Clear Sky Science · en
Safety and preliminary efficacy of adding tocilizumab to cisplatin/docetaxel for the treatment of locally advanced triple-negative breast cancer patients: prospective phase 1/2 clinical trial
Why this study matters to patients and families
Triple-negative breast cancer is one of the most challenging forms of breast cancer to treat. It tends to grow quickly, comes back more often, and lacks the usual hormone or HER2 “handles” that many modern drugs grab onto. This study tested whether adding an immune‑modifying drug called tocilizumab, already used for arthritis and other inflammatory conditions, could make standard chemotherapy work better and more safely for people with advanced triple-negative breast cancer in the breast and nearby lymph nodes.
A tough cancer with limited options
Triple-negative breast cancer accounts for only about one in five breast cancers but causes a disproportionate share of deaths because it is aggressive and has fewer effective targeted treatments. For many patients whose tumors are large or have spread to nearby lymph nodes, the main option is strong chemotherapy given before surgery to shrink the cancer. Even with modern regimens that include platinum drugs like cisplatin, a significant number of patients are left with residual tumor after treatment, which is linked to a higher risk of the cancer returning.
Turning down a harmful signal
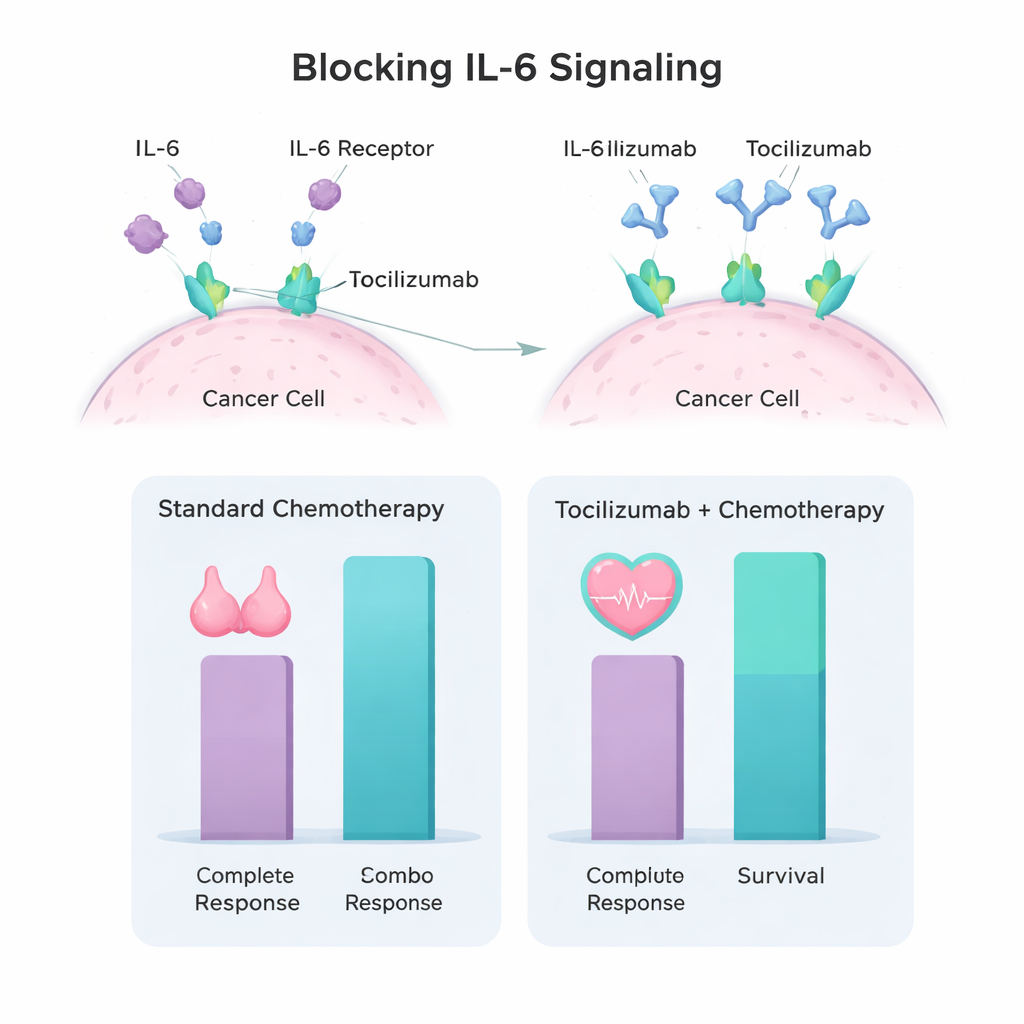
Scientists have found that triple-negative tumors and the cells surrounding them often produce high levels of a signaling protein called interleukin‑6 (IL‑6). IL‑6 acts like a constant “grow and resist treatment” message for the cancer. Tocilizumab is an antibody drug that blocks the IL‑6 receptor, shutting down this signal. Previous laboratory and animal studies from the same research group showed that giving tocilizumab before cisplatin made triple-negative breast cancer cells more sensitive to chemotherapy and slowed tumor growth. These findings led the team to design a first‑in‑human clinical trial to see if this approach was safe and showed early signs of benefit in patients.
How the trial was carried out
The study was a single‑center phase 1/2 trial in Riyadh that enrolled 30 women with locally advanced triple-negative breast cancer (tumor size at least 4 cm, no distant spread). All patients received tocilizumab by vein on day 1, followed by cisplatin and docetaxel on day 2, every four weeks for six cycles. The first part of the trial used a standard “3+3” dose‑escalation design to find a safe chemotherapy dose to pair with a fixed dose of tocilizumab. Once that dose was chosen, more patients were treated in an expansion phase. After chemotherapy, patients proceeded to surgery, and pathologists carefully examined breast tissue and lymph nodes to determine whether any invasive cancer remained. The main goals were to assess safety and to measure how many patients achieved a pathological complete response—meaning no detectable invasive cancer in the breast or underarm lymph nodes.
What the researchers found
Of the 30 enrolled patients, 28 completed treatment and 26 underwent surgery. The group was relatively young (median age 42 years), and most had stage III disease and lymph node involvement at the start—features usually linked to poorer outcomes. Overall, 62% of patients had no remaining invasive cancer after treatment, and 81% had no cancer in the lymph nodes. When near‑complete responses (tiny residual spots of tumor with clean lymph nodes) were included, 81% of patients fell into the complete or near‑complete response category, and none of these patients had a recurrence during the follow‑up period. Patients carrying inherited BRCA1 or BRCA2 mutations did especially well: all of them achieved complete response. Early survival results were encouraging, with estimated three‑year disease‑free survival of 80% and overall survival of 90%, even though many patients started with very advanced local disease.
Side effects and safety
Safety was a key focus because the regimen combined a biologic drug with intensive chemotherapy. Among the 28 patients evaluated, there were no treatment‑related deaths. The most common mild to moderate side effects were nausea, joint and muscle pain, vomiting, diarrhea, rash, and temporary changes in blood counts. More serious events included low white blood cell counts and fever, liver enzyme elevations, and significant but controllable increases in cholesterol and triglycerides—changes that are known with tocilizumab and were managed with standard medications. Only a few patients needed chemotherapy dose reductions, and kidney problems from cisplatin were rare and reversible, suggesting that the overall combination was tolerable.
What this could mean going forward
For patients and families facing triple-negative breast cancer, these early results suggest that adding an IL‑6–blocking drug before standard chemotherapy may substantially increase the chances that no cancer is found at surgery, particularly in people with BRCA mutations, and may do so without adding dangerous side effects. However, the study was small, conducted at a single center, and did not allow certain modern add‑on treatments for patients with residual disease. Tocilizumab was also stopped after surgery, so longer‑term benefits remain to be fully defined. Larger, randomized trials will be needed to confirm whether this approach should become part of standard care, but this work opens a promising new avenue: targeting inflammatory signals in and around the tumor to help existing chemotherapy work harder and more precisely against a very difficult‑to‑treat cancer.
Citation: Al-Tweigeri, T., Tulbah, A., Akhtar, S. et al. Safety and preliminary efficacy of adding tocilizumab to cisplatin/docetaxel for the treatment of locally advanced triple-negative breast cancer patients: prospective phase 1/2 clinical trial. Sci Rep 16, 7029 (2026). https://doi.org/10.1038/s41598-026-38465-z
Keywords: triple-negative breast cancer, tocilizumab, IL-6 blockade, neoadjuvant chemotherapy, pathologic complete response