Clear Sky Science · en
CNOT2 /c-Myc/STAT3 signaling is critically involved in glycolysis mediated apoptosis of benzyl isothiocyanate in hepatocellular carcinoma
A Cancer-Fighting Ingredient on Your Plate
Benzyl isothiocyanate, or BITC for short, is a natural chemical found in familiar vegetables like broccoli, cabbage, and kale. While doctors have long known that people who eat more of these foods tend to have lower cancer rates, how specific plant ingredients slow or kill cancer cells is still being worked out. This study looks at how BITC affects liver cancer cells in the lab and uncovers a surprising link between the way cancer cells burn sugar and how they are pushed into self-destruction.
Why Liver Cancer Needs Better Options
Liver cancer is one of the deadliest cancers worldwide, and hepatocellular carcinoma is its most common form. Even with surgery, chemotherapy, and targeted drugs, many tumors come back or stop responding to treatment. One reason is that cancer cells rewire how they use energy: instead of relying mainly on oxygen in their powerhouses (mitochondria), they burn large amounts of sugar very quickly in a process often called the “Warburg effect.” This warped sugar use helps cancer cells grow faster and resist dying. Finding safe ways to interrupt this energy supply, especially using food-derived molecules, is an appealing strategy.
Testing a Vegetable Chemical on Liver Cancer Cells
The researchers treated two human liver cancer cell lines, SK-Hep1 and Huh7, with increasing doses of BITC. As the dose went up, fewer cells survived, showing that BITC is toxic to liver cancer cells under these conditions. When they looked more closely at what was happening inside the cells, they saw classic hallmarks of programmed cell death, or apoptosis. Key proteins that normally exist in an “inactive” form before being cut during apoptosis, such as PARP and caspase-3, were reduced in their intact, or “pro‑” forms. Flow cytometry tests also showed more cells in the sub‑G1 fraction of the cell cycle and more cells positive for Annexin V, both signs that the cancer cells were actively undergoing apoptosis rather than simply being damaged.
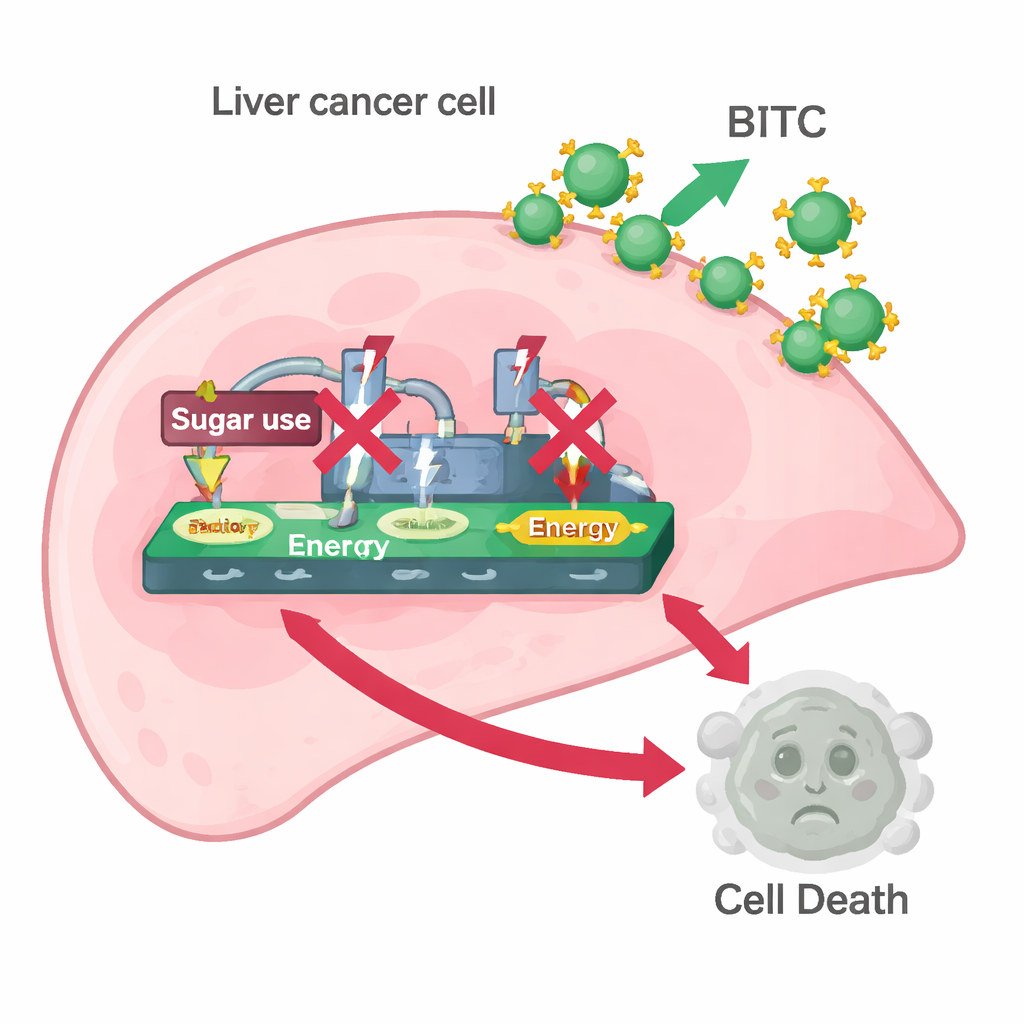
Shutting Down Cancer’s Sugar Craving
The team then explored whether BITC was affecting the Warburg effect. They measured proteins that help cancer cells gobble sugar and turn it into energy and lactate—specifically HK2, PKM2, and LDH. BITC treatment lowered the levels of all three in both liver cancer cell lines and reduced how much lactate the cells released and how much glucose they consumed from the culture medium. In other words, BITC made liver cancer cells less able to run their high-speed sugar-burning machinery. When the scientists added pyruvate, a downstream product of sugar breakdown that can still feed cell metabolism, many of BITC’s effects were reversed: the cells regained higher levels of PARP, caspase-3, and the glycolysis enzymes. This rescue suggested that interfering with sugar processing is a key part of how BITC triggers cell death.
Unraveling the Control Switches Inside the Cell
To understand deeper control layers, the researchers focused on a signaling trio: CNOT2, c-Myc, and STAT3. These proteins help drive growth, survival, and metabolic rewiring in many tumors. Data from liver cancer patients showed that CNOT2 tends to be higher in people with poorer outcomes. In the lab, BITC treatment lowered the active, phosphorylated form of STAT3, its upstream activator JAK1, the growth-promoting protein c-Myc, and CNOT2 itself. Using genetic tools to reduce CNOT2 or STAT3 levels made BITC’s killing effect even stronger, further lowering PARP and caspase-3. Protein-binding experiments revealed that BITC also disrupted the physical partnerships between CNOT2 and STAT3 and between CNOT2 and c-Myc. When CNOT2 or c-Myc were artificially boosted, BITC lost much of its ability to shut down glycolysis enzymes and trigger apoptosis, underscoring that this signaling axis acts like a master switch linking sugar metabolism and survival.
What This Could Mean for Future Therapies
Taken together, the findings paint a coherent picture: BITC attacks liver cancer cells by cutting off their preferred sugar-based fuel line and flipping cell-death switches that depend on the CNOT2/c-Myc/STAT3 axis. By blocking both growth signals and sugar use, BITC pushes cancer cells past a metabolic tipping point into apoptosis. These results come from cell culture experiments, not patients, and only two liver cancer cell types were studied, so much more work in animals and humans is needed. Still, the research strengthens the idea that certain compounds from everyday vegetables could be refined or combined with other treatments to more precisely starve liver tumors while encouraging them to self-destruct.
Citation: Koh, W., Park, SY., Kim, B. et al. CNOT2 /c-Myc/STAT3 signaling is critically involved in glycolysis mediated apoptosis of benzyl isothiocyanate in hepatocellular carcinoma. Sci Rep 16, 7000 (2026). https://doi.org/10.1038/s41598-026-38416-8
Keywords: liver cancer, benzyl isothiocyanate, cancer metabolism, apoptosis, cruciferous vegetables