Clear Sky Science · en
Novel histological observation method using the scattered light of unstained collagen sections
Why looking at invisible fibers matters
Our skin, tendons, and many organs are held together by collagen, a tough, ropelike protein. When these collagen fibers are damaged or rearranged, scars form and tissues stiffen—a process known as fibrosis that underlies many common conditions, from liver cirrhosis to heart failure and skin aging. Yet doctors and researchers still struggle to see the fine details of how collagen changes during disease, because existing imaging tools are either slow, expensive, or require elaborate staining. This study introduces a new way to look at collagen in ordinary lab samples using only light and clever image processing, potentially making detailed fiber analysis much more accessible.
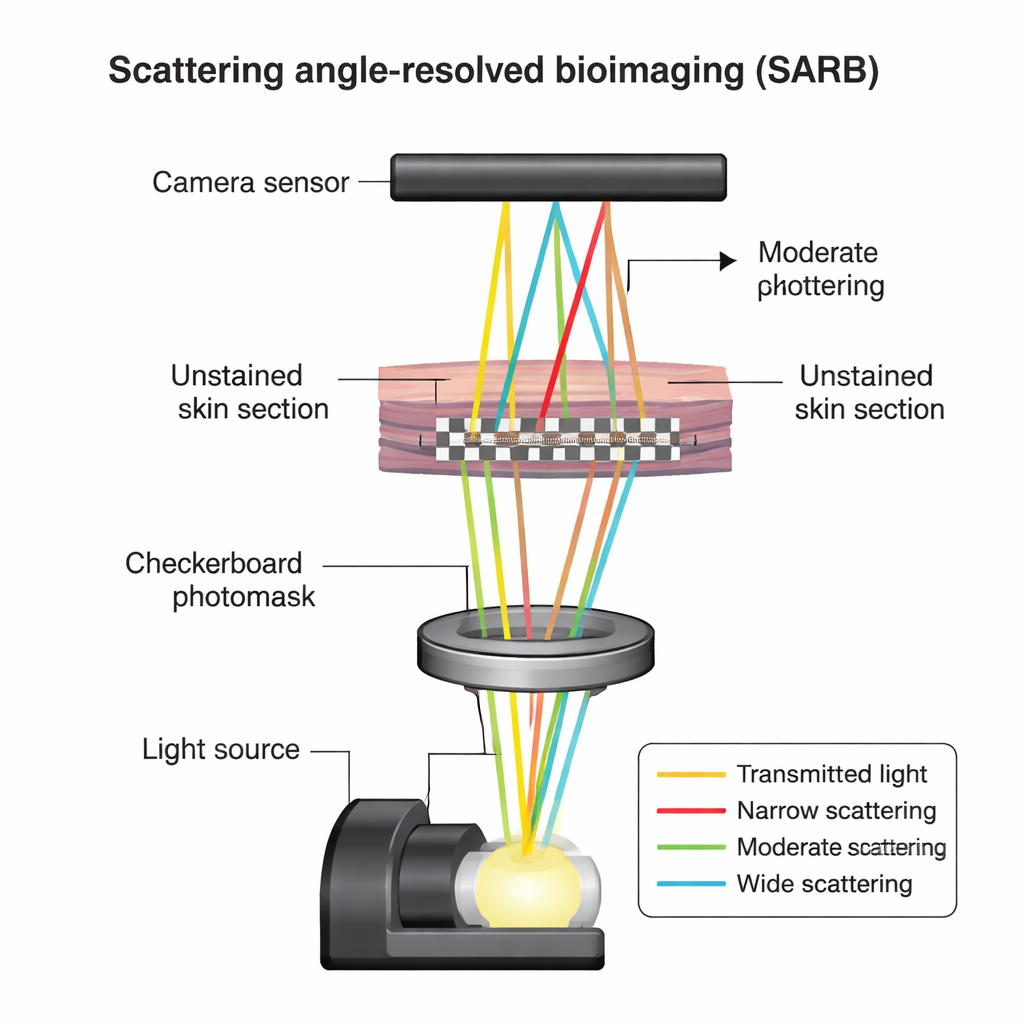
A new way to see with scattered light
The researchers focused on standard pathology samples: thin slices of mouse skin that had been fixed and embedded in paraffin, just like tissues examined every day in hospitals. Instead of coloring these sections with chemical stains, they kept them unstained and used a technique called scattering angle-resolved bioimaging, or SARB. In simple terms, when light passes through tissue, some light goes straight through, while some is scattered in different directions depending on the tiny structures it encounters. SARB shines a finely patterned checkerboard of light through the tissue in a regular microscope and, by shifting this pattern and analyzing how the image changes, mathematically teases apart light that has been scattered at different angles. This turns an ordinary microscope into a tool that can “read” subtle structural differences without dyes.
Turning ordinary sections into rich structural maps
Using SARB on 4-micrometer-thick mouse skin slices, the team could clearly see the major skin layers and hair follicles, but the real gain was inside the collagen bundles in the dermis. By separating images into components dominated by narrowly, moderately, and widely scattered light, then assigning these to red, green, and blue color channels, they created composite views where differences in scattering showed up as distinct colors. Collagen bundles that looked uniformly pale pink in standard hematoxylin-and-eosin (H&E) staining instead revealed internal streaks and speckles in SARB images, hinting at finer substructures that conventional light microscopy normally hides.
Checking against electron microscopes
To test whether these colorful scattering patterns actually reflected real fiber architecture, the same tissue sections were later examined with scanning and transmission electron microscopes, which can resolve individual collagen fibrils. SARB had revealed two main internal patterns within collagen bundles: linear streaks and dot-like spots. Electron microscopy showed that the streaky SARB regions corresponded to collagen fibrils seen lengthwise, while the dotted regions matched fibrils seen in cross-section. In other words, although SARB cannot see single fibrils, it can report their overall orientation and organization within a bundle. This establishes a crucial link between the scattering signal and true microstructure, validating SARB as more than a pretty coloring trick.
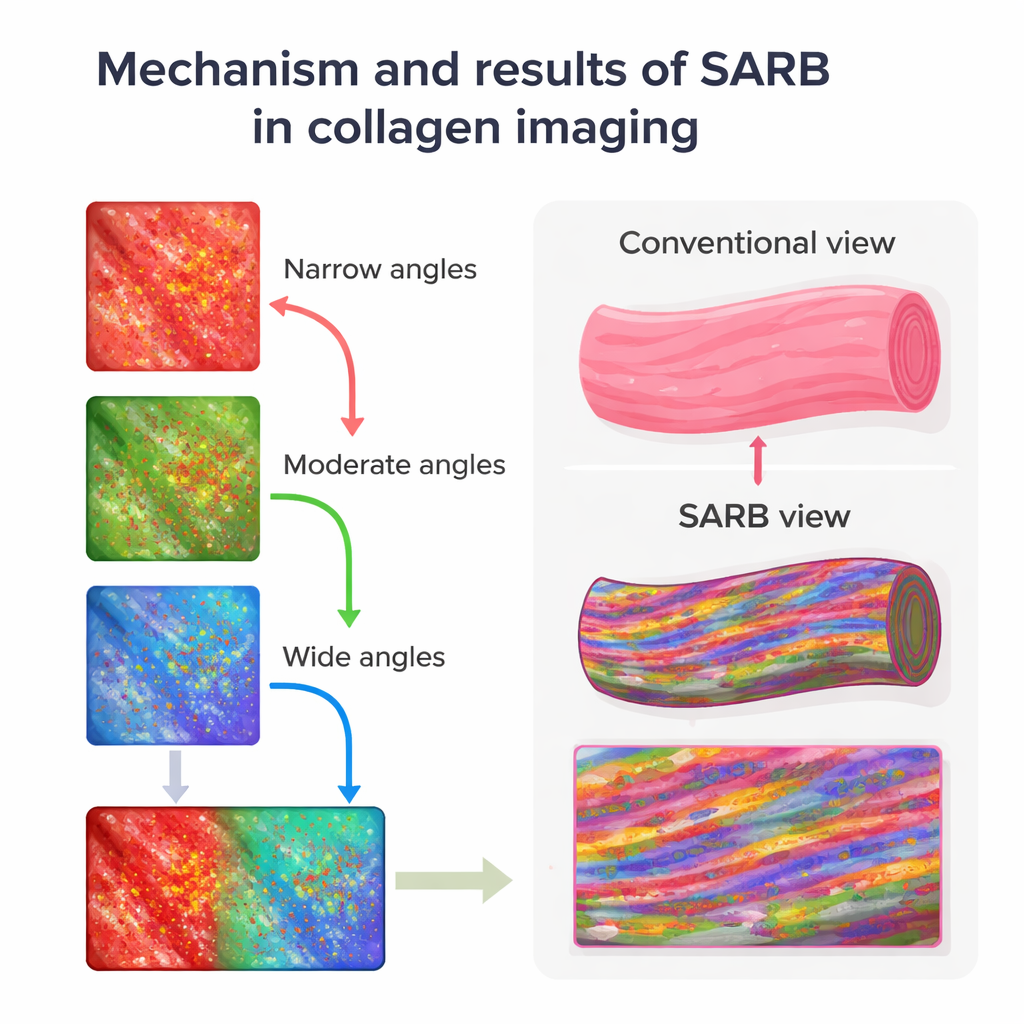
Seeing how collagen ages and loosens
The team then asked whether SARB could detect known changes in collagen with aging. Comparing skin from young and old mice, conventional Picrosirius red staining under polarized light mainly showed a shift in collagen types—from one type to another—but gave limited insight into how fibers were arranged. SARB, by contrast, revealed that aged skin had larger gaps between collagen bundles and fewer internal scattering signals, suggesting looser or more disorganized fibrils. Electron microscopy supported this impression, showing more irregular fibers in older animals. By converting SARB images into black-and-white maps and measuring the fraction of strongly scattering areas, the researchers found that older mice had a significantly lower “high-scattering area” ratio, providing a simple number that tracked structural decline.
From lab curiosity to practical tool
Because SARB is built on a regular transmitted-light microscope with an added patterned filter and camera, it could, in principle, be deployed in many labs that already handle paraffin-embedded tissue. It avoids time-consuming staining, reduces variability between laboratories, and produces both visual and quantitative readouts of collagen organization. While more work is needed to relate specific scattering signatures to exact fiber thickness, density, and 3D arrangement, this study shows that scattered light from unstained sections can serve as a sensitive, label-free marker of collagen health. In the future, SARB could help screen antifibrotic or anti-aging treatments, monitor wound healing, and perhaps flag early tissue changes in cancers—all by extracting new structural information from the tissue slides pathologists already prepare.
Citation: Otaki, M., Shimano, M., Asano, Y. et al. Novel histological observation method using the scattered light of unstained collagen sections. Sci Rep 16, 7574 (2026). https://doi.org/10.1038/s41598-026-38504-9
Keywords: collagen imaging, fibrosis, skin aging, light scattering, microscopy