Clear Sky Science · en
Histatin-1 promotes the expression of markers associated with odontoblastic differentiation in the dental pulp and apical papilla
Why Spit Might Help Save Your Teeth
When a tooth is badly damaged by decay or injury, dentists try to preserve it by protecting the soft inner tissue, called the pulp. The dream of future dentistry is to help the tooth heal itself, rebuilding the hard dentin layer instead of relying only on fillings and crowns. This study explores whether a natural protein found in human saliva, called histatin‑1, could coax cells inside young teeth to turn into dentin‑forming cells and help the tooth repair from within.
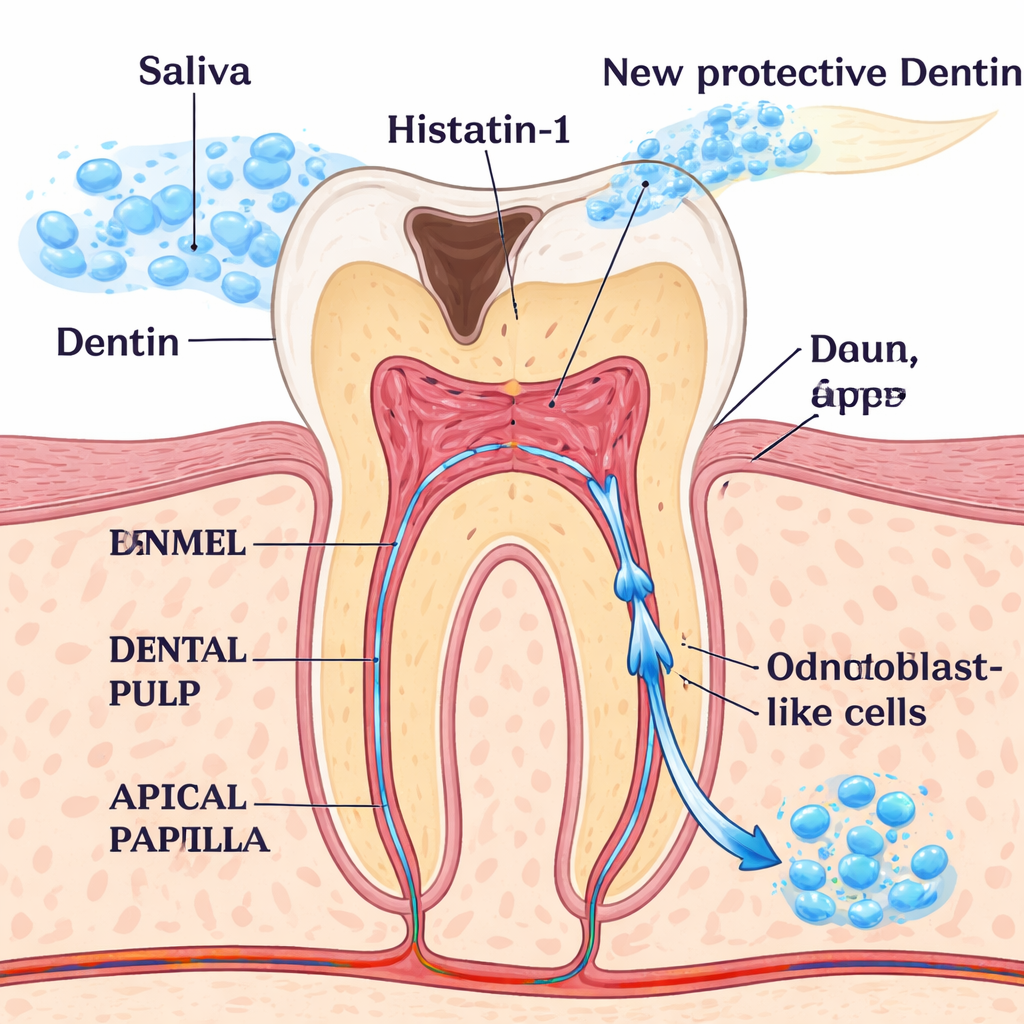
Hidden Repair Crews Inside Young Teeth
Inside every tooth, the dental pulp and a neighboring region at the root tip called the apical papilla contain versatile cells that can mature into different cell types. Among the most important are odontoblasts, the specialized cells that build and maintain dentin, the protective hard tissue beneath enamel. In deep cavities or trauma, odontoblasts are damaged or lost, and successful regenerative treatments depend on replacing their function. Scientists have long searched for safe signals that nudge pulp and apical papilla cells toward an odontoblast‑like state so they can rebuild the tooth from the inside.
A Salivary Protein with Surprising Talents
Histatin‑1 is known mainly as a small protein in saliva that protects teeth from microbes and helps wounds heal. Previous work showed that it can also encourage bone‑related cells to move, stick, and deposit mineral, hinting that it might influence hard tissue formation more broadly. The authors of this study asked whether histatin‑1 could specifically push pulp and apical papilla cells from immature permanent teeth toward an odontoblast‑like identity rather than just a generic bone‑like state. If so, this saliva‑derived molecule could become a natural tool for targeted dental regeneration.
Turning Pulp Cells into Dentin Builders
To test this idea, the researchers obtained freshly removed immature molars and split each tooth’s pulp–apical papilla complex in half, treating one half with histatin‑1 and leaving the other as a control. After a week, they examined both tissues under the microscope. Histatin‑1‑treated samples showed more of two hallmark dentin proteins—DSPP and DMP1—and more cells that stained positive for these markers, especially in the apical papilla. When the team isolated the resident mesenchymal cells and grew them in dishes, histatin‑1 again boosted DSPP levels and increased activity of beta‑catenin, a signaling protein known to drive tooth cell maturation. Under the microscope, treated cells also acquired shape features seen in developing odontoblasts: more primary cilia—tiny antenna‑like projections—and a more focused, polarized Golgi apparatus, which hints that the cells are gearing up to secrete dentin matrix in one direction.
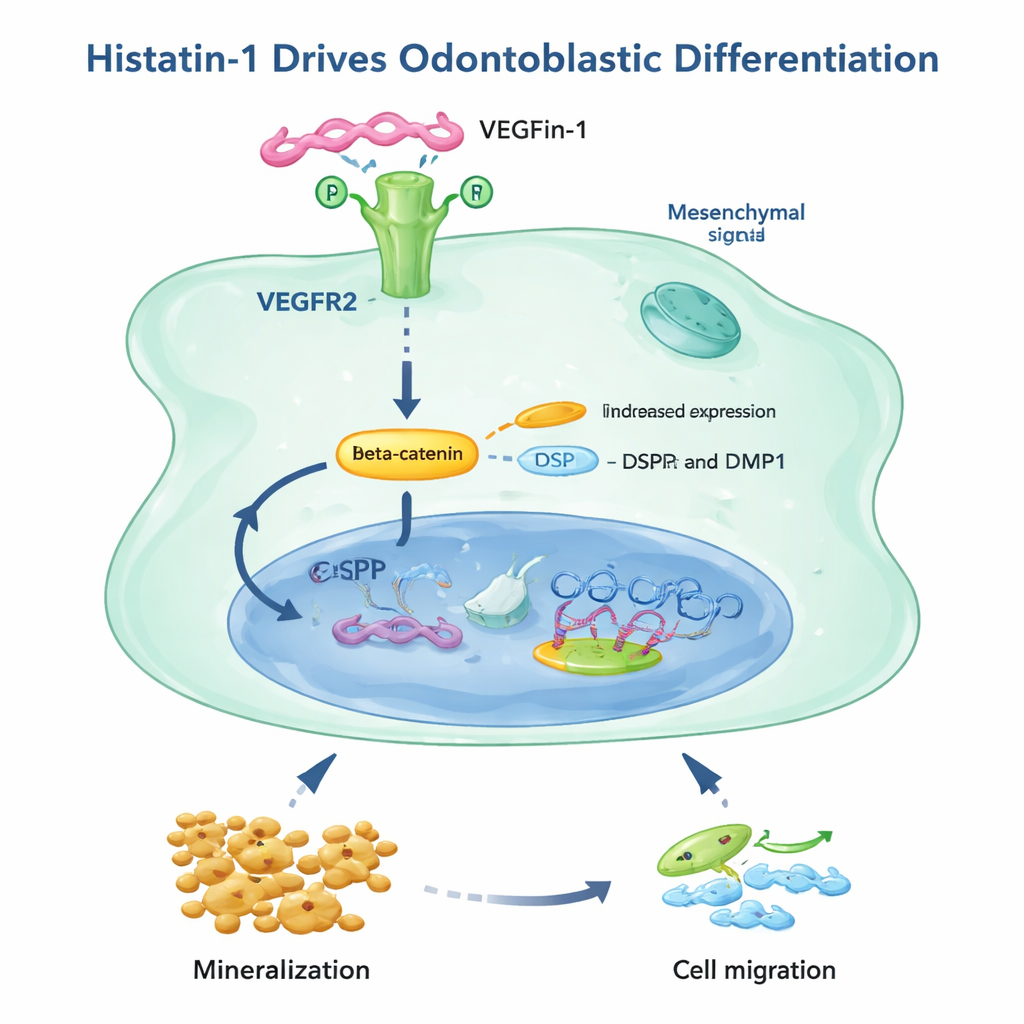
How Histatin‑1 Sends the Signal
The study went on to ask how histatin‑1 delivers its message. Earlier work had identified a receptor on cell surfaces, VEGFR2, that can bind histatin‑1. Here, the researchers confirmed that both pulp and apical papilla tissues express VEGFR2 and that histatin‑1 raises its levels. Blocking VEGFR2 with a specific drug, or using a modified form of histatin‑1 that can no longer attach to this receptor, sharply reduced the ability of histatin‑1 to trigger mineral deposit formation in these cells. The same receptor was also crucial for histatin‑1‑driven cell movement in apical papilla cells, though pulp cells appeared to have an extra, VEGFR2‑independent way to migrate. Overall, the findings point to a pathway in which histatin‑1 binds VEGFR2, boosts beta‑catenin signaling, switches on odontoblast markers, and enhances both mineralization and movement of key repair cells.
What This Could Mean for Future Dentistry
For non‑specialists, the take‑home message is that a natural component of saliva can help instruct young tooth cells to behave like dentin builders through a defined signaling route. While this work was done in cell and tissue samples rather than in patients, it identifies histatin‑1 as a promising, seemingly tissue‑friendly candidate for regenerative dental therapies. In the future, tailored gels, rinses, or pulp‑capping materials containing histatin‑1 could one day help guide the tooth’s own cells to migrate into damaged areas and rebuild lost dentin, offering a more biological alternative to traditional restorative procedures.
Citation: Silva, P., Garrido, M., Tapia, H.A. et al. Histatin-1 promotes the expression of markers associated with odontoblastic differentiation in the dental pulp and apical papilla. Sci Rep 16, 7360 (2026). https://doi.org/10.1038/s41598-026-38578-5
Keywords: dental regeneration, histatin-1, odontoblasts, dental pulp stem cells, dentin repair