Clear Sky Science · en
Controlling sodium silicate sol-gel transition time through encapsulation of hydrochloric acid using tunable polymeric microcapsules
Why timing matters when liquids turn into gels
Many everyday and industrial materials start out as thin liquids and then slowly thicken into gels. In oil and gas wells, this transformation is used on purpose: special liquids are pumped underground so they can solidify and plug unwanted cracks in the rock, steering water and oil along better paths. The challenge is timing. If the liquid turns into a gel too soon, it clogs the wellbore; too late, and it races past the target zone. This study explores a way to put that “gel switch” on a timer using microscopic capsules that carry acid and break open only when and where they are needed.
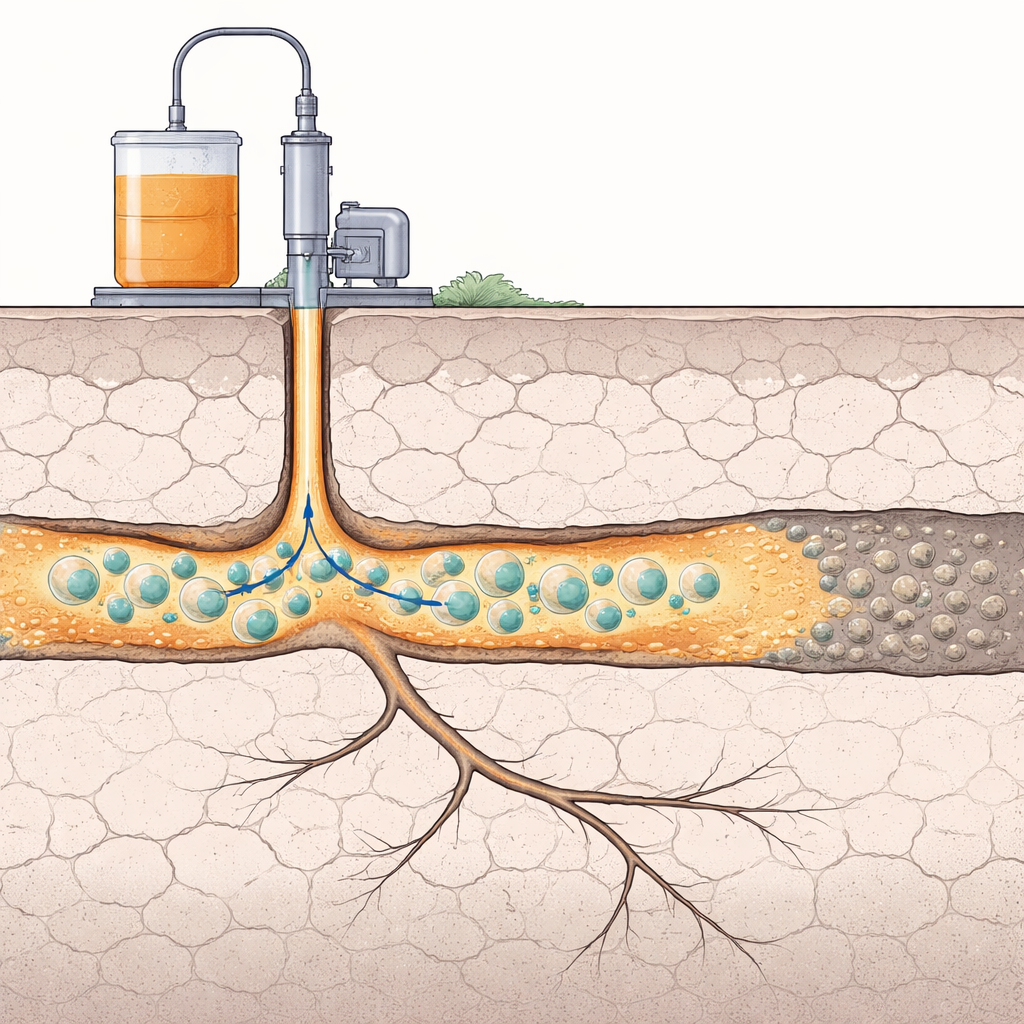
A smart way to plug leaky underground pathways
The work focuses on sodium silicate, a water-based liquid that can be turned into a solid-like gel by adding an acid such as hydrochloric acid. Because sodium silicate is stable, inexpensive and relatively environmentally friendly, it is widely used in detergents, construction materials, and especially in oil and gas wells to seal fractures and high‑permeability zones. But in real underground formations, temperature, salt content, and rock chemistry can all change how fast the gel forms, making it hard to predict where the plugging will actually occur. The authors propose separating the silicate liquid from the acid that triggers gelation, and enclosing the acid inside tiny polymer shells so that the gel forms only after a controllable delay.
Tiny shells that carry acid on a timer
To build this timer, the team used microfluidic devices—glass capillary systems that can generate extremely uniform droplets—to fabricate microscopic capsules made of a rubbery silicone material called PDMS. Each capsule has an inner droplet of concentrated hydrochloric acid surrounded by a PDMS shell and suspended in water. By adjusting the flow rates and the mixing ratio of PDMS base to curing agent, they could tune three key features of the capsules: shell thickness, shell stiffness (how rigid or soft it is), and whether the inner droplet sits right in the center or off to one side (eccentricity). These design choices allowed the researchers to create “thin,” “thick,” and “eccentric” capsules with different mechanical strengths and response to stress.
How water inflow makes capsules burst
When these capsules are transferred from a sugary storage solution into sodium silicate solution, they suddenly find themselves in a less concentrated environment. Water naturally flows through the PDMS shell into the more concentrated acid core, causing the capsule to swell. If the shell is thin or soft, it stretches and ruptures relatively quickly, releasing the acid; if it is thick or stiff, it can resist swelling for much longer. The released acid then mixes with the surrounding sodium silicate, lowers its pH, and triggers the chemical reactions that turn the liquid into a gel network. In this way, the physical design of each capsule programs how long it will wait before “switching on” the gelation process.

Measuring when liquid becomes solid
To track when the gel starts to form, the authors introduced a sensitive but simple method based on a tensiometer and a thin plate (the Wilhelmy plate method). As the plate repeatedly moves into and out of the sample, the instrument measures the vertical force on the plate. While the solution is still liquid, this force stays nearly constant. Once a gel network develops, the plate begins to drag on the material and the measured force rises sharply, signaling the sol‑gel transition time. Using this approach, the researchers compared sodium silicate mixed directly with free acid—where gelation began in about eight minutes—with mixtures in which all the acid was trapped in capsules.
Turning minutes into days with tailored capsules
The results show that encapsulating the acid can stretch the gelation time from minutes to many hours or even days. Thin‑shelled and softer capsules burst sooner, giving transition times on the order of one day, while thick, stiffer shells delayed gel formation for almost four days at room temperature. Eccentric capsules, with uneven shell thickness, produced intermediate delays. Temperature also mattered: at 60 °C, similar to many underground reservoirs, even the thick, stiff capsules ruptured much faster, and gelation began after about five hours instead of ninety. Across all tests, shell thickness emerged as the single strongest lever for tuning when the gel starts to form.
What this means for real‑world use
For non‑specialists, the key message is that the authors have built a microscopic “time‑release” system for turning a flowing liquid into a solid plug deep underground. By packing acid into tiny, tunable capsules rather than mixing it directly with sodium silicate, engineers could choose whether gelation begins within minutes, hours, or days, and adjust this timing for different reservoir temperatures and conditions. This level of control could improve how efficiently oil and gas wells are sealed and managed, and the same principle—using adjustable microcapsules to schedule when a reaction starts—may be useful in many other technologies where it matters exactly when and where a liquid turns into a solid.
Citation: Lima, M., Pessoa, A.C.S.N., de Medeiros, A. et al. Controlling sodium silicate sol-gel transition time through encapsulation of hydrochloric acid using tunable polymeric microcapsules. Sci Rep 16, 8094 (2026). https://doi.org/10.1038/s41598-026-38462-2
Keywords: sodium silicate gels, microcapsules, controlled gelation, oil and gas reservoirs, osmotic release