Clear Sky Science · en
Rapid, efficient, and thermal degradation of chlorophenols using polymer-coated or metal-doped magnetic nanoparticles, with and without the application of AMF
Cleaning Up Stubborn Toxins in Water
Industrial chemicals called chlorophenols are powerful ingredients in dyes, pesticides, and other products, but once they leak into rivers or groundwater they are very hard to remove and can harm both ecosystems and human health. This study explores a new way to scrub these stubborn molecules from water using tiny magnetic particles that act like reusable "heat-and-clean" capsules. By tuning the surface and composition of these particles, and even heating them remotely with a magnetic field, the researchers show how toxic pollutants can be broken down in seconds into safer substances, pointing to faster and more practical treatment methods for factory wastewater.
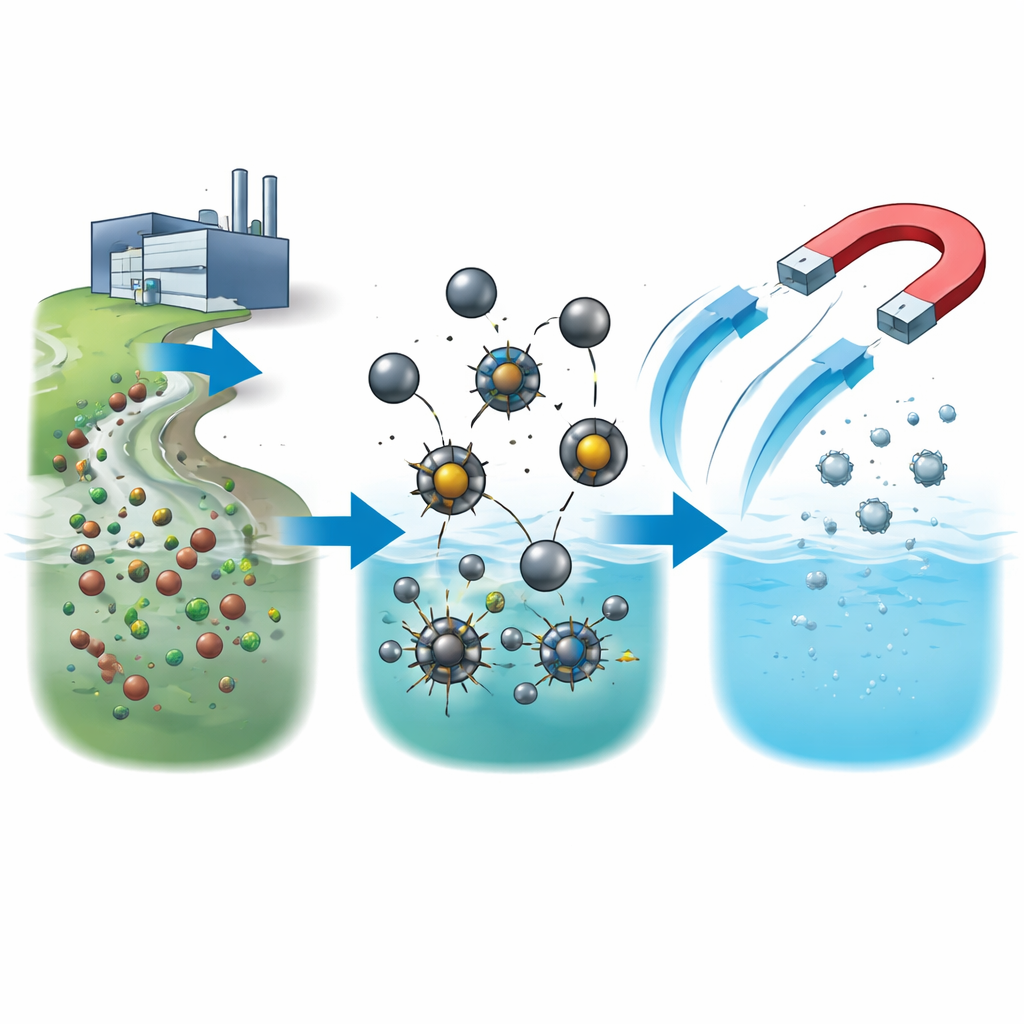
Tiny Magnets Built for Dirty Water
The team designed several families of iron oxide nanoparticles—grains so small they are only about 8 to 15 billionths of a meter across. Some particles were wrapped in thin shells of common polymers such as PVP, starch, or chitosan, which help them stay dispersed and stable in water. Others were “doped” by swapping some of the iron atoms for cobalt, nickel, or zinc, which changes how the particles behave magnetically and chemically. Careful imaging and structural tests confirmed that all of these particles had highly ordered crystal structures and strong, switchable magnetism. This means they can be quickly collected with a simple magnet after they have done their job, instead of being left behind as a new kind of waste.
Turning Weak Bleach into a Powerful Cleaner
On their own, chlorophenols resist normal water treatment and even simple hydrogen peroxide is too weak to destroy them quickly. The nanoparticles change that. When mixed with polluted water and a small amount of hydrogen peroxide, their iron and doped metal atoms help generate extremely reactive short-lived species that attack the chlorophenol rings. The researchers found that uncoated iron oxide particles cleaned both 2-chlorophenol and 4-chlorophenol well within minutes under mild conditions. Polymer-coated versions also worked, but their protective shells partly blocked access to the reactive surface, slowing the breakdown even as they improved stability and handling.
Boosting Power with Smart Metal Choices
The most dramatic improvements came from particles that contained additional metals. By comparing zinc-, nickel-, and cobalt-doped iron oxide, the team showed that the identity and position of these metal atoms inside the crystal lattice strongly control how fast pollutants are destroyed. Zinc-doped particles broke down chlorophenols faster than bare iron oxide, but cobalt-doped particles were the clear winners: they completely degraded typical test solutions at neutral pH within seconds, giving some of the highest reaction rates reported for this kind of chemistry. At the same time, the particles stayed magnetically responsive and structurally intact over at least six cleaning cycles, and simple magnetic separation plus water rinsing was enough to prepare them for reuse.
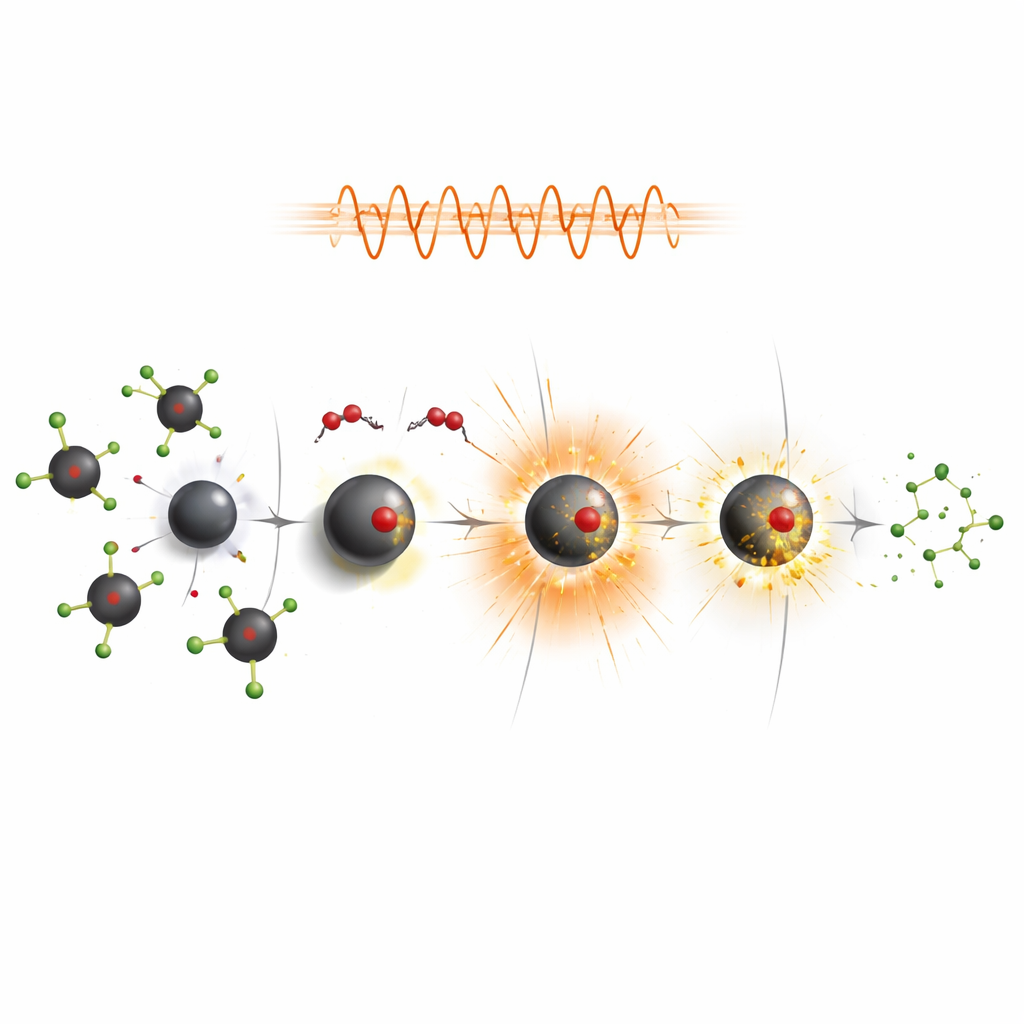
Cleaning Faster by Heating from the Inside
Because these particles are magnetic, they heat up when exposed to an alternating magnetic field, much like metal in an induction stove. The researchers took advantage of this effect by running degradation tests while switching on such a field. Under these conditions, the particles warmed the surrounding liquid, which in turn sped up the production and action of the reactive species that attack chlorophenols. For several formulations—especially polymer-coated particles that were slower at room temperature—this “magnetothermal” boost nearly doubled or more the amount of pollutant removed in a fixed time. Cobalt-doped particles again stood out, achieving complete removal of both target chlorophenols under field-driven heating while maintaining strong reusability.
From Toxic Streams to Safer Water
Overall, the study shows that carefully engineered magnetic nanoparticles can transform a relatively gentle oxidizing agent into a fast, efficient water cleaner and that their performance can be dialed in by adjusting surface coatings, metal dopants, and temperature. In clear tests, chlorophenols were not just removed from solution but broken apart, with chlorine released as harmless chloride ions and the carbon content falling to nearly zero, indicating full mineralization to carbon dioxide. Because the particles can be switched on by a magnetic field, recovered with a handheld magnet, and reused many times without complex regeneration steps, this approach offers a promising route toward compact, high-speed treatment units for industrial wastewater that currently carries some of the most persistent and dangerous organic contaminants.
Citation: Mohammed, H.A., Madkhali, N., Lemine, O.M. et al. Rapid, efficient, and thermal degradation of chlorophenols using polymer-coated or metal-doped magnetic nanoparticles, with and without the application of AMF. Sci Rep 16, 7922 (2026). https://doi.org/10.1038/s41598-026-38408-8
Keywords: chlorophenol pollution, magnetic nanoparticles, advanced oxidation, wastewater treatment, cobalt ferrite