Clear Sky Science · en
Application analysis of transfected cell method for detecting AChR antibodies in MG patients
Why this matters for people with weak muscles
Myasthenia gravis is a disease in which the body’s own defenses attack the places where nerves talk to muscles, causing drooping eyelids, trouble chewing, and general fatigue. Doctors rely on blood tests that look for these misguided antibodies, but today’s tests can miss a sizeable fraction of patients. This study explores a newer, cell-based way to find those antibodies more reliably, which could mean earlier diagnosis and better guidance for treatment.
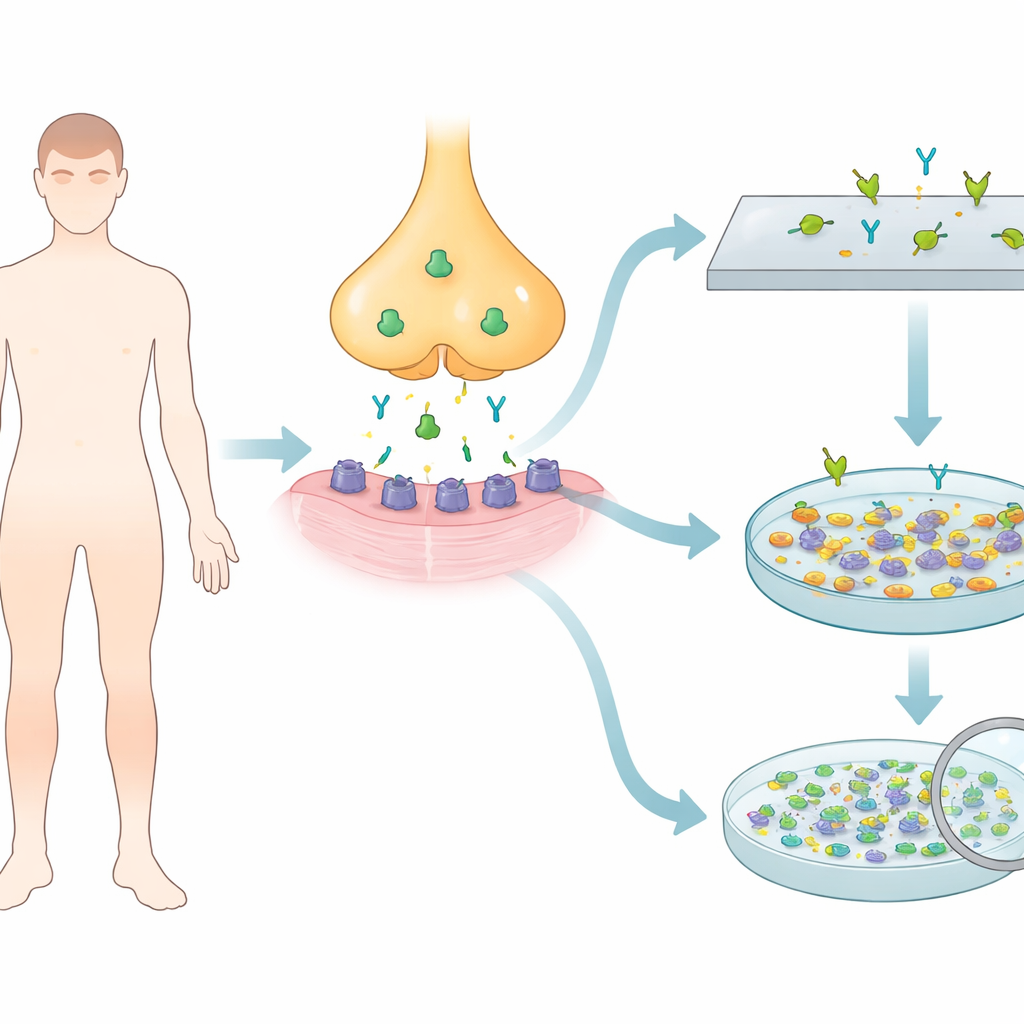
A hidden attack at the nerve–muscle meeting point
In myasthenia gravis, the immune system targets a key protein called the acetylcholine receptor, which acts like a gate that lets nerve signals trigger muscle movement. Most patients with generalized disease, and many with eye-only symptoms, carry antibodies against this receptor in their blood. Yet up to half of people whose symptoms are limited to the eyes, and around 10–15 percent of those with more widespread weakness, test negative with standard blood assays. One likely reason is that some antibodies cling only weakly to the receptor and slip past conventional tests that use purified or simplified forms of the protein.
Turning ordinary cells into sensitive test platforms
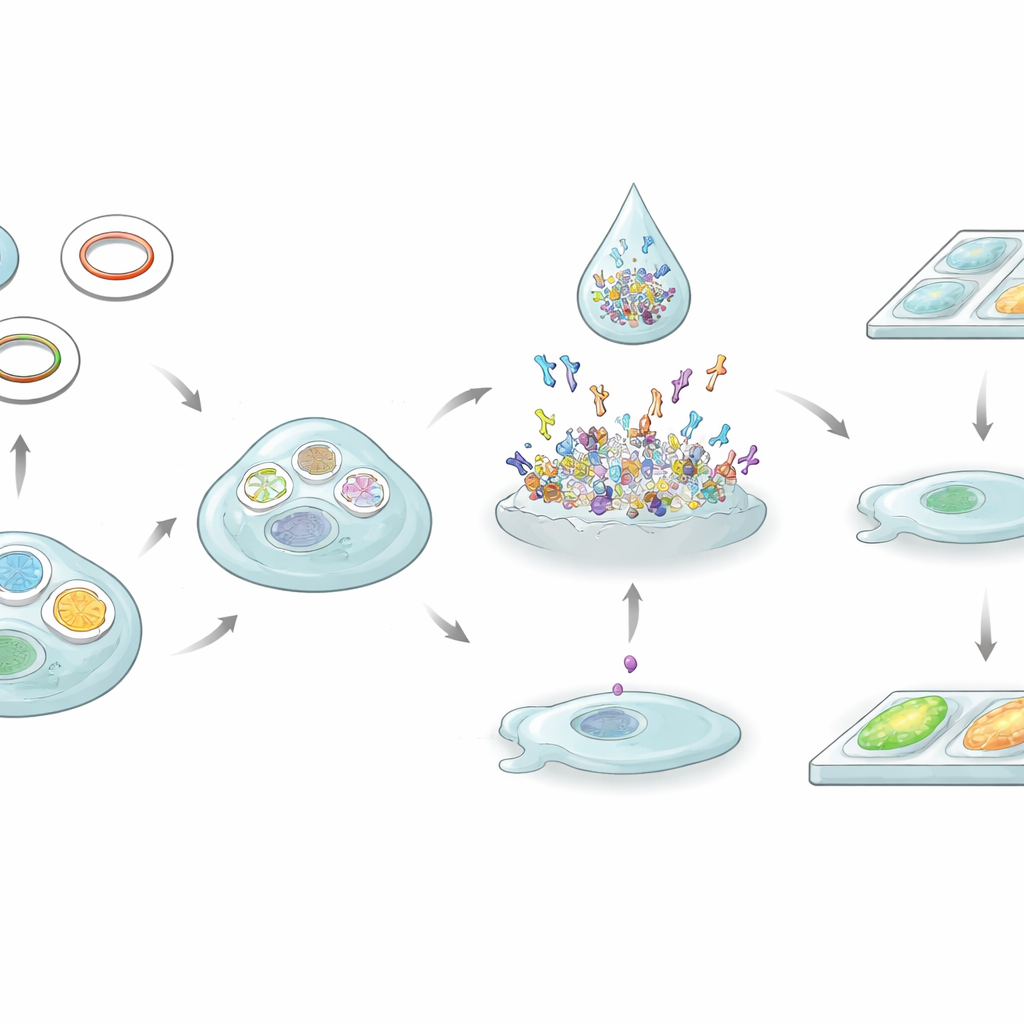
The researchers set out to build an in-house cell-based assay using a human cell line commonly used in laboratories. They inserted genetic instructions for different combinations of receptor building blocks—adult-type, fetal-type, and a mixture of both—plus a helper protein that clusters receptors on the cell surface. These engineered cells were grown on tiny glass slips and fixed so they could be stored as ready-made test slides. When diluted patient serum was added, any antibodies against the receptor would bind to these clustered proteins and could be revealed using a fluorescent secondary antibody viewed under a microscope.
Finding the best combination to catch more antibodies
The team first discovered that cells expressing only a single receptor subunit failed to light up, even when tested with 48 samples that were positive by a commercial kit. This suggested that antibodies from patients recognize the receptor’s full three‑dimensional shape on the cell surface, not lonely pieces still trapped inside the cell. In contrast, cells engineered with full adult or fetal receptor combinations showed clear fluorescent signals when exposed to positive sera, while remaining dark for healthy control samples. The strongest and most frequent signals appeared when cells expressed a full mix of both adult and fetal receptor forms together, especially in samples with lower antibody levels. In a set of 24 carefully chosen pediatric and adult patients, this “full combination” slide produced medium-to-strong signals in about two‑thirds of cases, far outperforming slides that displayed only adult or only fetal receptors.
Head-to-head with a standard blood test
To see how their cell-based approach compared with a widely used enzyme-linked immunosorbent assay (ELISA), the researchers tested 85 patients with myasthenia gravis using both methods. The ELISA detected receptor antibodies in about 68 percent of patients, whereas the cell-based assay detected them in more than 75 percent. Overall, the two tests agreed well—the statistical measure of agreement, called kappa, was high—yet there were eight patients where the results disagreed. When a radioimmunoassay, considered a reference method, was brought in to resolve these conflicts, it matched the cell-based assay in six of the eight cases. This pattern suggests that the cell-based test can pick up some low-affinity antibodies that ELISA misses, without sacrificing specificity.
What this means for patients and doctors
By making ordinary lab cells display densely packed, mixed-age versions of the acetylcholine receptor, this study created a more sensitive spotlight for disease-causing antibodies in myasthenia gravis. The new assay performs at least as well as existing commercial kits overall and may be better at catching hard-to-detect antibodies, particularly in patients with low levels or more complex antibody profiles. While larger, multi-center studies and live-cell refinements are still needed before widespread use, this work outlines a practical path toward more accurate blood testing, helping doctors confirm diagnoses earlier, monitor disease more precisely, and tailor treatment for people living with this muscle-weakening disorder.
Citation: Liu, W., Xia, Z., Hu, J. et al. Application analysis of transfected cell method for detecting AChR antibodies in MG patients. Sci Rep 16, 8130 (2026). https://doi.org/10.1038/s41598-026-38414-w
Keywords: myasthenia gravis, acetylcholine receptor antibodies, cell-based assay, autoimmune neuromuscular disease, diagnostic sensitivity