Clear Sky Science · en
Facile sol–gel fabrication of MnOx/Graphite nanostructured electrodes for sustainable produced water treatment
Cleaning Up a Hidden Pollution Problem
Every barrel of oil pulled from the ground brings up far more “produced water” — a salty, dirty mix laced with oil residues and stubborn organic chemicals. This waste stream is often hard to clean and can threaten rivers, farms, and seas if not treated properly. The study behind this article introduces a new, relatively simple kind of electrical filter that can strip much of this pollution from produced water, offering a path toward safer reuse and discharge.
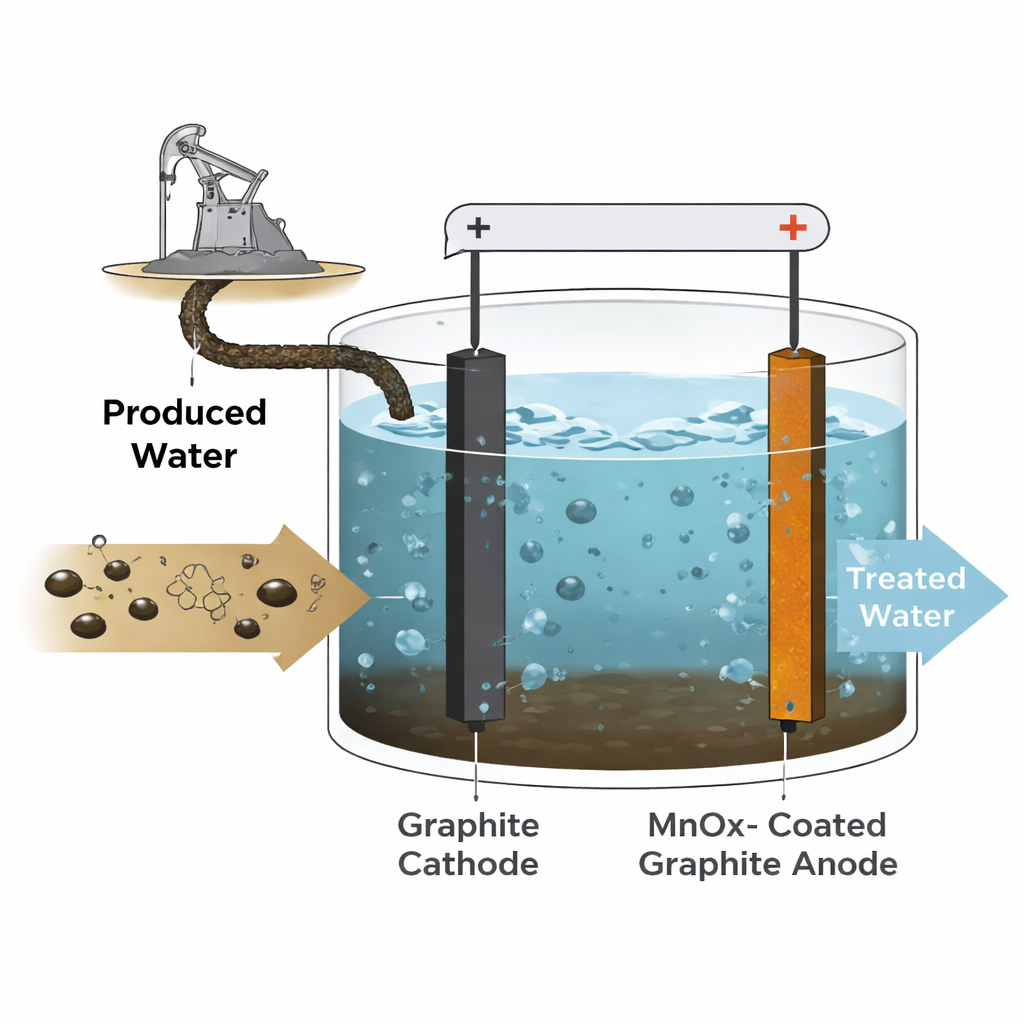
A Tough Wastewater Challenge
Produced water from oil and gas wells is not just salty; it also carries a cocktail of dissolved organics, tiny oil droplets, and other contaminants. Standard treatment methods such as biological reactors and membranes often struggle with such harsh mixtures. High salinity can poison microbes, and sticky organics quickly foul filters. Regulators, however, are tightening limits on how much organic material, often measured as chemical oxygen demand (COD), can be released into the environment. This pressure has pushed engineers to search for robust, low-sludge technologies that can polish the final effluent before it is discharged or reused.
Cleaning with Electricity Instead of Chemicals
Electrochemical oxidation tackles the problem by passing an electric current through the water between two electrodes. Unwanted organic molecules are broken down either directly on the surface of the positive electrode or indirectly by powerful oxidants formed in the salty water. The trick is finding an electrode material that is efficient, durable, and affordable. Some of today’s best-performing electrodes use exotic or toxic materials, such as lead-based oxides or boron-doped diamond, which are expensive or raise safety concerns. Manganese oxides, in contrast, are abundant, relatively benign, and known for their strong electrochemical activity, making them attractive candidates if they can be coated reliably onto a solid support.
Building a Better Electrode with a Simple Coating Method
The researchers developed a straightforward way to coat ordinary graphite plates with a thin, nanoscale layer of manganese oxide using a sol–gel dip-coating process. They dissolved a manganese salt and a stabilizing liquid in ethanol to form a dark, paint-like solution. Cleaned graphite strips were dipped into this liquid, pulled out at controlled speeds, dried, and then heated to fix the coating. By carefully tuning the recipe — adjusting the manganese concentration, the number of layers, the drying and heating steps, and the dipping speed — they created a family of test electrodes. Electrical tests showed that one particular set of conditions produced a highly porous and uniform film made of very small manganese oxide particles, dramatically increasing the electrode’s ability to store and transfer charge.
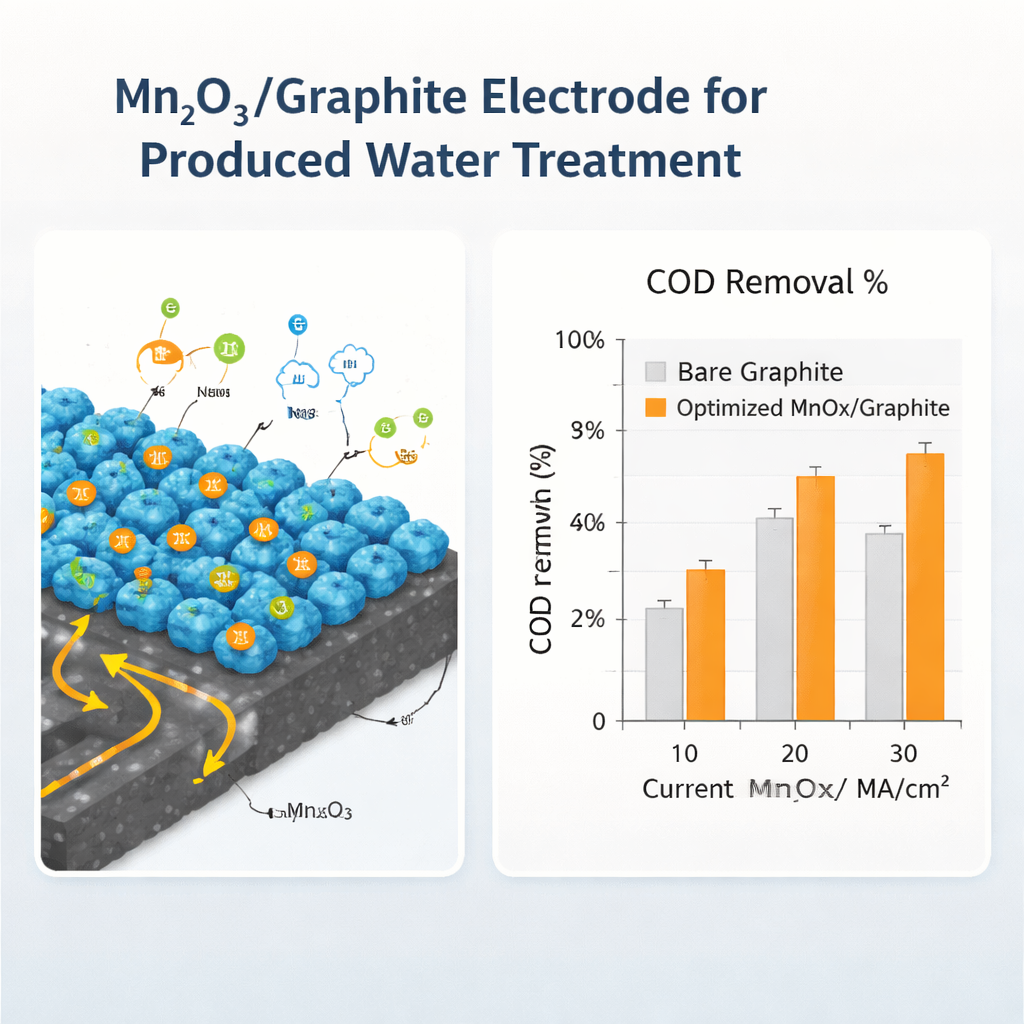
Turning Dirty Water Clear
The team then tested these coated electrodes on real produced water from an oil field in southern Iran. Using the best-performing manganese oxide on graphite electrode as the positive side and bare graphite as the negative side, they ran the water through an electrochemical cell at different current levels. Under practical settings, the optimized electrode removed about 87 percent of the organic load in just two hours, and at higher current it nearly eliminated it. Microscopy and X-ray measurements revealed that the coating consisted of well-crystallized manganese oxide nanoparticles tightly anchored to the graphite, helping it withstand extended operation. Accelerated tests suggested that, when run at typical industrial currents, the electrode could last on the order of hundreds of hours before needing replacement, much longer than plain graphite.
What This Means for Water and Energy
In everyday terms, this work shows that a relatively cheap, easy-to-make coated graphite plate can act like a powerful electric scrubber for some of the oil industry’s dirtiest water. By combining a simple sol–gel dip-coating step with careful heat treatment, the researchers created a surface that both conducts electricity well and provides countless tiny reaction sites where pollutants can be broken down. While not a complete treatment system on its own, this type of electrode could serve as an efficient final polishing step that helps operators meet strict discharge limits and reduce environmental risks, all while using less complex materials than many current high-end technologies.
Citation: Ghasemi, M., Afsham, N. & Fallah, N. Facile sol–gel fabrication of MnOx/Graphite nanostructured electrodes for sustainable produced water treatment. Sci Rep 16, 7344 (2026). https://doi.org/10.1038/s41598-026-38450-6
Keywords: produced water, electrochemical oxidation, manganese oxide electrode, wastewater treatment, graphite coating