Clear Sky Science · en
Algorithmic analysis of the structure of mixed odontogenic tumors
Why tiny jaw tumors matter
Most of us only think about our teeth when they ache or need a filling, but the tissues that form teeth can sometimes give rise to rare tumors. These growths, called odontogenic tumors, are uncommon but important because they can damage the jaws and be difficult to diagnose under the microscope. This study asks whether modern computer algorithms can measure the architecture of these tumors so precisely that they help pathologists tell look‑alike lesions apart and better understand how they behave.
Rare growths that mimic developing teeth
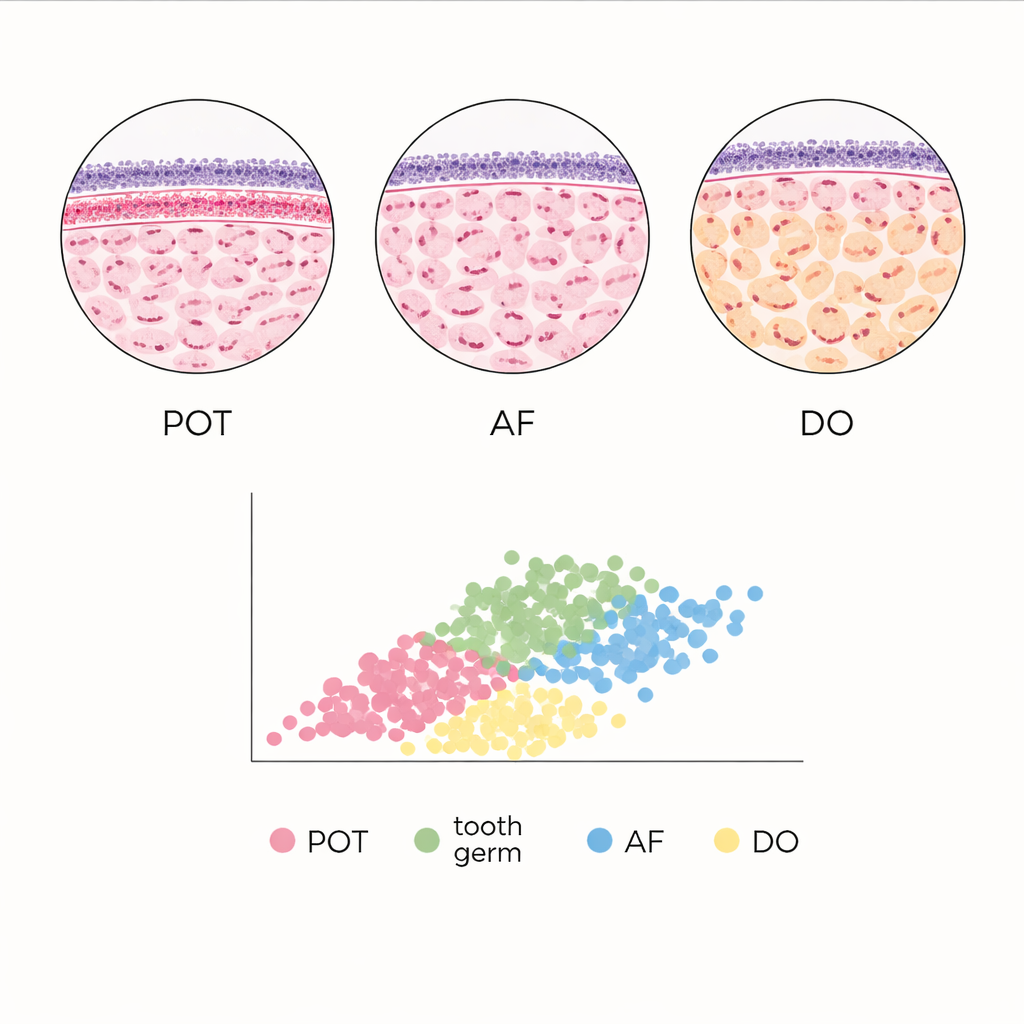
Odontogenic tumors arise only in tooth‑forming regions of the jaws. The team focused on a small group called mixed odontogenic tumors, which contain both the cell layer that normally gives rise to enamel (epithelium) and the supporting tooth‑forming tissue (ectomesenchyme). Within this group, three lesions—primordial odontogenic tumor (POT), ameloblastic fibroma (AF), and developing odontoma (DO)—can look remarkably similar to each other and to normal tooth germs, the early structures from which teeth develop. POT is especially new to science, having been formally recognized by the World Health Organization only in 2017, and remains so rare that every additional case is relevant to refining its definition.
Turning microscope slides into measurable maps
To move beyond the limits of eyeballing glass slides, the researchers digitized biopsy samples from 15 mixed odontogenic tumors (five each of POT, AF, and DO) and five normal tooth germs. After staining the tissue in the usual way, they scanned the slides at high magnification and used image‑analysis software to locate each cell nucleus. From there, mathematical tools known as watershed transforms defined a sort of “zone of influence” around every nucleus, creating thousands of “virtual cells” whose size and shape could be measured automatically. By stacking and aligning images, the team also examined how tightly packed the nuclei were at different depths below the surface layers of the tissue.
What the algorithms saw inside the tumors
In POT, the computer‑derived density maps confirmed a striking crowding of cells just beneath the surface layer of epithelium, with nuclei packed most tightly in the upper third of the tissue and thinning out toward the center. This pattern—called subepithelial condensation—was present in most POT samples but absent in a minority that lacked the dense band. The nuclei in the supporting tissue were slightly larger than those in the epithelium, but both compartments had similarly rounded shapes. When the team compared the virtual cells across all entities, they found that epithelial virtual cells were always the smallest, reflecting their higher packing density, while deeper regions contained much larger virtual cells. Developing odontoma showed the broadest spread of epithelial virtual cell areas, with generally larger values than in other lesions, whereas ameloblastic fibroma tended to have the smallest.
Normal tooth development as a benchmark
To place these tumors in context, the authors compared them with normal tooth germs captured at early “cap” and “bell” stages of development. In some regions, the architecture of POT without a condensed subepithelial band closely resembled that of tooth germs: the average areas of epithelial virtual cells were not statistically different. By contrast, developing odontoma and ameloblastic fibroma showed distinct quantitative signatures, including fewer layers of virtual cells and different epithelial cell‑area distributions, despite sometimes imitating tooth development under the microscope. These measurable differences suggest that, even when tissues look similar by eye, their underlying spatial organization can reveal subtle diagnostic clues.
What this means for patients and pathologists
The study shows that algorithm‑driven “virtual cell” analysis can capture architectural patterns that support the idea of primordial odontogenic tumor as a distinct type of jaw lesion, one that in some areas behaves like an early, actively interacting tooth germ. At the same time, it quantifies how other mixed odontogenic tumors differ in the size and layering of their epithelial compartments. For patients, this does not yet change treatment, but it points toward a future in which computer‑assisted measurements supplement traditional pathology, especially in small or ambiguous biopsies. The authors stress that their dataset is still small, and larger multi‑center studies are needed before such methods can routinely guide diagnosis, but the work illustrates how digital tools can turn a pathologist’s qualitative impressions into reproducible numbers.
Citation: Pereira-Prado, V., Sicco, E., Silveira, F.M. et al. Algorithmic analysis of the structure of mixed odontogenic tumors. Sci Rep 16, 7538 (2026). https://doi.org/10.1038/s41598-026-38399-6
Keywords: odontogenic tumors, primordial odontogenic tumor, digital pathology, virtual cell analysis, tooth development