Clear Sky Science · en
meso-2,3-dibromosuccinic acid as a temperature-dependent catalyst for the selective synthesis of syn and anti-highly functionalized tetrahydropyridines: experimental and theoretical study
Why this matters for future medicines
Chemists often need to build complex, three-dimensional molecules that behave like tiny keys fitting into biological locks. A subtle change in 3D shape can turn a helpful drug into an inactive or even harmful one. This paper describes a simple, metal-free way to steer such shapes using nothing more exotic than temperature, offering a cleaner and more precise route to potential medicines and other useful chemicals. 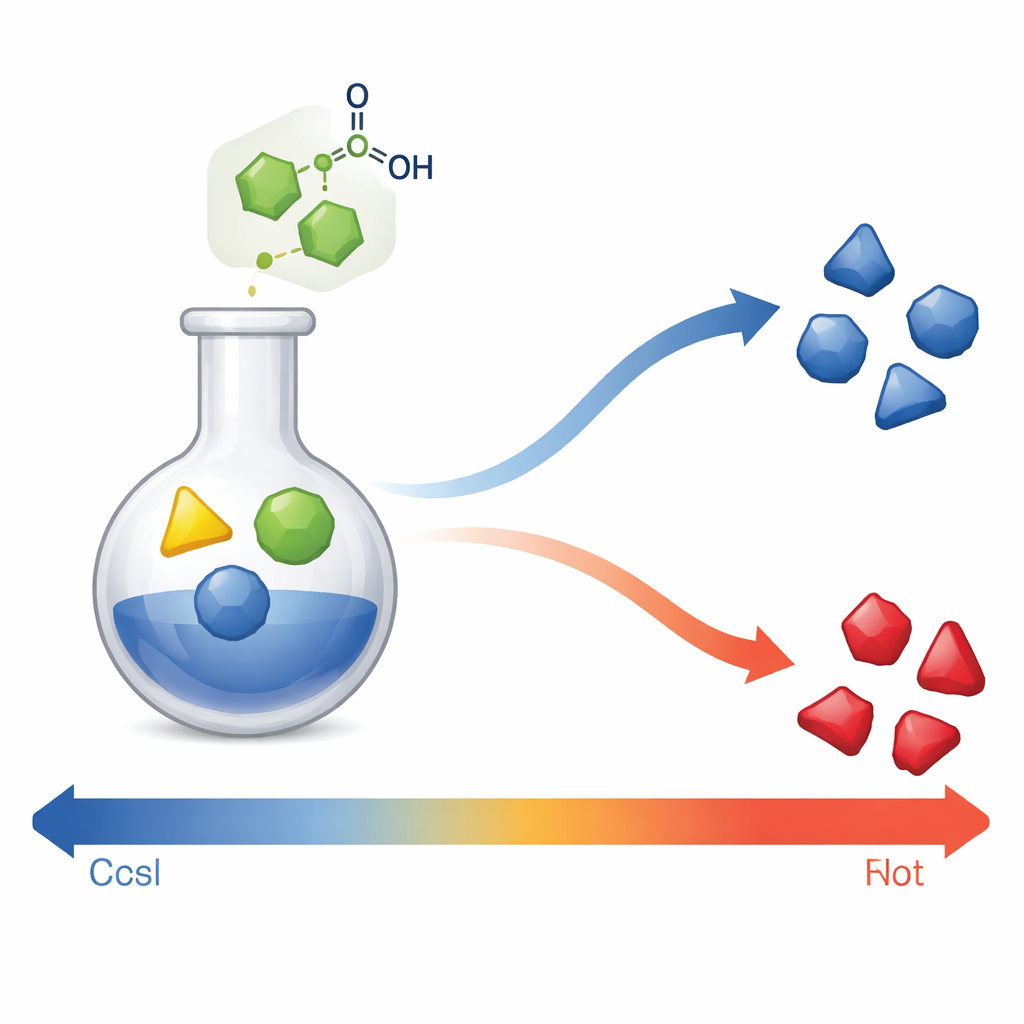
Shaping tiny rings that power modern drugs
Many modern drugs, natural products, and agrochemicals contain six‑membered rings that include nitrogen atoms. Two closely related families of these rings, called tetrahydropyridines and piperidines, appear in treatments for high blood pressure, bacterial and malarial infections, brain disorders, and cancer. These rings are three‑dimensional, so their atoms can be arranged in different relative orientations, known as syn and anti forms. Even though these forms have the same atoms and bonds, they can behave very differently in the body. Being able to choose which form to make, quickly and cleanly, is therefore a central goal in drug‑oriented chemistry.
A one-pot recipe with a simple acid helper
The authors developed a one‑pot process in which three common building blocks—an aromatic aldehyde, an aromatic amine, and a 1,3‑dicarbonyl compound—combine in a single flask to form richly decorated tetrahydropyridines. The key ingredient is a small organic acid, meso‑2,3‑dibromosuccinic acid, which acts as a catalyst. It speeds up the reaction without being consumed and, importantly, contains no metals. Compared with earlier methods that often rely on metal salts or harsh conditions, this approach uses inexpensive materials, works in ordinary ethanol, and gives high yields of products with many different substituents, making it attractive for medicinal chemistry and green chemistry alike.
Turning the temperature knob to pick syn or anti
A striking feature of this catalyst is its sensitivity to temperature. At low temperature (around 5 °C), the reaction produces only the syn form of the tetrahydropyridine, giving chemists complete control over that shape. At moderate temperature (about 25 °C), the mixture contains both syn and anti forms in a roughly 60:40 ratio. At higher temperature (around 65 °C), the outcome flips: only the anti form appears. Other tested acids and metal salts could not match this behavior; most gave only the anti form regardless of temperature. The team also explored many combinations of aldehydes and amines. They found that bulky groups near the reacting sites favor the syn product, while other patterns favor the anti form, revealing how subtle changes in structure guide the reaction. 
Peeking under the hood with theory
To understand why temperature has such a strong effect, the researchers used advanced quantum‑chemical calculations. They modeled the key step of the reaction, an aza‑Diels–Alder cyclization where two reactive partners join to form the nitrogen‑containing ring. The calculations show that the anti product is more stable overall, like a deeper valley in an energy landscape. However, the pathway leading to the syn product has a lower energy barrier, meaning it is easier to reach at first. At low temperature, the system follows this easier route and gets trapped in the syn valley. As the temperature rises, the molecules gain enough energy to climb out and rearrange into the more stable anti form. The team backed up this picture by analyzing orbital energy gaps, aromaticity measures, and by comparing predicted nuclear magnetic resonance spectra with the experimental data; the excellent agreement supports their mechanistic explanation.
What this means in simple terms
In practical terms, the study shows that a small, inexpensive organic acid can act like a “thermostat” for molecular shape. By cooling or heating the reaction, chemists can choose whether they obtain mainly the syn or the anti version of a valuable ring system, without resorting to heavy metals or complex setups. This level of control is crucial when designing new drugs, because biological targets can distinguish sharply between such near‑twins. The combination of straightforward experiments and detailed theoretical analysis in this work provides both a useful synthetic tool and a clear blueprint for how temperature and catalyst choice can be harnessed to sculpt molecules in three dimensions.
Citation: Aboonajmi, J., Mandegani, Z., Rabor, J.T. et al. meso-2,3-dibromosuccinic acid as a temperature-dependent catalyst for the selective synthesis of syn and anti-highly functionalized tetrahydropyridines: experimental and theoretical study. Sci Rep 16, 8117 (2026). https://doi.org/10.1038/s41598-026-38400-2
Keywords: tetrahydropyridines, temperature-controlled catalysis, metal-free synthesis, multicomponent reactions, stereoselective chemistry