Clear Sky Science · en
Activated T cells induce apoptosis in A549 lung adenocarcinoma cells via TRPV4-mediated calcium influx
Why this research matters for lung cancer
Lung cancer is still one of the deadliest cancers, in part because tumors often learn to resist standard treatments like chemotherapy and radiation. In recent years, harnessing the body’s own immune system has offered new hope. This study explores a fresh twist on that idea: it shows how a specific type of immune cell, called an activated T cell, can push lung cancer cells toward self-destruction by forcing them to take in calcium through a molecular “gate” known as TRPV4. Understanding this pathway could help scientists design more powerful and precise immunotherapies.
Soldiers of the immune system meet lung tumor cells
The researchers focused on human lung adenocarcinoma cells known as A549 cells, a widely used model for non-small cell lung cancer. They paired these cancer cells with Jurkat T cells, a stand-in for the body’s killer T cells, which normally seek and destroy infected or malignant cells. By chemically activating the Jurkat cells, the team mimicked the highly alert state of T cells engaged in a real immune response. They then mixed activated or inactive T cells with the lung cancer cells at different ratios and watched what happened over one to three days, paying special attention to how many cancer cells survived and how many underwent programmed cell death, or apoptosis.
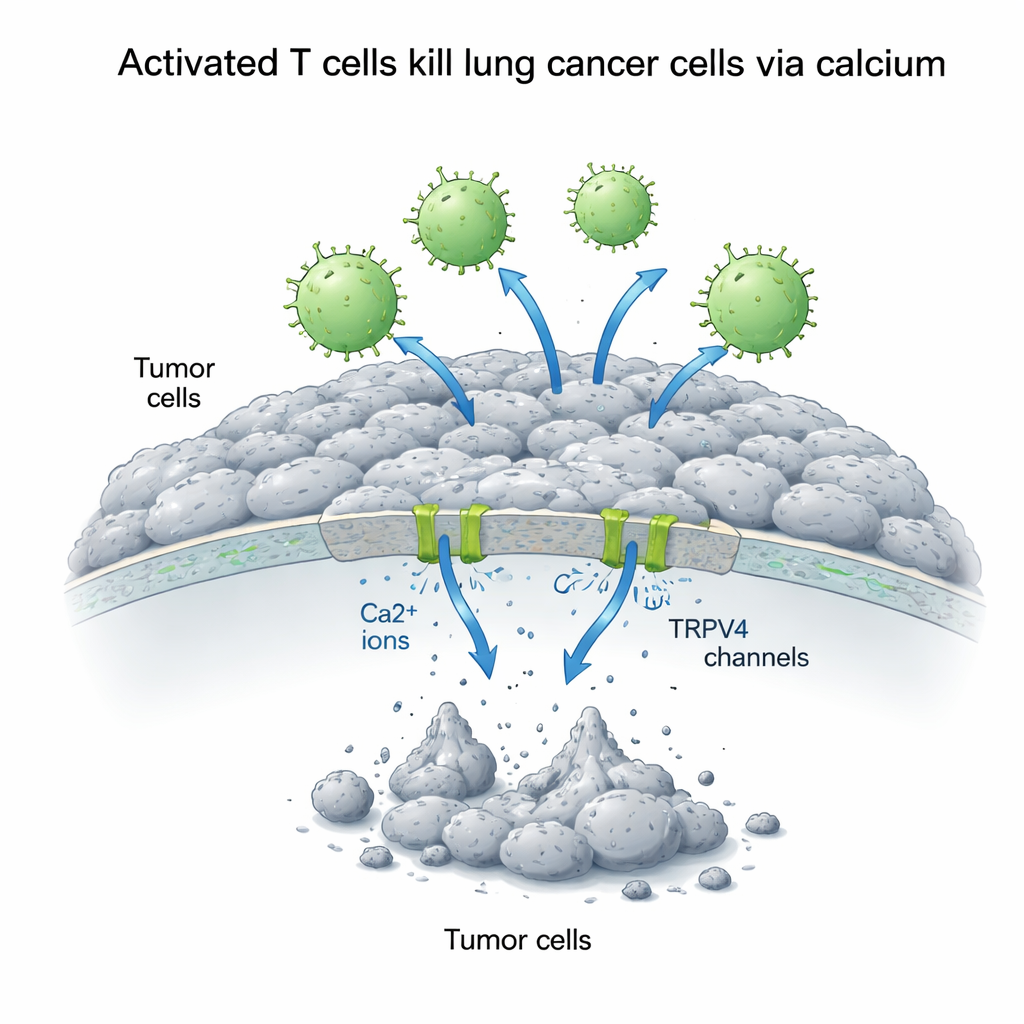
Calcium as a double-edged signal for cancer cells
Calcium inside cells acts like a versatile signal switch that can either promote growth or trigger death, depending on how it is handled. The scientists boosted the amount of calcium outside the A549 cells and measured how this affected the battle between T cells and tumor cells. Surprisingly, extra calcium alone made the lung cancer cells grow better, suggesting that under calm conditions calcium fuels survival pathways. But when activated T cells were present, the story flipped: higher external calcium dramatically strengthened T cell–driven killing. Over 72 hours, cancer cell survival plunged to less than a third of normal when both activated T cells and extra calcium were combined, whereas inactive T cells had little effect and could even support growth when calcium was added.
A calcium “gate” linked to stress and cell death
To understand how this switch from growth to death occurs, the team looked at TRPV4, a protein channel in the cell membrane that allows calcium to flow inside. Using protein analysis, they found that TRPV4 levels rose sharply when A549 cells were exposed to added calcium, whether or not T cells were present, and were highest when calcium and activated T cells were combined. At the same time, the cancer cells showed clear signs of oxidative stress: the balance tipped away from protective antioxidants (lower total antioxidant capacity) and toward harmful oxidants (higher total oxidant status). Flow cytometry, a technique that tags cells with fluorescent dyes, confirmed that activated T cells caused a surge in both early and late stages of apoptosis, especially when calcium was abundant. Together, these observations point to a chain of events in which TRPV4 channels admit more calcium, which then drives chemical stress inside the cell and pushes it toward death.
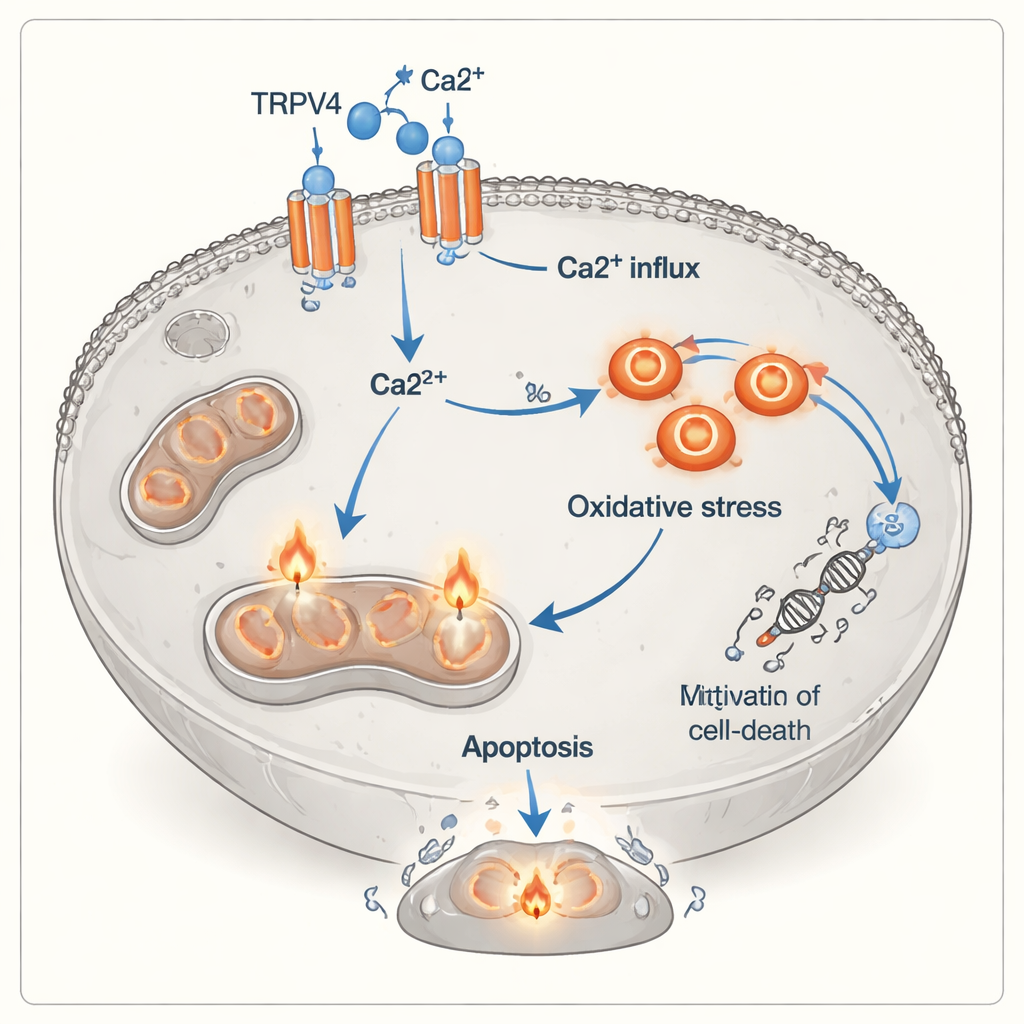
A new layer to how immune cells kill tumors
Classically, killer T cells destroy their targets by releasing toxic proteins or by triggering death receptors on the cancer cell surface. This study suggests there is another, more indirect route: signals from activated T cells—including inflammatory molecules and close cell-to-cell contact—appear to make lung cancer cells more reliant on TRPV4 and more vulnerable to calcium overload and oxidative damage. In this view, TRPV4 acts like a sensitizing switch that turns otherwise growth-promoting calcium into a lethal signal. Although the experiments were done in dishes and used a single cancer cell line, they highlight a potentially important bridge between immune attack, ion channels, and the internal stress machinery of tumor cells.
What this could mean for future treatments
For non-specialists, the takeaway is that the success of immunotherapy may depend not only on making T cells stronger, but also on making cancer cells easier to kill. By targeting TRPV4 or related calcium channels, doctors might one day boost the ability of T cells to eliminate lung tumors or fine-tune treatments to avoid damaging healthy tissues. The work is still at an early stage, and the authors stress the need for further experiments in animals and more complex models. Even so, the discovery of a TRPV4–calcium–oxidative stress axis adds a promising new target to the toolbox for improving immune-based therapies against lung cancer.
Citation: Alavi, F., Kazemi-Lomedasht, F., Eftekhari, Z. et al. Activated T cells induce apoptosis in A549 lung adenocarcinoma cells via TRPV4-mediated calcium influx. Sci Rep 16, 7155 (2026). https://doi.org/10.1038/s41598-026-38589-2
Keywords: lung cancer immunotherapy, T cells, calcium signaling, TRPV4 channel, oxidative stress