Clear Sky Science · en
Optimized paraquat removal using Bi₄O₅Br₂: synthesis, performance evaluation, and mechanistic insights
Why cleaner water matters
Many modern farms rely on powerful weed killers to protect crops, but some of these chemicals are so persistent and toxic that even tiny amounts in drinking water can pose serious health risks. This study tackles one of the most controversial of these herbicides, paraquat, and explores a new material based on bismuth compounds that can almost completely strip it out of water using a process as simple as mixing and filtering. The work shows how careful tuning of material structure and operating conditions can turn a laboratory powder into a realistic tool for safer water.

A stubborn weed killer in our water
Paraquat is a fast-acting weed killer used worldwide, valued by farmers but feared by toxicologists. It dissolves very easily in water and clings strongly to soil particles, which helps it linger in the environment and find its way into rivers, lakes, and wells. Even microgram-per-liter levels are worrisome because paraquat is extremely poisonous: accidental or intentional ingestion is often fatal, and long-term exposure has been linked to serious illnesses such as Parkinson’s disease. Some regions, like the European Union and Brazil, have banned it outright, but many countries still use it, making affordable ways to remove it from water supplies a pressing need.
Building a smarter sponge for toxins
Instead of trying to break paraquat down with energy-intensive methods like advanced oxidation or high-pressure membranes, the authors focus on adsorption — essentially, making a smart sponge that captures the molecules and lets clean water pass. They start from a porous bismuth-based framework called CAU-17 and transform it using a double-solvent process into tiny rods of a compound named Bi₄O₅Br₂. Detailed tests show that these rods have a layered, mesoporous architecture: lots of evenly sized pores around 7 nanometers wide and a moderate but effective surface area. Electron microscopy and elemental mapping reveal that the building blocks (bismuth, oxygen, bromine, and carbon) are uniformly distributed, ensuring that active sites are spread throughout the material rather than clumped in a few spots.
Tuning conditions for maximum cleanup
To find the best way to use this new adsorbent, the team turns to a statistical optimization approach called response surface methodology. Instead of changing one factor at a time, they systematically vary four at once: how much paraquat is in the water, how long the material and water stay in contact, how much Bi₄O₅Br₂ is added, and the solution’s acidity (pH). Across 29 carefully planned tests, removal efficiency ranges from about 40% to over 97%. The analysis reveals that the amount of adsorbent and the pH of the water matter most: adding slightly more Bi₄O₅Br₂ dramatically boosts removal, while near-neutral conditions around pH 6–7 are ideal. Contact time plays a lesser role because most of the adsorption happens quickly, within about half an hour, and the initial paraquat concentration mainly affects how easily the available sites become saturated.
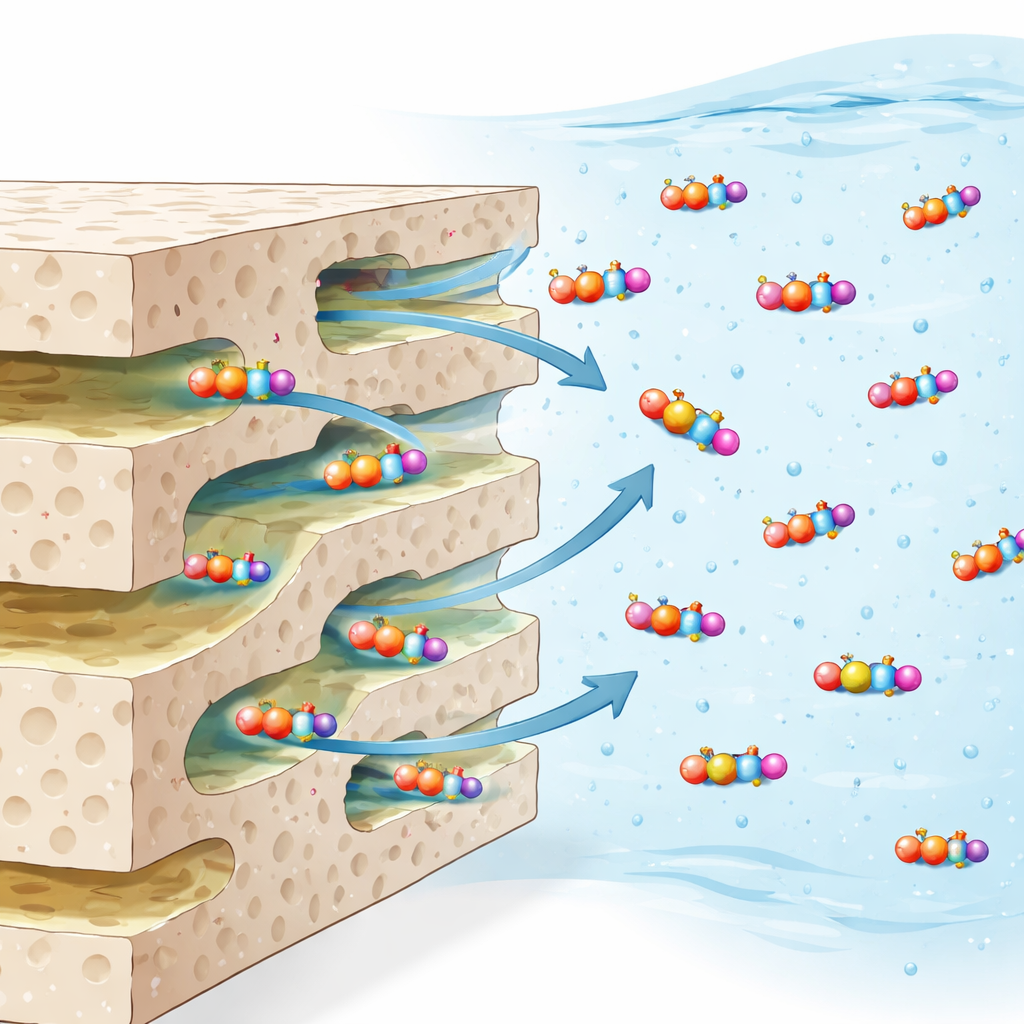
How the trap holds on to paraquat
At the microscopic level, the material acts like a negatively charged, finely layered sponge when the water is close to neutral or slightly alkaline. Paraquat molecules carry a positive double charge, so they are strongly attracted to the oppositely charged surface, much like pieces of lint drawn to a sweater fresh from the dryer. The pores allow the molecules to diffuse rapidly into the interior, where they encounter abundant binding sites. Spectroscopic fingerprints taken before and after adsorption show subtle shifts in signals associated with surface –OH and related groups, indicating that hydrogen bonding helps lock paraquat in place alongside the dominant electrostatic pull. The combination of accessible pores, good surface area, and favorable charge makes it possible to achieve very high removal using an impressively low amount of material.
What this means for safer water
By the end of the study, the researchers demonstrate that Bi₄O₅Br₂ can strip roughly 97–99% of paraquat from mildly contaminated water using only a tiny dose of adsorbent and simple mixing for under an hour. When compared with other advanced materials, it matches or exceeds their performance while using less material and operating under gentler conditions. For a layperson, the key takeaway is that a carefully engineered powder can reliably “soak up” one of the most dangerous herbicides from water without complex equipment. The authors note that the next steps are to test how many times the material can be reused, how stable it is in real wastewater, and how it behaves in continuous flow systems — all crucial for turning this promising lab result into a practical safeguard for communities that still live with paraquat in their environment.
Citation: Dehghani, Z., Fekri, M., Mahmoodabadi, M. et al. Optimized paraquat removal using Bi₄O₅Br₂: synthesis, performance evaluation, and mechanistic insights. Sci Rep 16, 8229 (2026). https://doi.org/10.1038/s41598-026-38566-9
Keywords: paraquat, water treatment, adsorption, nanomaterials, herbicide pollution