Clear Sky Science · en
In silico discovery of natural compound-derived multi-target inhibitor for Huntington’s disease therapy
A New Angle on a Devastating Brain Disease
Huntington’s disease is a rare but devastating brain disorder that slowly robs people of movement, thinking, and independence. Today’s medicines can ease some symptoms, but they do not stop or reverse the disease. This study explores a new, computer-driven way to hunt for treatments, looking for a single natural compound that can hit several weak points in the disease process at once—an approach that could be more effective than drugs aimed at just one target.
Why Huntington’s Is So Hard to Treat
Huntington’s disease is caused by a faulty gene that leads to the gradual breakdown of nerve cells, usually starting in midlife. Early on, people may notice subtle mood changes, small involuntary movements, or mild trouble with planning and focus. Over 10 to 12 years, this can progress to severe movement problems, loss of speech, dementia, and full dependence on caregivers. Scientists know that many things go wrong inside brain cells in Huntington’s—energy production falters, harmful proteins clump together, and chemical signals between cells become toxic. Because so many pathways are disrupted at once, a “one protein, one drug” strategy has struggled to produce real breakthroughs.
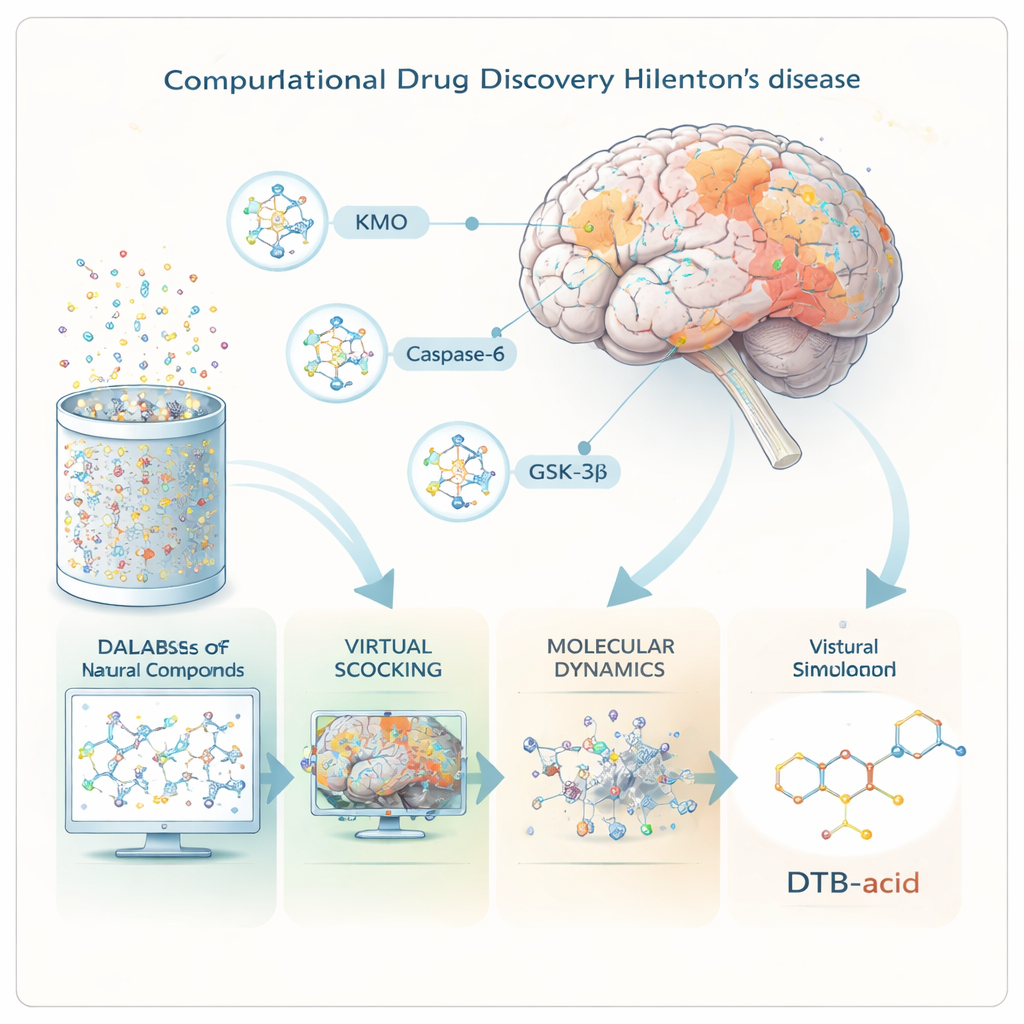
Three Critical Pressure Points in the Brain
The researchers focused on three proteins that sit at key control points in Huntington’s disease. The first, KMO, helps steer the breakdown of the amino acid tryptophan toward either toxic or protective by-products; when this balance tilts toward toxins, brain cells suffer. The second, Caspase-6, cuts the mutant huntingtin protein into smaller, highly toxic fragments that build up early in the disease. The third, GSK-3β, is a signaling enzyme linked to abnormal protein tangles and cell death in several brain disorders. By finding one molecule that can dial down the harmful activity of all three at once, the team hoped to design a therapy better matched to the disease’s complexity.
Screening Nature’s Library with Supercomputers
Instead of mixing chemicals in test tubes, the scientists worked entirely “in silico,” using advanced software to model how molecules behave. They began with more than 695,000 natural compounds drawn from a public database, preparing each one in three-dimensional form. Powerful virtual screening tools then predicted which compounds were likely to slip across the blood–brain barrier, behave like real drugs inside the body, and avoid major safety problems. Only about 60,000 passed these filters and were tested, on the computer, for how snugly they might fit into the three target proteins’ binding pockets.
One Standout Molecule: DTB-Acid
From this enormous search, one molecule—called DTB-acid, a natural alkaloid related to matrinic acid—rose to the top. Detailed docking studies showed that DTB-acid could form strong, well-placed contacts inside all three proteins. Next, the team ran long molecular dynamics simulations, which mimic how atoms move in water over time, to check whether the compound would stay put rather than drift away. Across 100 billionths of a second of simulated motion, the protein–drug complexes remained stable. Additional energy calculations suggested that binding to KMO was especially strong, while a separate analysis of water molecules in the binding sites indicated that interactions with GSK-3β were also thermodynamically favorable.
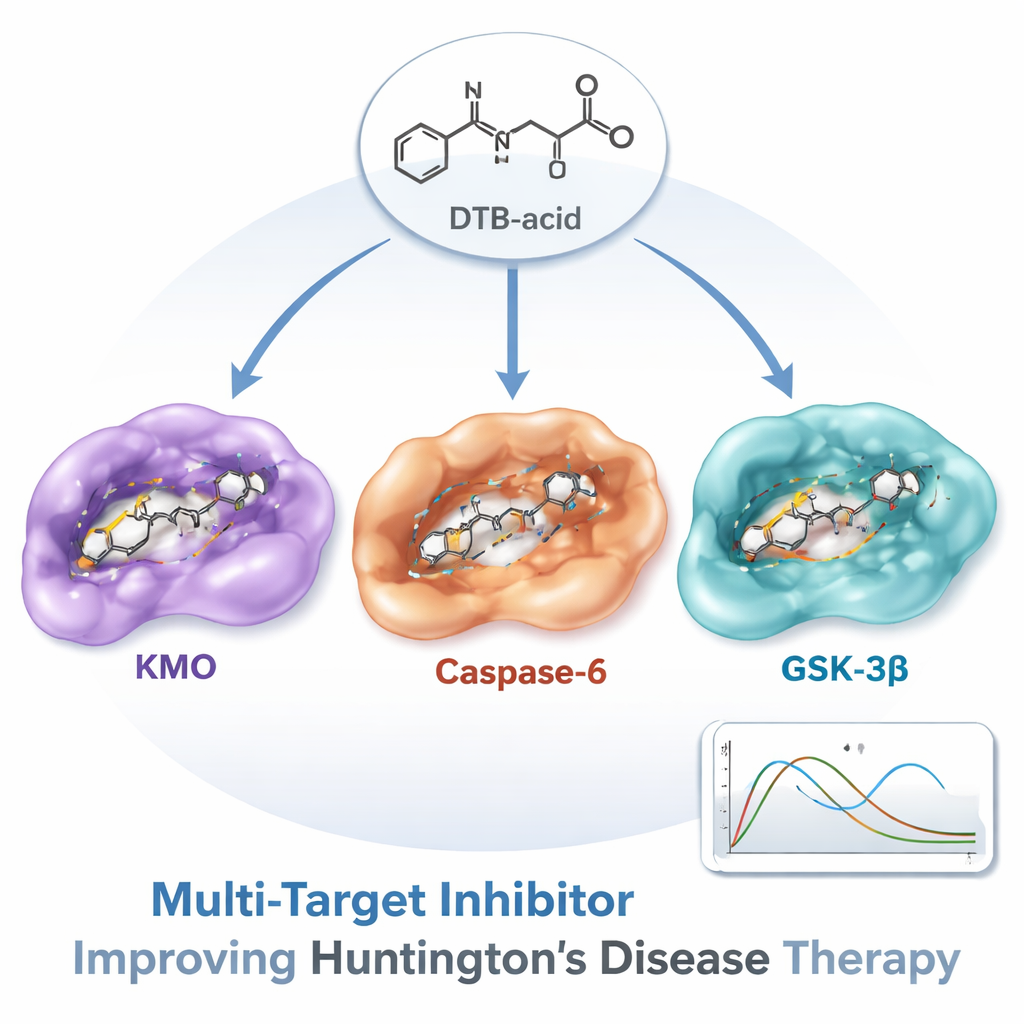
From Computer Hit to Future Medicine
Of course, a molecule that looks promising on a screen is only a first step. The authors stress that DTB-acid still needs to be tested in cells and animals to confirm that it reaches the brain, hits its intended targets, and actually protects neurons without harmful side effects. Even so, this work demonstrates a powerful, efficient path to discovering multi-target drugs for complex brain diseases. For non-specialists, the key message is that instead of treating Huntington’s disease with a series of separate, narrowly focused drugs, it may be possible to design one carefully tailored compound like DTB-acid that tackles several drivers of damage at once—offering new hope for slowing or altering the course of this relentless disorder.
Citation: Zheng, B., Banday, M., Gangwar, S. et al. In silico discovery of natural compound-derived multi-target inhibitor for Huntington’s disease therapy. Sci Rep 16, 7716 (2026). https://doi.org/10.1038/s41598-026-38430-w
Keywords: Huntington’s disease, multi-target drug, computational drug discovery, natural compounds, neurodegeneration