Clear Sky Science · en
Fabrication of an electrochemical sensor based on magnetic molecularly imprinted polymer for detection of sunset yellow dye
Why the color in your drink matters
Bright orange sodas, candies, and even some medicines often owe their appeal to a synthetic dye called Sunset Yellow. While regulators limit how much of this dye can be added to food, it can still escape into rivers, lakes, and tap water, and may be linked to health concerns such as allergies and possible effects on children’s behavior. This study describes a new, low-cost sensor that can quickly and selectively detect Sunset Yellow in water and beverages, helping protect both environmental quality and consumer safety.
A stubborn dye in everyday life
Sunset Yellow is popular in the food, pharmaceutical, and cosmetics industries because it dissolves easily in water and remains stable across a wide range of conditions. Those same strengths become weaknesses once the dye is released into the environment. It does not break down easily, can linger in waterways, and has been associated with potential toxic and allergic effects in laboratory and clinical studies. Traditional methods to remove or detect such dyes can be complex, expensive, or not very selective. There is a clear need for simple tools that can pinpoint specific dyes like Sunset Yellow in complex mixtures such as river water, wastewater, or soft drinks.
Building a smart magnetic sponge
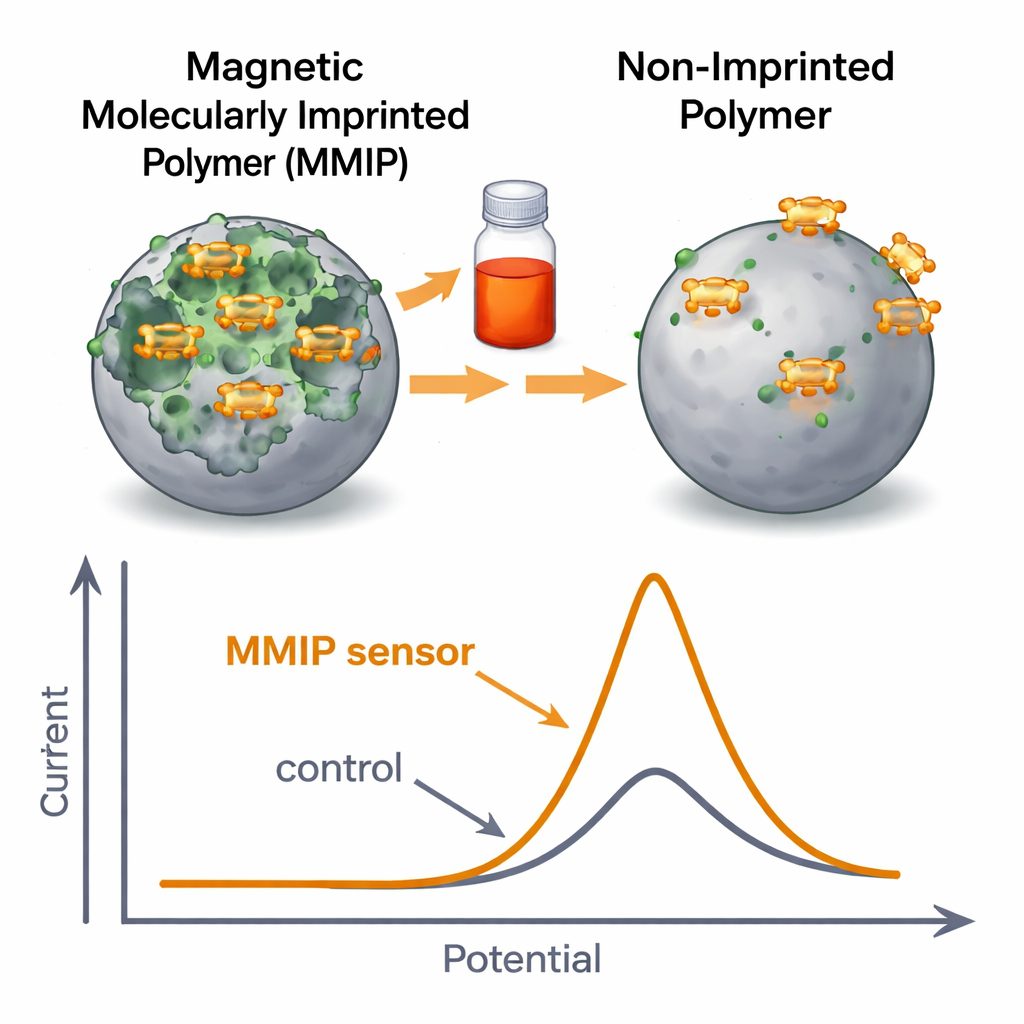
The researchers tackled this challenge by creating tiny “smart sponges” known as magnetic molecularly imprinted polymers. These particles have a magnetic core so they can be moved or collected with a magnet, and a surrounding shell made of polymer that is molded around Sunset Yellow molecules during fabrication. When the dye is later washed out, it leaves behind matching cavities—like a lock shaped to fit a single key—that strongly and selectively rebind Sunset Yellow from a solution. Electron microscope images showed that the finished particles were roughly 69 nanometers across, with a rough, cavity-rich surface ideal for capturing the dye. Batch tests revealed a high sorption capacity of about 80 milligrams of dye per gram of material under acidic conditions, following well-known adsorption patterns that indicate efficient, single-layer coverage on the particle surface.
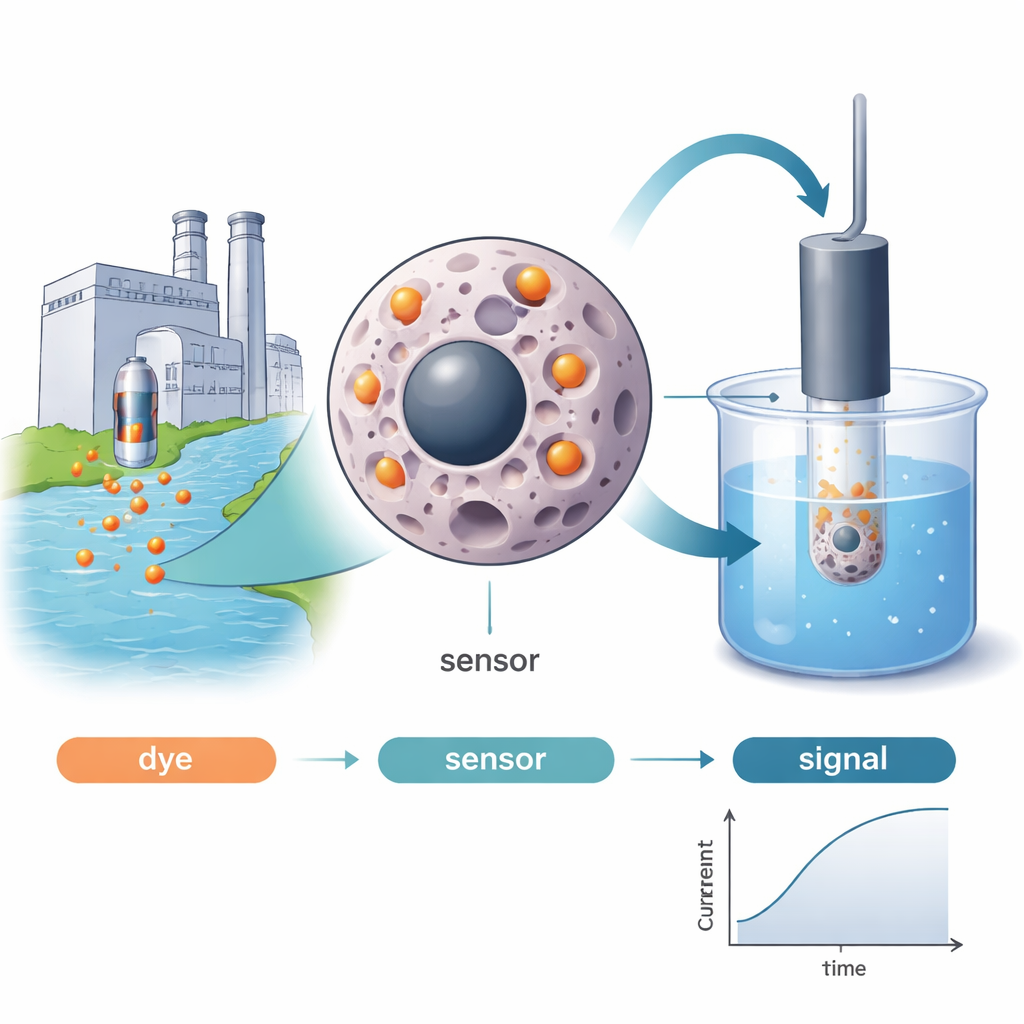
Turning molecular recognition into an electrical signal
To convert this selective binding into a practical analytical tool, the team embedded the magnetic imprinted particles into a carbon paste electrode, forming the heart of an electrochemical sensor. When this sensor is dipped into a solution and a small voltage is applied, Sunset Yellow that has collected in the cavities undergoes an oxidation reaction, producing a measurable electrical current. Compared with non-imprinted particles and blank electrodes, the imprinted sensor showed much sharper and higher current peaks, a clear sign that its tailor-made binding sites concentrate the dye near the electrode surface. The sensor worked best at neutral pH (around 7) and an accumulation time of about one and a half minutes, conditions that balance fast capture of the dye with strong, stable electrical signals.
Performance in real water and drinks
Beyond controlled laboratory solutions, the sensor was tested on river water, industrial wastewater, and commercial fruit juices. The samples were lightly filtered and, in some cases, spiked with known amounts of Sunset Yellow to see how much the sensor could recover. Across these tests, the sensor returned 72.9 to 99.3 percent of the added dye, indicating good accuracy even in the presence of many other substances. It also showed little interference from common compounds such as glucose, vitamin C, or another dye, Congo Red, underscoring its selectivity. The device could reliably detect Sunset Yellow at low concentrations, within a range relevant for environmental monitoring and quality control in the food industry.
What this means for consumers and the environment
In plain terms, this work delivers a reusable, magnetically controllable “nose” for Sunset Yellow that is sensitive, selective, and relatively inexpensive to make. By matching the shape and charge of the target dye, the magnetic polymer particles act like custom-made traps that not only pull the dye out of water but also allow an electrode to measure how much is present. Such sensors could help regulators, water treatment plants, and manufacturers quickly check whether dye levels stay within safe limits, and could be adapted to watch for other artificial colorants or pollutants. As synthetic additives continue to flow through our food and water systems, tools like this offer a practical way to keep an eye on what we cannot see with the naked eye.
Citation: Malik, S., Ahmad, W., Khan, A. et al. Fabrication of an electrochemical sensor based on magnetic molecularly imprinted polymer for detection of sunset yellow dye. Sci Rep 16, 6369 (2026). https://doi.org/10.1038/s41598-026-38556-x
Keywords: sunset yellow dye, electrochemical sensor, molecularly imprinted polymer, water quality monitoring, food colorant safety