Clear Sky Science · en
Synthesis of highly conducting molybdenum disulfide electrode for asymmetric supercapacitor applications
Why Better Energy Storage Matters
From electric cars to backup power for our homes, modern life depends on devices that can store and release energy quickly and reliably. Today’s batteries pack a lot of energy but tend to charge slowly and wear out over time, while conventional supercapacitors charge fast but hold relatively little energy. This study explores a new way to build the heart of a supercapacitor—the electrode—using a special material called molybdenum disulfide arranged in ultra-thin sheets. The aim is to combine fast charging with high energy storage and long lifetime in a cost-effective, environmentally friendly device.

Building a Better Electrode
The researchers focused on a compound called molybdenum disulfide (MoS2), which has a layered structure a bit like a stack of paper sheets. These layers can host electric charge both on their surfaces and deeper inside, making them attractive for advanced energy storage. Instead of mixing MoS2 powder with sticky binders and pressing it onto metal, the team grew it directly on a light, sponge-like metal support called nickel foam. They used a process known as chemical vapor deposition, in which vaporized molybdenum and sulfur atoms react and settle onto the foam, forming a tightly attached coating of interconnected MoS2 nanosheets without any glue-like additives. This “binder-free” approach preserves more open space for liquid electrolyte to reach the active material and lowers electrical resistance.
Seeing the Tiny Architecture
To understand what they had made, the scientists examined the electrode with several powerful tools. X-ray measurements showed that the MoS2 formed a well-ordered crystalline structure, while Raman spectroscopy confirmed that the chemical bonds matched those expected for high-quality MoS2. Electron microscope images revealed dense networks of thin, overlapping sheets with rough, porous regions and open channels throughout the nickel foam. Gas adsorption tests indicated a large surface area and pores spanning a range of sizes, all of which help ions from the electrolyte move in and out quickly. This tiny architecture is crucial: more accessible surface and pathways means more charge can be stored and released in a short time.
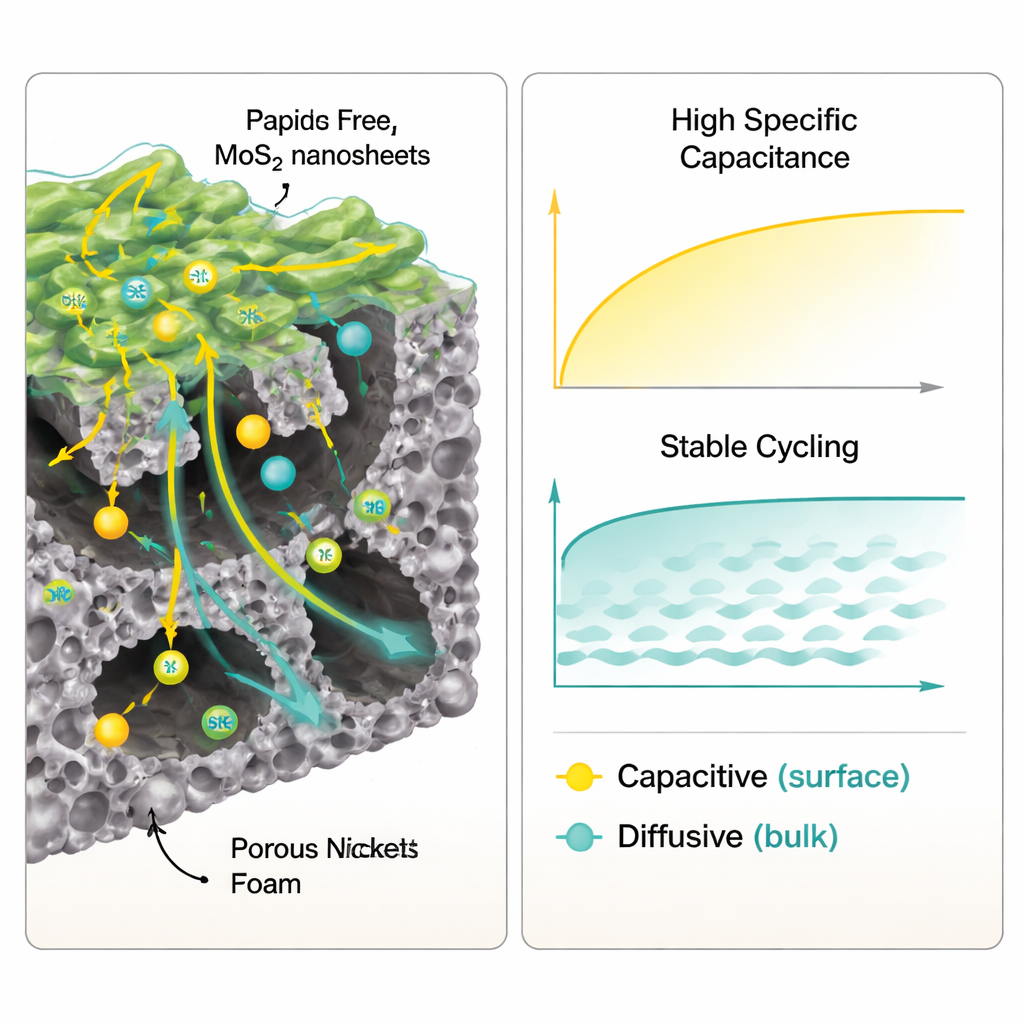
How Well It Stores and Delivers Energy
The real test is how the electrode behaves in an actual supercapacitor environment. In a water-based alkaline solution, the MoS2-on-foam electrode showed extremely high specific capacitance, a measure of how much electric charge can be held per unit mass. It significantly outperformed many similar materials reported in earlier studies. Even when the device was charged and discharged at higher rates, the electrode retained much of its storage capacity, indicating that ions could still reach active sites rapidly. Electrical impedance measurements showed low resistance for charge transfer and ion movement, helping to explain the strong performance. After 10,000 rapid charge–discharge cycles, the electrode still kept about four-fifths of its original capacity and nearly full charge–discharge efficiency, signaling good durability.
Turning the Material into a Practical Device
To move beyond a single electrode, the team built an asymmetric supercapacitor device. They used their MoS2-coated nickel foam as the positive side and a conventional activated carbon electrode as the negative side, separated by a thin membrane in an alkaline solution. This pairing allowed the device to operate over a wider voltage window than a typical symmetric supercapacitor, which boosts the energy it can store. Tests showed that the assembled device delivered both high capacitance and an impressive combination of energy density (how much energy per kilogram) and power density (how quickly that energy can be delivered). The performance surpassed many similar MoS2-based supercapacitors reported in the literature, suggesting that this design could be competitive for real-world applications.
What This Means for Future Devices
For non-specialists, the key message is that the researchers have found a clever way to grow a thin, highly conductive, and strongly attached layer of MoS2 nanosheets on a metal foam, without the usual inactive binders that block useful surface area. This architecture lets ions and electrons move easily, so the electrode can store a lot of charge and release it quickly over many cycles. When built into a full device, it offers a promising balance between battery-like energy and capacitor-like power. While more work is needed before commercialization, this study points toward supercapacitors that could one day help electric vehicles, portable electronics, and grid systems charge faster, last longer, and operate more efficiently.
Citation: Khan, A.R., Badshah, F., Awais, M. et al. Synthesis of highly conducting molybdenum disulfide electrode for asymmetric supercapacitor applications. Sci Rep 16, 7547 (2026). https://doi.org/10.1038/s41598-026-38362-5
Keywords: supercapacitors, molybdenum disulfide, energy storage, nanomaterials, nickel foam electrodes