Clear Sky Science · en
Fecal metabolic signals are associated with changes in microbiota and systemic metabolic pathways in Crohn’s disease
Why Your Gut Chemistry Matters
Crohn’s disease is a chronic condition in which the immune system attacks the digestive tract, causing pain, diarrhea, and long-term damage. While we know that gut bacteria are involved, this study asks a deeper question: how do the tiny chemicals produced in the gut and circulating in the blood connect the microbes in our intestines to whole-body health? By tracking these chemical signals in stool and blood from people with and without Crohn’s, the researchers show that shifts in gut chemistry may help harmful microbes thrive and could open new doors for dietary and metabolic treatments.
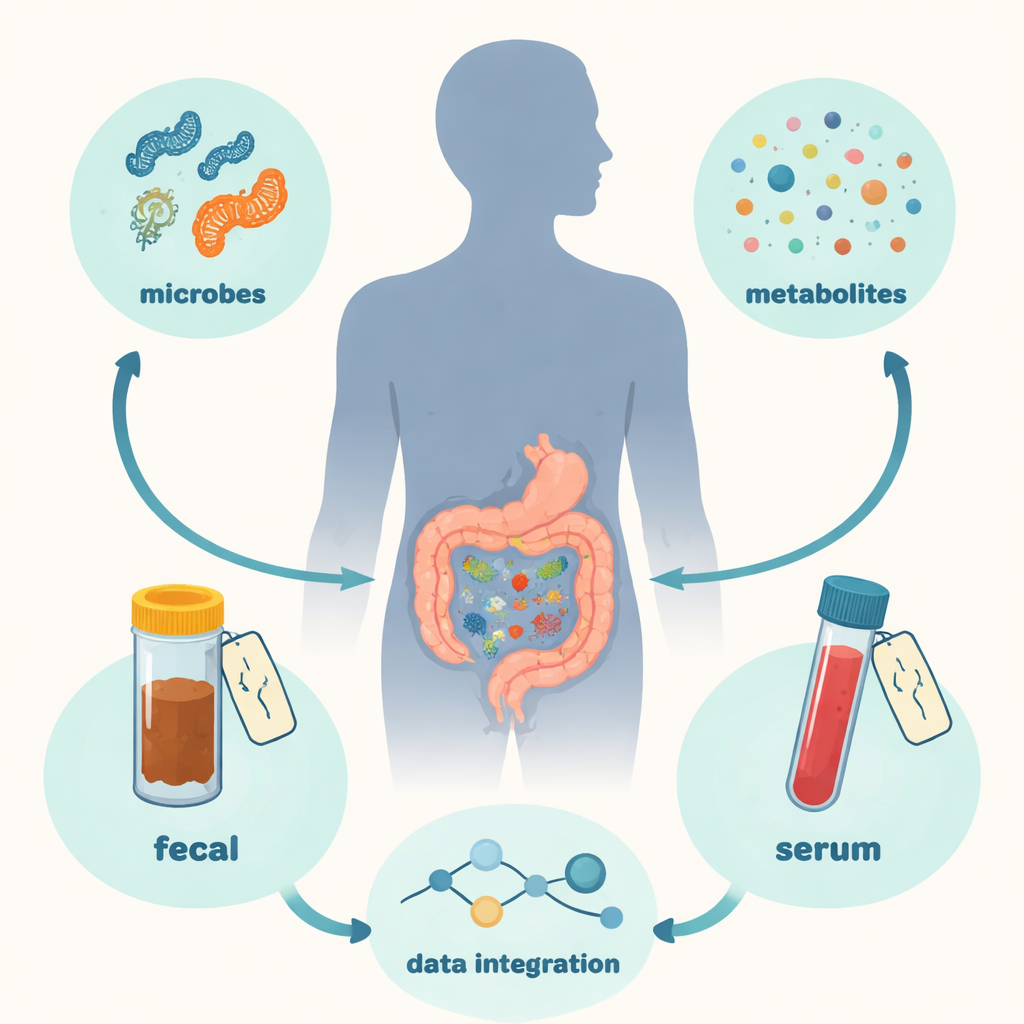
The People Behind the Data
The research team followed 123 adults, including 80 with Crohn’s disease and 43 healthy volunteers. For each person, they collected stool and blood samples, sometimes repeatedly over time. From these samples they measured hundreds of small molecules, called metabolites, using a sensitive technique known as mass spectrometry, and mapped the community of gut bacteria using DNA sequencing. This “multi-omics” approach let them see not just which microbes were present, but also which chemical pathways were active in the gut and in the bloodstream, and how these patterns changed with disease flare-ups or remission.
Gut Sugars and Uninvited Mouth Bacteria
One of the most striking findings came from the stool samples. People with Crohn’s had higher levels of several complex sugars, including trehalose, raffinose, stachyose, glucose, and fructose, in their feces. At the same time, these sugars were strongly linked to bacteria normally found in the mouth, such as Veillonella and Streptococcus, that had migrated into the inflamed gut. The data suggest that excess or poorly absorbed sugars in the intestine may act as “fuel” that helps these out-of-place, inflammation-associated microbes settle in and persist. This fits with dietary approaches to Crohn’s that restrict certain carbohydrates and processed additives, hinting at a concrete chemical reason why such diets may help some patients.
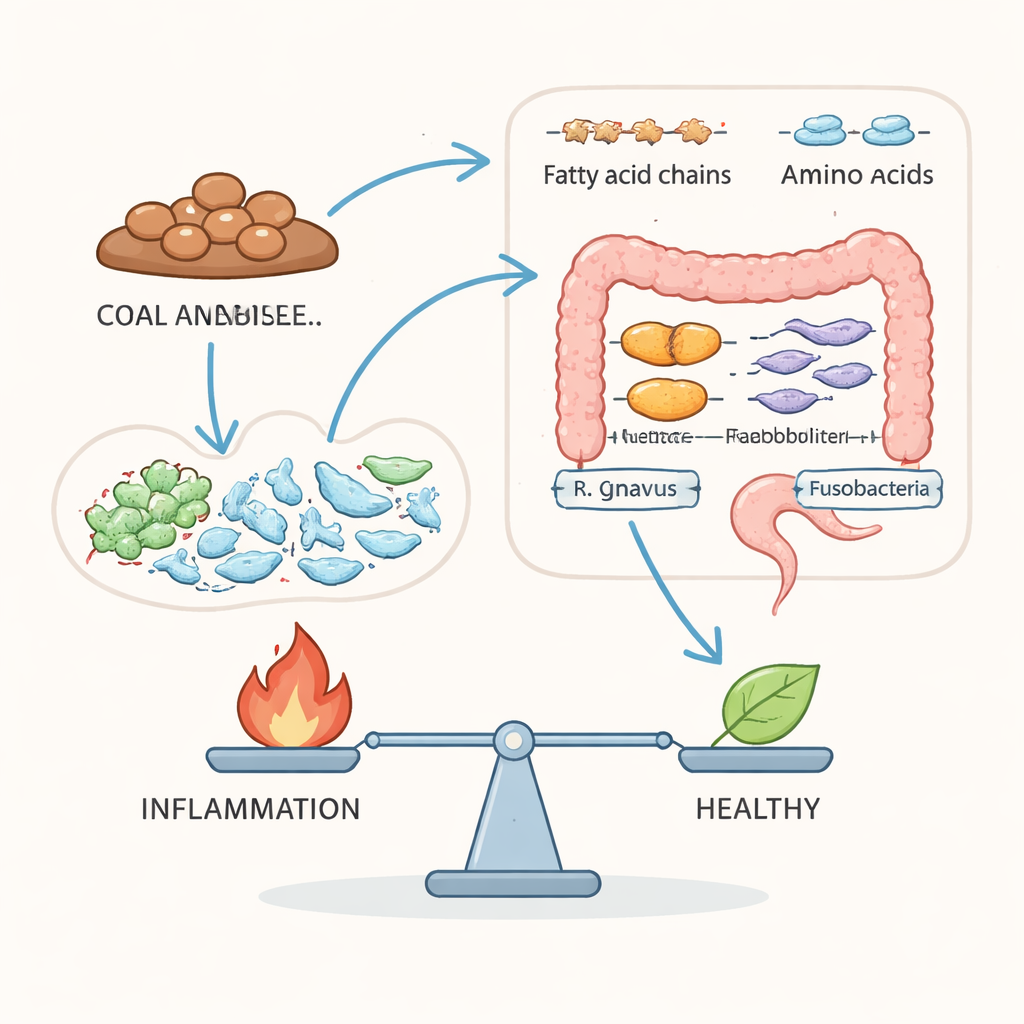
Fatty Molecules, Amino Acids, and a Shift in Energy Use
The study also uncovered a broader reshaping of energy metabolism in Crohn’s disease. In the blood, people with Crohn’s showed signs of reduced use of classic energy pathways tied to sugar breakdown and the cell’s powerhouses, along with increased signals related to fat use. In the stool, the pattern was reversed: pathways linked to sugar breakdown and core energy cycles were more active, while the breakdown of long-chain fats, an important fuel for cells lining the colon, appeared impaired. Certain inflammatory fatty molecules derived from arachidonic acid were enriched in the stool and tightly correlated with gut bacteria previously linked to Crohn’s, such as Ruminococcus gnavus and Fusobacteria. At the same time, essential amino acids like tryptophan, tyrosine, phenylalanine, and histidine built up in feces during active disease but tended to drop in the blood, suggesting disrupted absorption and altered microbial processing during flares.
Signals That Track Disease Activity
Because patients were sampled during both quiet and active phases of their disease, the team could ask which chemical patterns tracked with symptoms and with objective markers of inflammation in blood and stool. Many stool metabolites that distinguished Crohn’s from health also changed in step with flare-ups, high C-reactive protein, or elevated fecal calprotectin. These same metabolites were tied to a previously established “dysbiosis index” that measures how far a person’s gut microbiome has drifted from a healthy state, as well as to the overall diversity of gut bacteria. In other words, specific chemical signatures in the stool were consistently linked both to inflammatory activity and to how disrupted the microbial community had become.
What This Means for Patients
Taken together, the findings portray Crohn’s disease as a disorder of intertwined microbes and metabolism. Extra sugars and altered fat and amino acid processing in the gut appear to support inflammation-linked bacteria, which in turn may produce more inflammatory molecules, creating a self-reinforcing cycle. While this study does not prove cause and effect, it highlights fecal metabolites as potential levers to nudge the gut ecosystem toward a healthier state, whether through diet, targeted supplements, or future drugs that reshape microbial metabolism. For patients, this work strengthens the idea that what happens chemically in the gut is not just a by-product of disease—it may be part of the engine driving it, and therefore a promising target for more precise, less invasive therapies in the years ahead.
Citation: Levhar, N., Hadar, R., Braun, T. et al. Fecal metabolic signals are associated with changes in microbiota and systemic metabolic pathways in Crohn’s disease. Sci Rep 16, 6991 (2026). https://doi.org/10.1038/s41598-026-38558-9
Keywords: Crohn’s disease, gut microbiome, metabolomics, intestinal inflammation, diet and gut health