Clear Sky Science · en
The dual G9a inhibitor and histamine H3 receptor antagonist A-366 improves repetitive and social behaviors and attenuates neuroinflammation in BTBR T + tf/J mice
Why this mouse study matters for autism
Families living with autism spectrum disorder (ASD) often face a patchwork of treatments that ease only a few symptoms at a time. This study explores a new experimental compound, called A-366, that was designed to act on two very different biological systems at once. In a widely used mouse model of autism-like behavior, A-366 reduced repetitive actions, improved social interaction, and calmed brain inflammation, hinting at a future in which a single medicine might tackle several core features of ASD together.
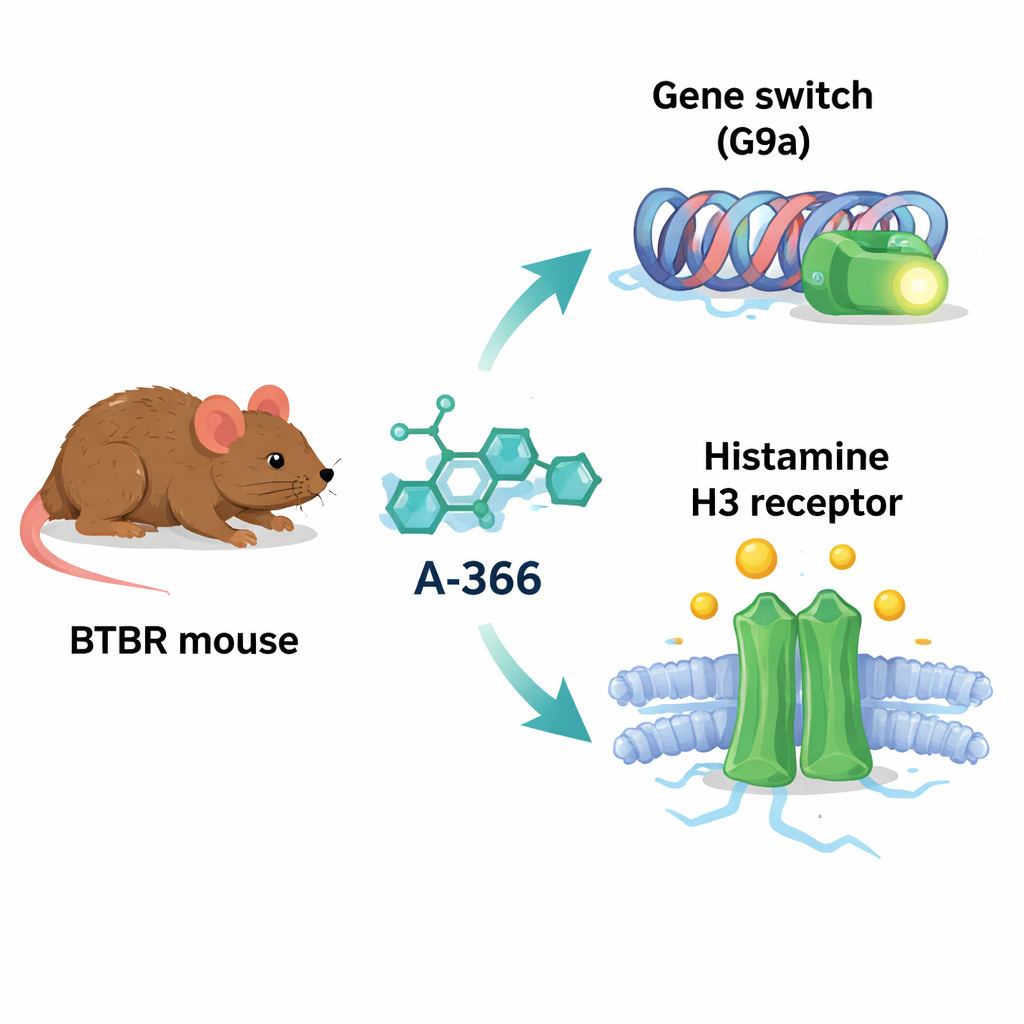
A condition with many moving parts
ASD is not a single disease with a single cause. It involves difficulties with social communication, repetitive behaviors, and often other challenges such as anxiety or attention problems. Behind these outward signs lies a tangle of changes in brain chemistry and gene regulation. Current drugs, like the antipsychotic aripiprazole, mainly target brain messenger systems such as dopamine and serotonin, and are approved only for associated symptoms like irritability. At the same time, research has revealed that the way DNA is packaged and chemically marked inside brain cells—epigenetics—also shifts in ASD, as does the activity of the brain’s own immune and inflammatory responses. These multiple layers suggest that a “one-target-at-a-time” approach may be too narrow.
Hitting both gene switches and brain messengers
A-366 was originally developed to block a protein called G9a, an enzyme that adds chemical tags to histone proteins and tends to turn genes down. In several brain disorders, G9a activity and its marks are increased, potentially silencing genes important for learning, memory, and healthy brain wiring. Intriguingly, the chemical shape of A-366 also resembles that of drugs that block the histamine H3 receptor, a switch that controls the release of histamine and other transmitters and has been linked to attention, wakefulness, and social behavior. Because of this overlap, the authors suspected that A-366 might act as a “dual agent”: loosening harmful gene silencing through G9a inhibition while at the same time boosting histamine signaling by blocking H3 receptors.
Putting A-366 to the test in autistic-like mice
The team worked with BTBR T+tf/J mice, a strain that naturally shows low sociability, strong repetitive grooming and digging, and elevated brain inflammation—features that echo key aspects of ASD. Male BTBR mice received daily injections of A-366 for three weeks at three different doses, and their behavior was compared with typical C57 mice and with BTBR mice treated with either pitolisant (a standard H3 receptor blocker) or aripiprazole. Across several tasks, A-366 reduced repetitive digging, shredding, and self-grooming in a dose-dependent fashion and improved performance in a Y-maze test of spatial working memory. In a three-chamber social test, A-366 not only restored the mice’s preference for spending time with another mouse over an empty cage, but at the highest dose brought social scores up to the level of typical mice and surpassed the effects of pitolisant and aripiprazole.
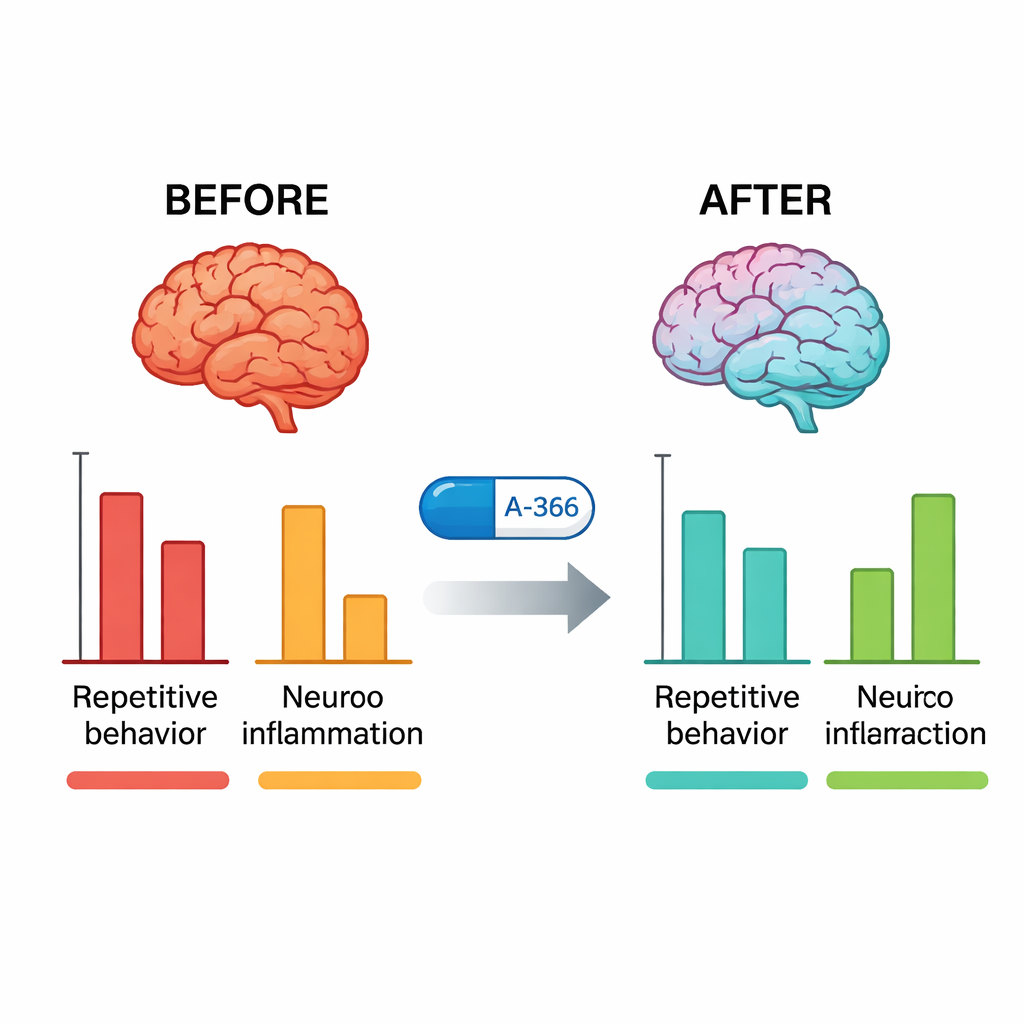
Quieting inflammation and confirming the targets
After behavioral testing, the researchers examined the mice’s brains. In untreated BTBR mice, G9a activity in the hippocampus and cerebellum was markedly higher than in typical mice. Treatment with A-366 sharply reduced this activity back toward normal, whereas pitolisant alone did not, confirming that A-366 truly engages its epigenetic target. At the same time, key inflammatory molecules—TNF-α, IL-6, and IL-1β—were greatly elevated in BTBR mice and were significantly lowered by A-366, more so than by aripiprazole. When the scientists added a drug that activates H3 receptors (RAMH) on top of A-366, the behavioral and anti-inflammatory benefits were only partly reversed, and the drop in G9a activity remained. This pattern supports a combined mechanism: stable epigenetic relief through G9a inhibition plus a histamine-mediated contribution through H3 receptor blockade.
What this could mean for future treatments
To a non-specialist, these findings suggest that it may be possible to design medicines that work on both the “software” and the “messaging system” of the brain at once. In this mouse model of autism-like behavior, A-366 eased repetitive actions, improved social engagement, and reduced signs of chronic brain inflammation by simultaneously loosening harmful gene shutoffs and adjusting histamine signaling. A-366 itself was built for other diseases and would need optimization and extensive safety testing before any use in people. Still, it provides a blueprint for a new class of multi-target drugs aimed at the complex biology of ASD, where addressing only one pathway at a time has so far offered limited relief.
Citation: Hajar, M., Jayaprakash, P., Stark, H. et al. The dual G9a inhibitor and histamine H3 receptor antagonist A-366 improves repetitive and social behaviors and attenuates neuroinflammation in BTBR T + tf/J mice. Sci Rep 16, 7105 (2026). https://doi.org/10.1038/s41598-026-38481-z
Keywords: autism spectrum disorder, epigenetic therapy, histamine H3 receptor, neuroinflammation, mouse model