Clear Sky Science · en
GluN2A-mediated currents and calcium signal in human iPSC-derived neurons
Why tiny lab-grown brain cells matter
Scientists are increasingly turning to lab-grown human brain cells to study how our brains develop and to probe what goes wrong in conditions such as autism, epilepsy, and Alzheimer’s disease. This study asks a simple but crucial question: do these lab-made neurons grow up and wire themselves in ways that truly resemble the human brain, especially in how they handle a key chemical signal called glutamate? The answer reveals how to build more realistic “mini-brain” models and points to new ways of examining brain health and disease.
Building neurons from reprogrammed cells
The researchers started with human induced pluripotent stem cells—ordinary cells that have been “reprogrammed” back into a stem-cell state. From there, they steered the cells toward becoming neural precursor cells and finally into networks of neurons and supporting astrocytes, mimicking the developing human cortex. They compared two widely used recipes, or culture conditions. One, called BrainPhys medium, is designed to promote active, electrically responsive neurons and tends to produce more astrocytes. The other, a more traditional neural maintenance medium, favors neurons with fewer astrocytes. Over sixty days, the team tracked how these cultures changed in appearance, gene activity, and cell types, finding that BrainPhys-grown cultures developed more elaborate neuron shapes and a neuron-to-astrocyte ratio closer to that of the real human brain.
Signs that the cells are growing up
To see how “mature” the neurons were, the team measured global gene activity using RNA sequencing at day 60. Cells grown in BrainPhys showed almost two thousand genes expressed at different levels compared with the standard medium, with many linked to neuron differentiation, synapse formation, and electrical activity. Genes associated with astrocyte function and inflammation were also higher, reflecting the larger astrocyte population. Together, these patterns suggest that the BrainPhys cultures better recapitulate later stages of brain development, with more refined communication points between neurons and a supporting environment that more closely resembles living tissue.
Key switches in chemical communication
A hallmark of brain maturation is a shift in the makeup of NMDA receptors—molecular gates on neurons that open in response to glutamate and help set up learning-related connections. Early in development, a subunit called GluN2B dominates; later, GluN2A takes over, changing how long and how strongly the receptors stay open. Using microscopy, the team showed that over time more NMDA receptors moved into synapses, the contact points between neurons, especially in BrainPhys cultures. Targeted gene measurements revealed a drop in the “early” GluN2B subunit and a rise in the “later” GluN2A subunit, indicating that the neurons were undergoing this classic developmental switch.
Following currents and calcium inside the cells
Functional tests confirmed that these molecular changes mattered. With fine-tipped electrodes, the researchers recorded electrical currents triggered by NMDA, a drug that selectively activates NMDA receptors. Neurons from both culture methods showed robust currents, but blocking the GluN2A subunit reduced these currents by roughly half, demonstrating that GluN2A was now a major player in signal flow. The team then tracked calcium, a key internal messenger, using a fluorescent dye. When exposed to NMDA, a far larger fraction of BrainPhys-grown neurons showed strong calcium surges, and the size of the signal was greater. Gene analysis suggested why: BrainPhys cultures upregulated multiple components of calcium channels and release pathways, equipping the cells to translate NMDA receptor activation into broad, coordinated calcium signals.
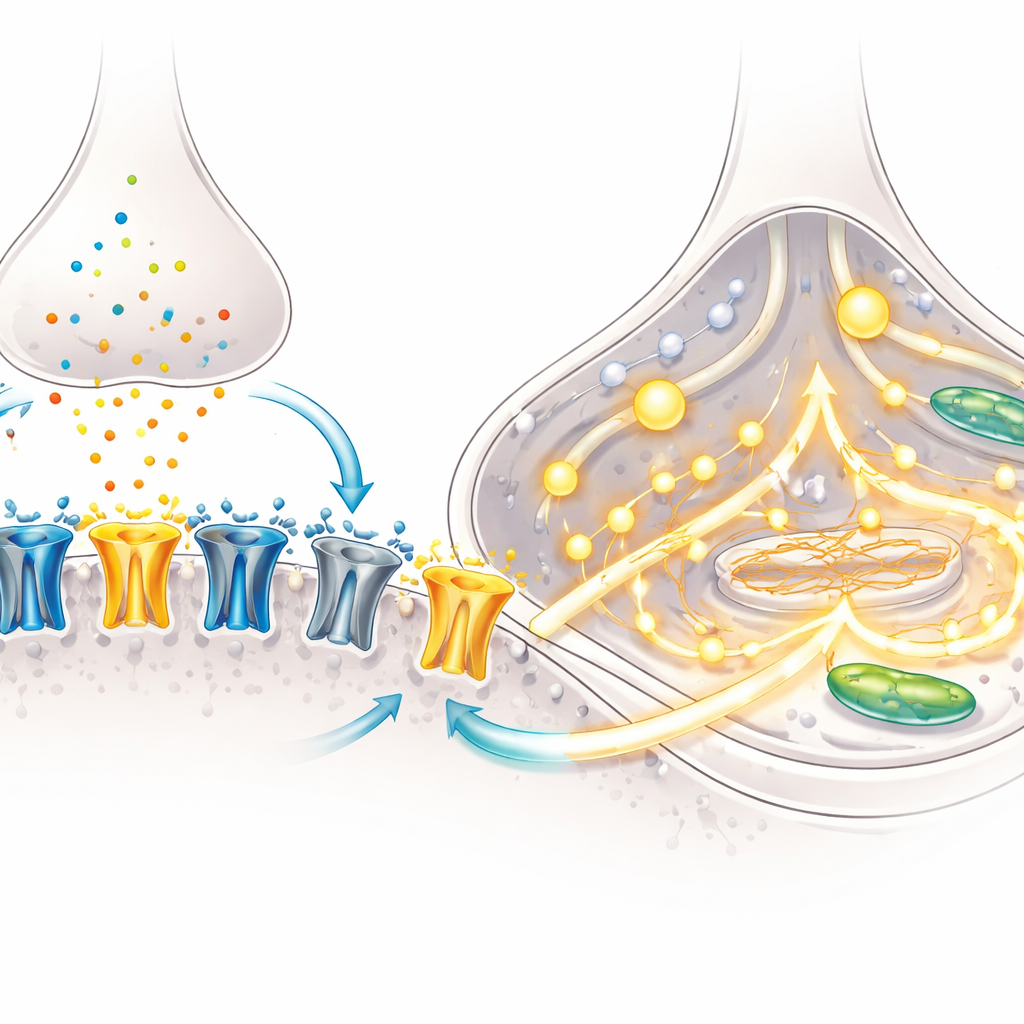
What this means for studying brain disorders
Together, these findings show that human stem-cell-derived neurons can reach a functionally mature state in just two months, especially when grown under conditions that favor active networks and a realistic mix of neurons and astrocytes. In this state, the “adult-like” GluN2A subunit dominates NMDA receptor currents and contributes strongly to calcium signaling, much as in the developing human brain. For non-specialists, the takeaway is that researchers now have a more faithful lab model of how human excitatory neurons grow up, wire together, and respond to glutamate. This makes it possible to probe how subtle genetic changes or disease processes disrupt these finely tuned switches, opening a window onto conditions ranging from epilepsy to Alzheimer’s disease and guiding the design and testing of future therapies.
Citation: Escamilla, S., Avilés-Granados, C., Peralta, F.A. et al. GluN2A-mediated currents and calcium signal in human iPSC-derived neurons. Sci Rep 16, 9736 (2026). https://doi.org/10.1038/s41598-026-38482-y
Keywords: NMDA receptors, stem cell neurons, brain development, calcium signaling, synaptic maturation