Clear Sky Science · en
Targeted HDAC8 inhibition with non-hydroxamate [1,2,4]triazolo[4,3-a] quinoline compounds
New Hope for a Childhood Cancer
Neuroblastoma is an aggressive childhood cancer that arises from nerve cells and is often difficult to treat once it has spread. This study explores a new way to slow down or kill neuroblastoma cells by turning off a key cellular switch called HDAC8. By building a new family of drug-like molecules that act very selectively on this switch, the researchers aim to create treatments that are both more effective against tumors and gentler on the rest of the body.
A Molecular Switch at the Heart of Cancer
Inside our cells, genes are constantly being turned on and off. One important control system relies on tiny chemical tags called acetyl groups that attach to proteins associated with DNA. Enzymes called histone deacetylases (HDACs) remove these tags, tightening the packaging of DNA and usually dialing down gene activity. HDAC8 is one member of this enzyme family and has been linked to several cancers, including neuroblastoma. When HDAC8 is overactive, cancer cells can grow, survive, and spread more easily, making it an attractive target for new drugs.
Limitations of Older HDAC Drugs
Several HDAC-blocking drugs are already approved for some blood cancers, but they tend to hit many HDAC types at once. Most of these medicines use a hydroxamate chemical group to grip a metal ion in the enzyme’s active site. While effective, this group binds metals so strongly that it can lead to side effects and limits how precisely the drug can distinguish between different HDACs. As a result, patients may experience toxicity, and normal cells can be harmed along with cancer cells. Researchers have therefore been searching for alternative chemical designs that keep the cancer-fighting benefits while reducing unwanted damage.
Designing a New Class of Targeted Inhibitors
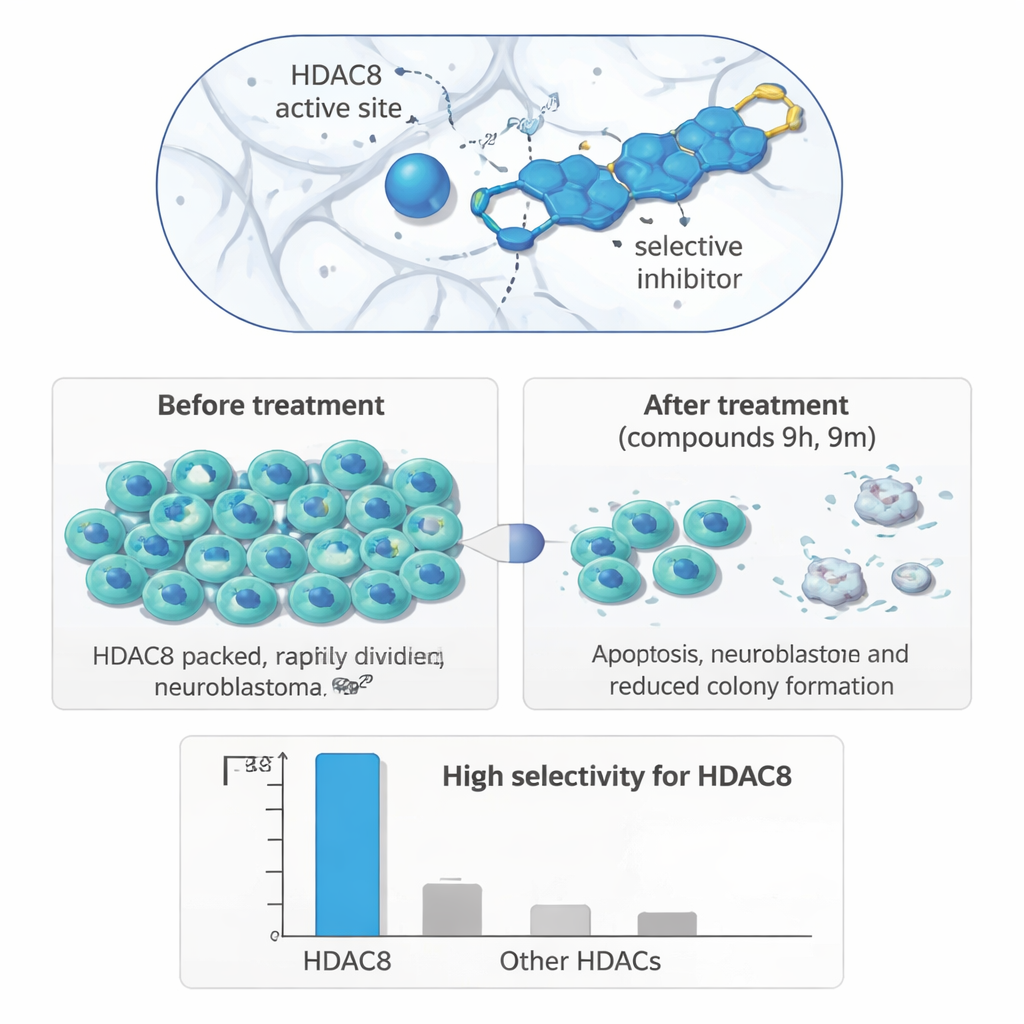
In this work, the team designed and synthesized 21 new compounds built around a triazoloquinoline scaffold, connected through a flexible linker to an "+alpha-amino amide" region that can bind the zinc atom in HDAC8’s active site. Using computer-guided design, they chose this architecture to fit into a narrow “acetate release channel” unique to HDAC8, a structural feature that helps achieve high specificity. Detailed docking studies and molecular dynamics simulations showed that several of the new molecules—especially those called 9m and 9r—form stable, long‑lasting interactions inside the HDAC8 pocket and bind as tightly as, or tighter than, a well-known reference inhibitor. Importantly, biochemical tests confirmed that the best compounds strongly block HDAC8 while leaving other HDAC family members largely untouched.
From Test Tube to Tumor Cells
To see whether this precise enzyme targeting translates into useful biological effects, the researchers tested their compounds on a panel of human cell lines. In neuroblastoma cells (IMR‑32), two molecules, 9h and 9m, clearly stood out: they reduced cell growth at micromolar concentrations and did so more strongly in neuroblastoma than in breast or colon cancer cells, or in noncancerous kidney cells. Long-term colony formation experiments showed that treated neuroblastoma cells lost much of their ability to regrow after drug exposure. In a wound-healing assay, which tracks how quickly cells migrate to close a scratch in a cell layer, both compounds slowed the movement of neuroblastoma cells, hinting at reduced metastatic potential.
Probing How the Compounds Kill Cancer Cells
Flow cytometry experiments revealed that 9h and 9m trigger programmed cell death (apoptosis) in neuroblastoma cells and cause an accumulation of cells in a phase of the cell cycle associated with DNA damage or death (Sub‑G1). To confirm that these effects truly stem from HDAC8 blockade, the team measured the acetylation state of a protein called SMC3, a known HDAC8 target involved in keeping sister chromosomes paired during cell division. When HDAC8 is inhibited, acetylated SMC3 builds up. After treatment with 9h or 9m, acetylated SMC3 levels rose sharply, while total SMC3 protein remained unchanged—strong evidence that these compounds directly hit HDAC8 in living cells and disrupt its normal function in chromosome control.
What This Could Mean for Future Treatments
Taken together, the chemistry, computer modeling, enzyme tests, and cell-based experiments paint a consistent picture: the newly developed triazoloquinoline compounds are potent, highly selective blockers of HDAC8 that can curb the growth and spread of neuroblastoma cells while sparing other HDAC enzymes. Because they avoid the conventional hydroxamate group, they may offer a safer route to harness HDAC8 as a therapeutic target. Although much work remains—especially studies in animals and eventual clinical trials—this research lays a solid foundation for developing more precise drugs against childhood neuroblastoma and possibly other HDAC8-driven diseases.
Citation: Bandaru, N.V.M.R., Fathima, A., Sengar, S. et al. Targeted HDAC8 inhibition with non-hydroxamate [1,2,4]triazolo[4,3-a] quinoline compounds. Sci Rep 16, 7472 (2026). https://doi.org/10.1038/s41598-026-38490-y
Keywords: HDAC8 inhibitors, neuroblastoma, epigenetic therapy, triazoloquinoline, targeted cancer drugs