Clear Sky Science · en
aFGF rescues high glucose-induced senescent fibroblasts and improves diabetic wound healing by regulating SIRT1/STAT3 pathway
Why slow-healing wounds matter
For many people with diabetes, a small cut on the foot or leg can turn into a stubborn wound that lingers for months, sometimes leading to infection or even amputation. These chronic sores are not just a surface problem: the skin cells that should rebuild the damaged tissue often grow old before their time and stop working properly. This study explores a promising way to “rejuvenate” some of those cells using a naturally occurring repair signal, with the goal of helping diabetic wounds close faster and more completely.
Cells that grow old too soon
Healthy skin relies on fibroblasts—support cells that live in the deeper layers of the skin—to multiply, move into a wound, and lay down fresh scaffolding for new tissue. In diabetes, long-term high blood sugar and sugar-damaged molecules push these fibroblasts into a state called cellular aging or senescence. Senescent fibroblasts no longer divide well, produce less of the structural proteins needed for repair, and instead release a cocktail of irritation-promoting factors that fuel ongoing inflammation. The authors show that in diabetic rats, skin around a wound contains more of these aged fibroblasts and fewer of the helpful matrix proteins, correlating with much slower healing.
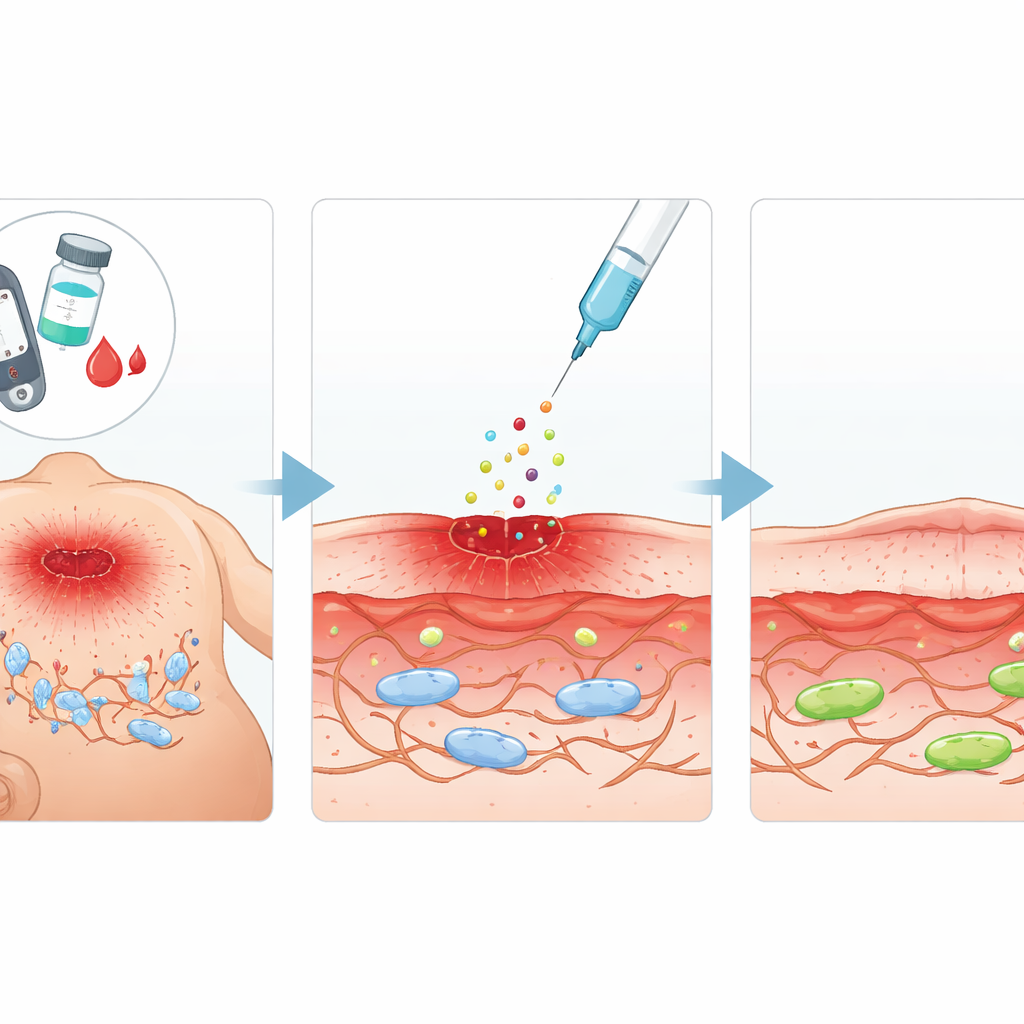
A repair signal with promise
The team focused on acidic fibroblast growth factor (aFGF), a naturally occurring protein known to encourage cell growth and tissue repair. They wondered whether aFGF could not only boost fibroblast activity, but also reverse aging-like changes caused by high sugar. In diabetic rats, they created standardized skin wounds and injected aFGF around the injured area every few days. Compared with untreated diabetic animals, aFGF-treated rats showed faster shrinkage of the wound surface, better rebuilding of the underlying tissue, and lower levels of markers that signal cellular aging. In parallel lab experiments using a mouse fibroblast cell line exposed to very high glucose, aFGF restored the cells’ ability to divide and migrate—two behaviors essential for closing a wound.
Calming oxidative stress inside the cells
High blood sugar does more than just bathe cells in extra fuel: it also drives the overproduction of reactive oxygen species, unstable molecules that damage proteins, fats, and DNA. This oxidative stress is a major trigger for fibroblasts to become senescent. The researchers measured several standard indicators of this stress in their cell model, including damaging byproducts and the activity of the cell’s own antioxidant enzymes. Under high glucose, fibroblasts showed more damage and weaker antioxidant defenses. When aFGF was added, the balance shifted: harmful markers fell, and protective enzymes became more active. This suggests that aFGF helps restore a healthier internal environment, making it less likely that fibroblasts will tip into a permanently aged state.
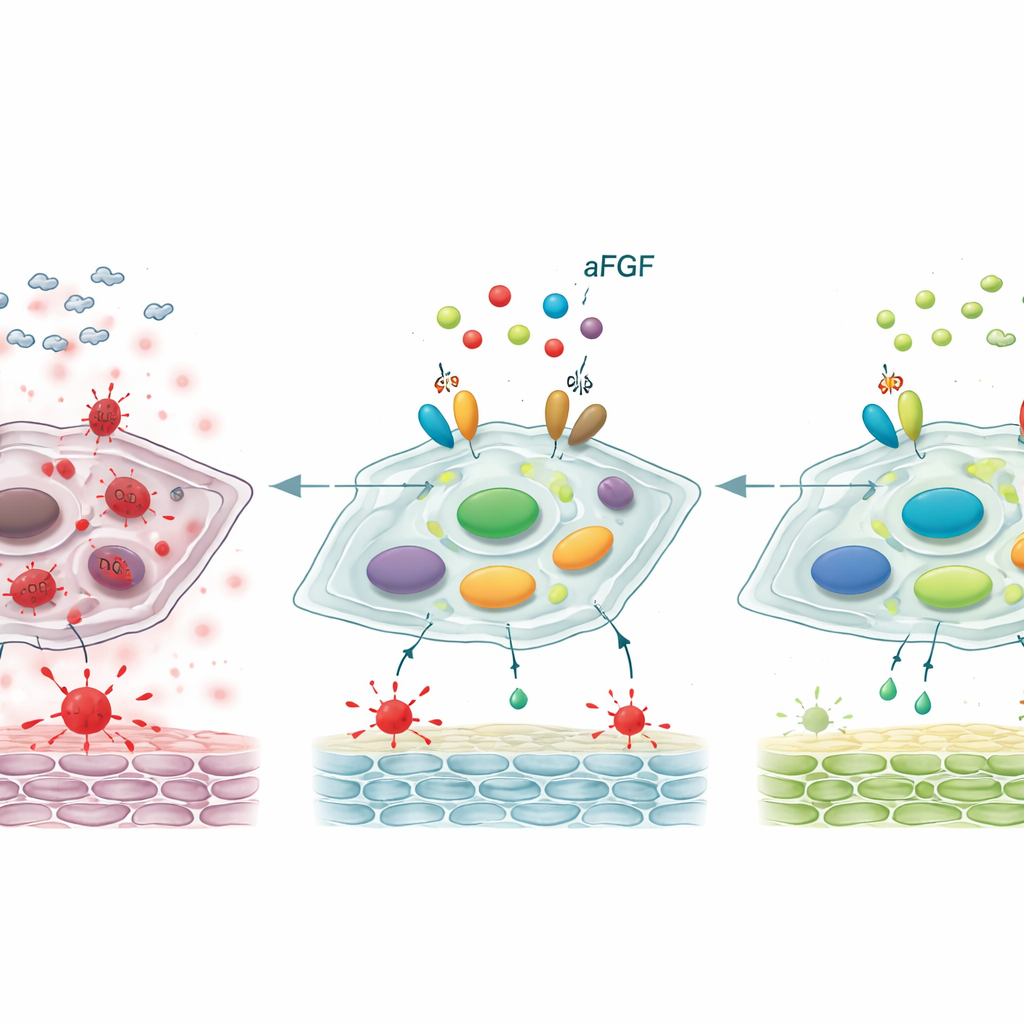
Resetting a key control switch
Digging deeper, the scientists examined a molecular control axis involving two proteins, SIRT1 and STAT3, that together influence whether fibroblasts stay youthful or become senescent. In diabetic wounds and high-glucose–treated cells, levels of SIRT1, a guardian associated with stress resistance, were reduced, while the activated form of STAT3—a driver of aging and inflammatory behavior—was increased. Treatment with aFGF flipped this pattern: SIRT1 rose, and activated STAT3 fell. When the team blocked SIRT1 with a specific inhibitor, aFGF largely lost its ability to reduce oxidative stress, dampen aging markers, and speed wound closure in rats. This indicates that aFGF’s beneficial effects depend strongly on reawakening SIRT1, which in turn reins in STAT3 and the harmful aging program it controls.
What this could mean for patients
Taken together, the findings suggest that aFGF does more than simply push cells to grow. It appears to rescue overworked fibroblasts in diabetic wounds from a destructive aging cycle, easing oxidative stress and resetting a key molecular switch so that these cells can once again help rebuild tissue. While the work was done in rats and cultured cells, and real diabetic wounds are even more complex than the models used here, the results point to aFGF as a promising candidate for future treatments. If similar benefits are confirmed in humans, carefully targeted aFGF therapy might one day help chronic diabetic ulcers heal faster, reducing pain, infection risk, and the chance of serious complications.
Citation: Wang, X., Lu, M., Jia, S. et al. aFGF rescues high glucose-induced senescent fibroblasts and improves diabetic wound healing by regulating SIRT1/STAT3 pathway. Sci Rep 16, 7856 (2026). https://doi.org/10.1038/s41598-026-38480-0
Keywords: diabetic wound healing, fibroblast aging, oxidative stress, growth factor therapy, SIRT1 STAT3 pathway