Clear Sky Science · en
Solvent-free synthesis of a binary organic material with spectroscopic, thermodynamic, dielectric and computational studies
A New Building Block for Future Electronics
From smartphones to cloud servers, modern life depends on electronic materials that are fast, efficient and increasingly sustainable to make. This study introduces a newly crafted organic solid, made without any liquid solvents, that behaves like a semiconductor and stores electric charge remarkably well. Because it combines eco-friendly preparation with properties attractive for memory chips and other organic devices, it offers a glimpse of how tomorrow’s electronics might be lighter, greener and more adaptable than today’s silicon-based technology.
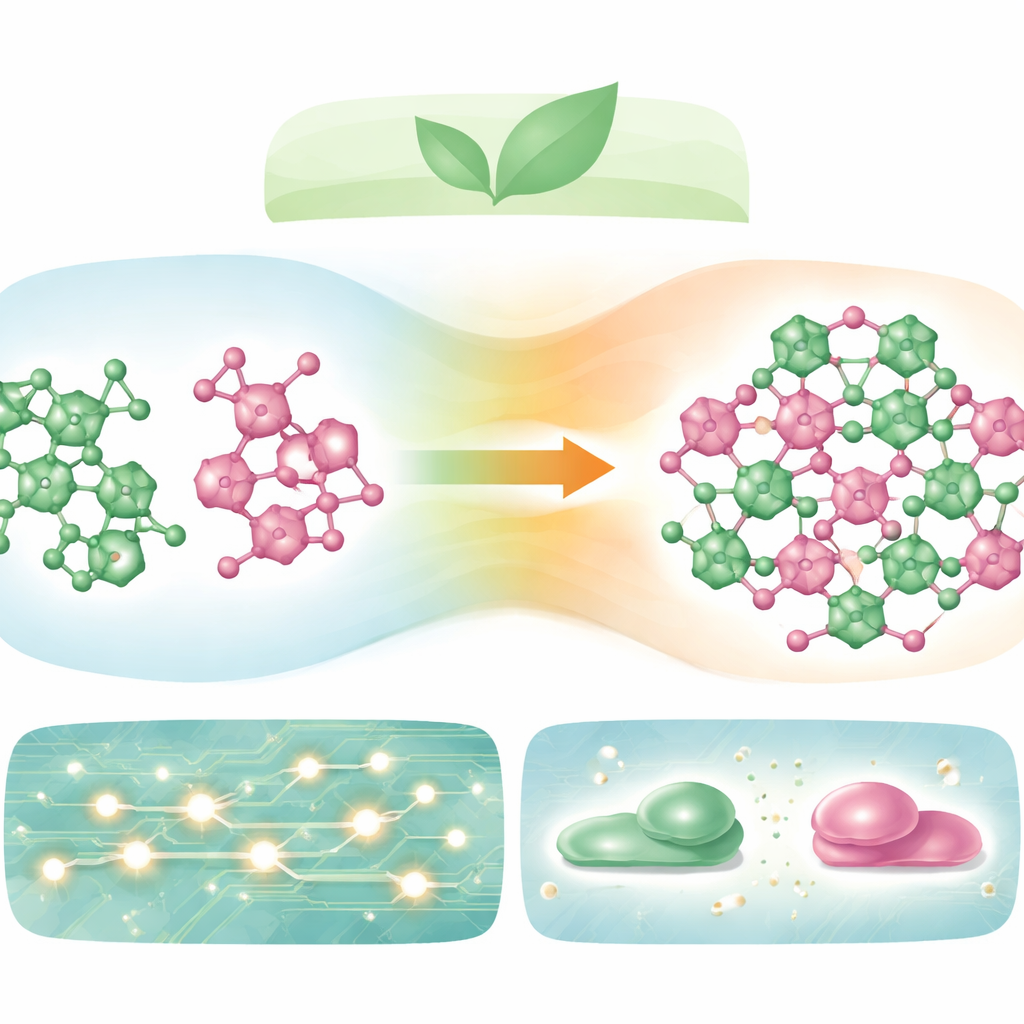
Making a Solid Without Any Liquid
The researchers set out to create a new "binary" organic material by combining two simple, inexpensive molecules: terephthalaldehyde and 2-amino-5-chloropyridine. Instead of dissolving them in a solvent, they mixed precise amounts of the two powders, gently melted them together in sealed tubes, and cycled the mixture through heating and cooling until it became fully uniform. By measuring how mixtures of different ratios melted and solidified, they constructed a phase diagram—a kind of map showing which solid forms at each composition. This map revealed that a distinct new compound appears when one part terephthalaldehyde reacts with two parts of the amine, flanked on both sides by special low-melting mixtures known as eutectics.
Proving a New Structure Is Born
To confirm that a genuine new material had formed—rather than a simple blend of the starting powders—the team used several structural probes. Infrared and Raman light spectra showed that the strong signal from the original aldehyde group vanished and a new signal characteristic of an imine (Schiff base) bond appeared, signalling that the molecules had chemically linked. Solid-state nuclear magnetic resonance further supported this transformation by showing the disappearance of carbonyl signatures and the appearance of new carbon environments. Powder X-ray diffraction patterns of the product displayed a completely different set of sharp peaks compared with either starting material, indicating a new, ordered crystal structure rather than a mechanical mixture.
Zooming In on Electrons and Interactions
Beyond establishing the structure, the authors used advanced computer calculations based on density functional theory to explore how electrons behave in the new solid, named PCPMA. They examined several possible three-dimensional shapes (conformers) of the molecule and found that one nearly linear arrangement is especially stable, allowing electrons to spread out along the backbone. Calculations of the energy gap between filled and empty electronic states, together with detailed maps of how electrons are distributed over the atoms, show that PCPMA should act as a semiconductor: it does not conduct like a metal, but it can move charge when enough energy is supplied. Additional analysis of subtle noncovalent contacts—such as weak attractions between stacked rings—showed that gentle, dispersion-like forces help shape how the molecules pack in the solid.
Heat, Stability and Electric Response
Thermal measurements added a practical perspective. Differential scanning calorimetry and related techniques showed that PCPMA melts at much higher temperatures than its parent molecules and remains stable with essentially no weight loss up to roughly 260 °C. Using the heat absorbed at melting, the team estimated quantities such as heat of mixing, interfacial energy and a "roughness" parameter that describe how crystals grow and how different phases meet at their boundaries. Most striking, when the new material was pressed into a pellet and its electrical behaviour was measured, it showed a very high dielectric constant at low frequencies—hundreds of times that of vacuum—meaning it can store large amounts of electric energy. This response weakened with increasing frequency but grew with temperature, a pattern consistent with strong polarization in the solid.
Why This Matters for Everyday Devices
Putting all of these threads together, the study demonstrates that a simple, solvent-free route can yield a robust new organic crystal that combines semiconductor-like charge transport with an unusually high ability to store electric charge. For non-specialists, that means PCPMA behaves a bit like a tunable plastic version of the materials inside computer chips and capacitors. Its stability, strong internal bonding and rich electronic structure make it a promising candidate for future organic electronics, especially memory devices that rely on storing and switching charge. While further work will be needed to process it into thin films and integrate it into real circuits, this research offers a clear proof of concept that greener chemistry can deliver functional materials tailored for next-generation, low-cost electronic technologies.
Citation: Rai, A., Rai, R., Chaudhary, S. et al. Solvent-free synthesis of a binary organic material with spectroscopic, thermodynamic, dielectric and computational studies. Sci Rep 16, 8242 (2026). https://doi.org/10.1038/s41598-026-38588-3
Keywords: organic semiconductor, Schiff base, dielectric material, solvent-free synthesis, memory devices