Clear Sky Science · en
Exploring the toxic mechanism of cypermethrin-induced neurodegeneration diseases via network toxicology and molecular docking
Everyday Bug Sprays and the Aging Brain
From farms to household kitchens, insecticides help control pests—but some of these chemicals don’t just vanish after use. This study examines cypermethrin, a widely used insecticide, and asks a question with real-world stakes: could long-term exposure quietly raise our risk of brain disorders such as Alzheimer’s and Parkinson’s disease? Using advanced computer-based methods, the researchers map how this chemical might interact with the body and the brain, offering an early warning about a pollutant many people encounter without realizing it.
Why Nerve Cells Are So Hard to Protect
Neurodegenerative diseases, including Alzheimer’s, Parkinson’s, and other forms of dementia, are illnesses in which nerve cells in the brain gradually die and cannot be replaced. As these cells fail, people lose memory, thinking ability, and control of movement. At the same time, the modern world is seeing greater use of pesticides, and several long-term population studies have reported that communities with heavier pesticide use have higher rates of these brain diseases. Animal and cell experiments suggest that pyrethroid insecticides such as cypermethrin can damage brain cells through oxidative stress (chemical “rusting”) and long-lasting inflammation. But until now, scientists have not had a clear picture of how this particular chemical might connect, at the molecular level, to human neurodegeneration.
Building a Digital Map of Chemical Harm
To tackle this, the team turned to “network toxicology,” a big-data approach that pulls together information from many biological databases. First, they predicted which human proteins cypermethrin could plausibly bind to by comparing its structure with thousands of known compounds. Then they gathered hundreds of genes and proteins already linked to neurodegenerative diseases from major medical and genetic resources. By overlapping these two lists, they discovered 108 shared targets—proteins that both interact with cypermethrin and are associated with brain degeneration. They then built a large interaction map showing how these proteins talk to each other inside cells and used it to highlight 19 “hub” proteins that appear especially central to cypermethrin’s potential toxic effects.
Key Proteins at the Heart of the Problem
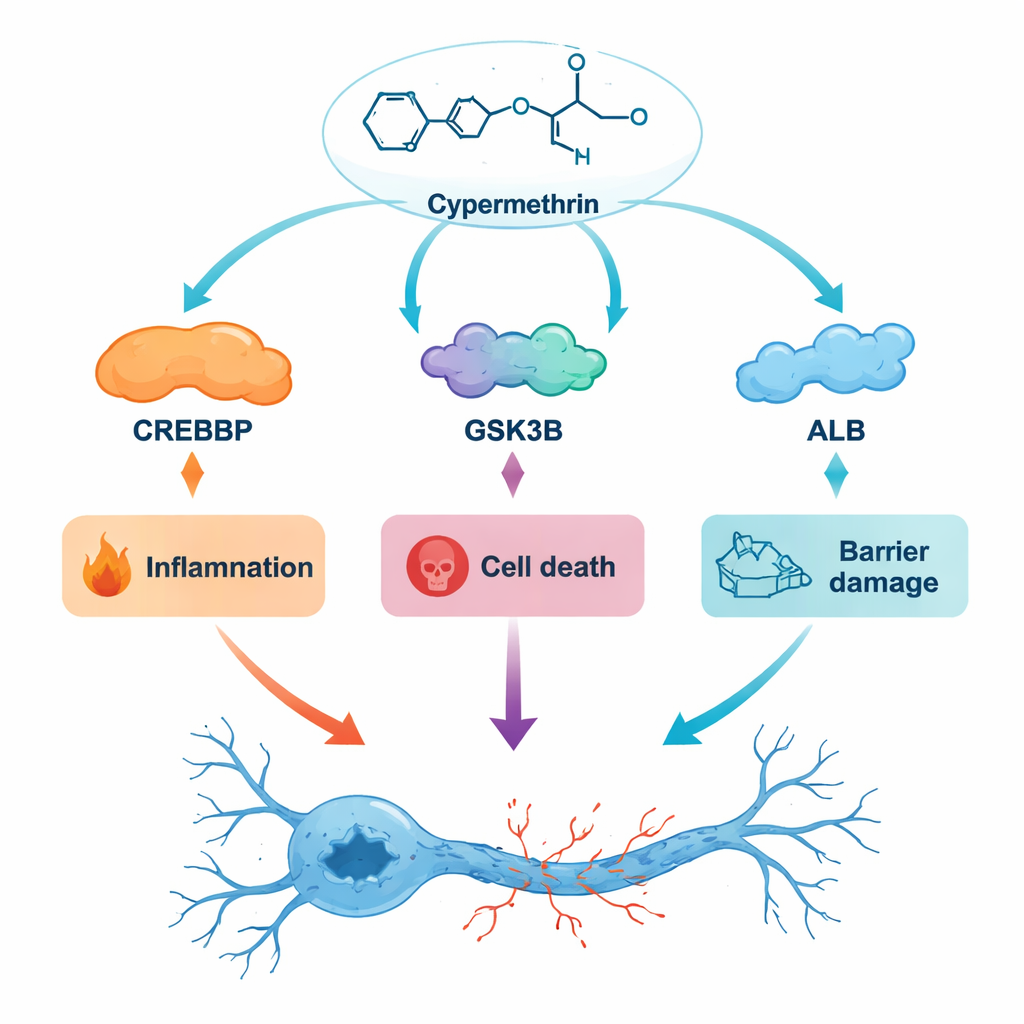
Several of the most important hub proteins have well-known roles in brain health. For example, APOE helps manage fats in the brain and is strongly tied to Alzheimer’s risk, while A2M helps clear away harmful protein clumps. Others, such as AKT1 and SRC, sit inside major signaling pathways that control whether cells survive or die. The researchers focused on three particular proteins: CREBBP, which helps switch genes on and off and supports memory formation; GSK3B, a signaling enzyme long linked to abnormal brain protein deposits and inflammation; and ALB, the main blood protein that ferries chemicals throughout the body and can cross into the brain when the blood–brain barrier is damaged. When they used detailed computer “docking” simulations, cypermethrin fit snugly into pockets on each of these proteins, suggesting it could realistically bind and alter their behavior.
Simulating Damage at the Molecular Level
To see whether these matches were stable or just fleeting, the team ran virtual “movies” of the proteins and cypermethrin interacting over 100 nanoseconds, tracking how much the structures wobbled. In all three cases, the complexes settled into stable shapes, supporting the idea that cypermethrin could stick to these proteins long enough to matter biologically. The patterns they observed fit with known disease mechanisms: GSK3B involvement points to increased inflammation and misfolded brain proteins; CREBBP interference could disturb gene programs that protect neurons; and strong binding to ALB supports the idea that cypermethrin may hitch a ride in the bloodstream and influence how much of the chemical reaches the brain.
What This Means for People and Public Health
Put simply, the study suggests that cypermethrin does not act on just one “off switch” in the brain but instead nudges an entire network of proteins toward stress, inflammation, and cell death—conditions that favor the development of neurodegenerative diseases. The work does not prove that cypermethrin causes Alzheimer’s or Parkinson’s in humans, because it relies on computer models rather than clinical trials. However, it highlights plausible biological routes by which everyday pesticide exposure could contribute to long-term brain damage and identifies specific proteins that future lab and clinical studies can test. As regulators and physicians weigh the safety of common insecticides, this network-level view provides an important scientific basis for more cautious use and for developing early diagnostic tools for people at risk.
Citation: Li, S., Ding, W., Yu, Y. et al. Exploring the toxic mechanism of cypermethrin-induced neurodegeneration diseases via network toxicology and molecular docking. Sci Rep 16, 7293 (2026). https://doi.org/10.1038/s41598-026-38547-y
Keywords: cypermethrin, pesticide neurotoxicity, neurodegenerative diseases, brain inflammation, environmental pollutants