading quickly, and responding poorly to treatment. Most patients are diagnosed with pancreatic ductal adenocarcinoma (PDAC), a particularly aggressive form of the disease. This study looks at a tiny change in a single gene, called DNMT3A, and asks a big question: can this subtle alteration help explain why some pancreatic cancers grow faster and resist cell death? Understanding such changes could open the door to earlier detection and more precise, targeted therapies.
A closer look at a deadly cancer
PDAC accounts for more than 90% of pancreatic cancers and has a five‑year survival rate of under 13%. It is usually driven by well‑known cancer genes such as KRAS and TP53, but many patients still do not benefit from current targeted treatments. Recent work suggests that not only gene mutations but also “epigenetic” changes—chemical markings on DNA that control which genes are turned on or off—play a key role in this cancer. DNMT3A is one of the main enzymes that writes these chemical marks, and its defects have already been linked to blood cancers like acute myeloid leukemia.
Hunting for dangerous DNA changes
The researchers collected tumor samples and nearby non‑cancerous tissue from three patients with moderately to poorly differentiated PDAC. They used whole‑exome sequencing, a technique that reads all the protein‑coding regions of the genome, and then applied strict computer filters to separate harmless DNA variations from those likely to damage cell behavior. From hundreds of alterations, they narrowed the list to 68 high‑risk changes. Among these, a rare variant in DNMT3A, known as p.R882C, stood out because it almost never appears in large population databases and was consistently flagged as harmful by multiple prediction tools.
What this mutation does inside cells
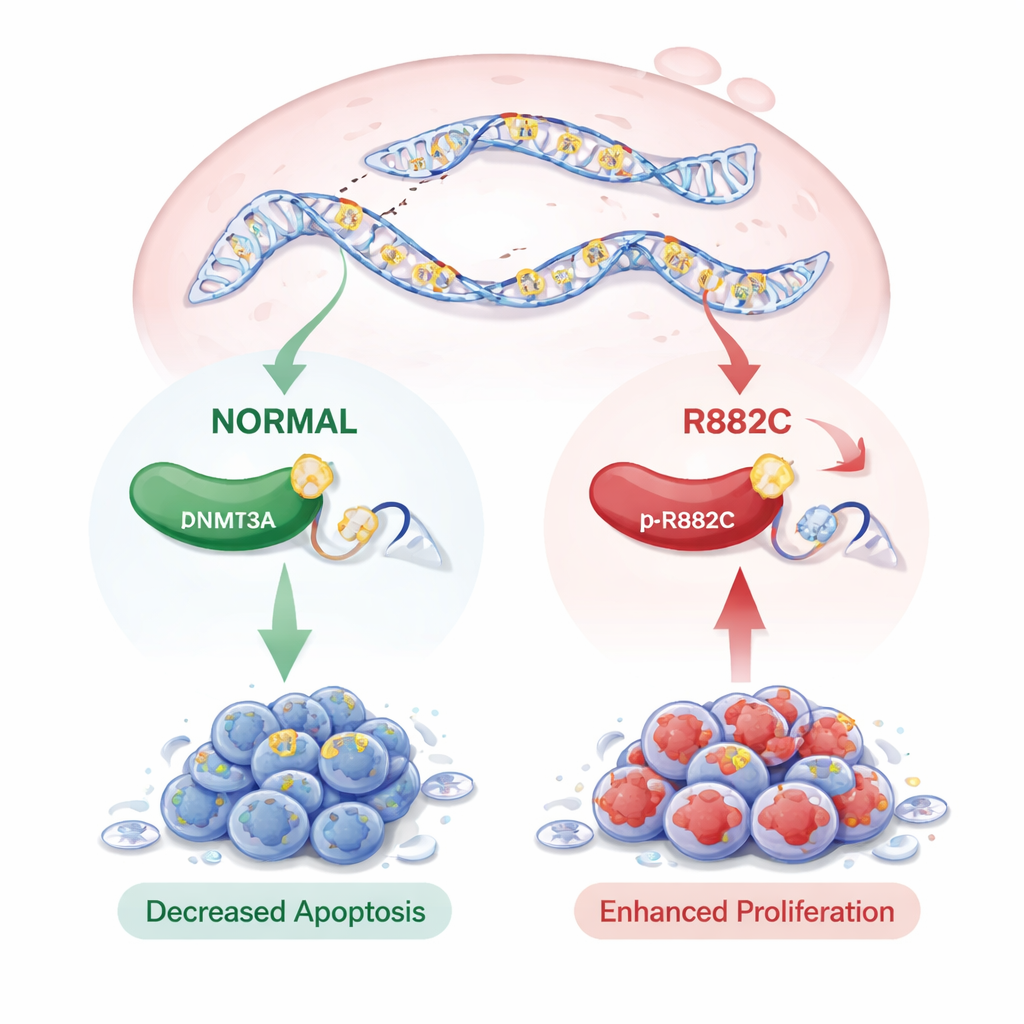
DNMT3A is a 912‑amino‑acid protein that helps establish DNA methylation patterns—chemical tags that act like “on” or “off” switches for genes. The p.R882C change sits in a crucial working region of DNMT3A, where it could disturb the enzyme’s activity. Using 3D computer models, the team found that the overall protein shape did not change dramatically, suggesting that the problem may lie in how the enzyme functions rather than how it folds. To test this, they engineered pancreatic cancer cell lines (PANC‑1 and PaTu 8988t) to produce either normal DNMT3A or the p.R882C version, and compared them with cells carrying an empty control vector.
Faster growth, more movement, less cell death
Surprisingly, the p.R882C mutation did not change how much DNMT3A was made at the RNA or protein level; the amount of enzyme stayed roughly the same. However, its presence had striking effects on cell behavior. In colony formation and growth assays, cells with the mutant DNMT3A formed more colonies and showed higher growth signals than those with the normal version. Wound‑healing and Trans‑well tests, which measure how quickly cells move, revealed that mutant‑bearing cells migrated more rapidly. When the team examined programmed cell death (apoptosis) using flow cytometry, cells with the p.R882C mutation had markedly fewer dying cells, both at early and late stages of apoptosis, indicating a strong survival advantage.
What this could mean for future care
Putting these findings together, the study suggests that the rare DNMT3A p.R882C mutation helps pancreatic cancer cells grow faster, move more easily, and resist self‑destruction—traits that can contribute to cancer progression. Although the mutation does not boost DNMT3A levels, it likely alters the enzyme’s activity and the pattern of DNA markings across the genome, echoing what has been seen in certain blood cancers. The authors note that this change is uncommon and did not yet show clear links to patient survival in public datasets, in part because available studies are small and often do not include DNMT3A. Still, uncovering this mutation in PDAC highlights a potentially important new player in the disease and raises the possibility that, in the future, DNMT3A p.R882C could serve as a marker for risk or a target for more tailored treatments.
Citation: Qu, Z., Mao, J., Qian, Y. et al. DNMT3A p.R882C driven proliferation and anti-apoptotic effects in pancreatic cancer cells. Sci Rep 16, 7659 (2026). https://doi.org/10.1038/s41598-026-38613-5
Keywords: pancreatic cancer, DNMT3A mutation, PDAC, DNA methylation, targeted therapy