Clear Sky Science · en
Phosphidated tungsten oxide@polyaniline hybrid electrode on nickel foam for dual-function supercapacitor and methanol oxidation
Smarter Materials for Storing and Making Clean Energy
Modern life runs on electricity, but our current batteries and fuel technologies are often bulky, slow to charge, or too expensive. This study reports a new “two-in-one” material that can both store energy like a fast, long‑lasting super‑battery and help convert liquid fuel into electricity more efficiently. By carefully stacking different layers of common and precious materials, the researchers build a tiny structure that could make future gadgets and clean power systems smaller, quicker, and cheaper.
Building a Tiny Energy Sponge
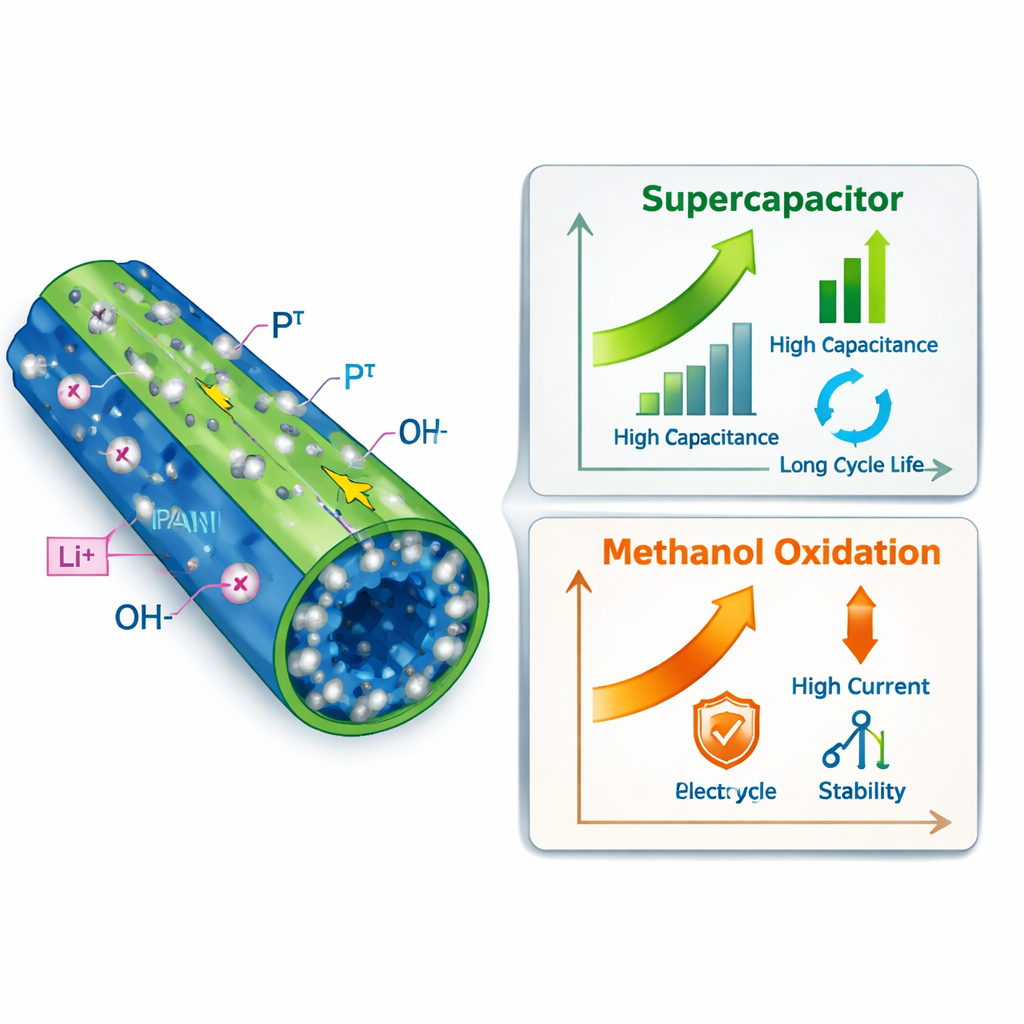
At the heart of this work is a carefully engineered electrode—the part of a device where electrical reactions happen. The scientists start with nickel foam, a metal that looks like a stiff sponge full of holes. They grow needle‑like crystals of tungsten oxide on this foam, and then partially convert them into tungsten phosphide, a related compound that conducts electricity better and offers more reactive sites. Next, they coat these nano‑needles with a thin layer of the conducting plastic polyaniline. This layered design creates a highly porous, three‑dimensional network that lets ions and electrons move quickly, while providing plenty of surface area for reactions.
Why This Hybrid Stores So Much Charge
The combination of tungsten phosphide and polyaniline is key to the material’s strong performance as a supercapacitor, a device that charges and discharges much faster than a conventional battery. Tungsten compounds provide rich “redox” sites—places where electrons can be taken up and released—while polyaniline acts like a fast, flexible highway for electrical charges. Tests in an alkaline water‑based solution show that the hybrid electrode can hold an impressive 1210 coulombs of charge per gram at moderate current, far more than either component alone. Even when the device is pushed to much higher charge–discharge speeds, it retains most of its capacity, thanks to the open, sponge‑like structure that allows ions in the liquid to reach deep into the material.
From Single Electrode to Practical Device
To see how this material would behave in a real‑world setting, the team built an asymmetric supercapacitor. They used their hybrid electrode as the positive side and common activated carbon—similar to that found in water filters—as the negative side, with a paper separator and the same alkaline solution. This device could be safely operated over a wide voltage range, which is crucial for storing more energy. It delivered an energy density of about 60 watt‑hours per kilogram, comparable to some battery technologies, while still providing the rapid power bursts typical of supercapacitors. After 10,000 charge–discharge cycles, the device kept nearly 90 percent of its original capacity, indicating that the layered structure resists the cracking and breakdown that often plague such materials over time.
Helping Fuel Cells Turn Methanol into Power
The same architecture also serves as a powerful platform for turning methanol, a liquid fuel, into electricity in alkaline fuel cells. For this task, the researchers added a very thin sprinkling of platinum nanoparticles onto the polyaniline layer. Platinum is the gold‑standard catalyst for methanol oxidation, but it is rare and expensive, so using it efficiently is vital. The nitrogen‑rich polyaniline helps anchor the platinum as tiny, well‑spaced particles, and the tungsten phosphide supplies additional chemical helpers that ease the removal of carbon‑based residues that would otherwise clog the surface. As a result, the hybrid electrode shows much higher activity—nearly two and a half times more current per unit of platinum—than a similar electrode without the tungsten‑based layer, and it maintains over 80 percent of its activity after 1000 test cycles.
What This Means for Future Energy Devices
In simple terms, the researchers have built a kind of “Swiss Army knife” electrode: without any precious metals it works as a high‑performance, long‑lasting energy storage material, and with a tiny amount of platinum added it becomes an efficient, durable catalyst for methanol fuel cells. This dual‑purpose design could help reduce the number of different materials needed in advanced energy systems and cut down on the use of costly metals. While more work is required to scale up the technology and test it in full devices, the study points toward compact, multi‑functional components that could underpin the next generation of portable electronics and clean power systems.
Citation: Adriyani, T.R., Ensafi, A.A. Phosphidated tungsten oxide@polyaniline hybrid electrode on nickel foam for dual-function supercapacitor and methanol oxidation. Sci Rep 16, 7008 (2026). https://doi.org/10.1038/s41598-026-38573-w
Keywords: supercapacitor, fuel cell, electrode materials, conducting polymer, methanol oxidation