Clear Sky Science · en
Inhibition of galanin receptor 3 slows down retina degeneration in retinitis pigmentosa through modulation of inflammatory and oxidative stress response
Why protecting fading night vision matters
Retinitis pigmentosa is a group of inherited diseases that slowly steal people’s night and side vision, often leading to serious sight loss in midlife. There is no broadly effective treatment, in part because many different gene mutations can cause the disease. This study explores a different angle: instead of fixing the broken gene, it asks whether turning down harmful inflammation and “rust-like” damage in the retina can slow the loss of light-sensing cells and help preserve vision.
A stress switch hidden in the retina
The light-sensing cells at the back of the eye, called photoreceptors, must constantly balance energy use, oxygen, nutrients, and waste removal. In one common form of retinitis pigmentosa, a mutation in the rhodopsin protein (known as P23H) makes this visual pigment misfold, clogging the cell’s internal machinery and triggering chronic stress. That stress in turn sets off inflammation and oxidative damage, both of which can kill photoreceptors. The authors focused on a signaling protein called galanin receptor 3 (GALR3), present in several retinal cell types. They found that in mice carrying the P23H mutation, GALR3 and its natural partner molecules were switched on more strongly than in healthy eyes, suggesting that GALR3 might act as a stress amplifier in degenerating retinas.
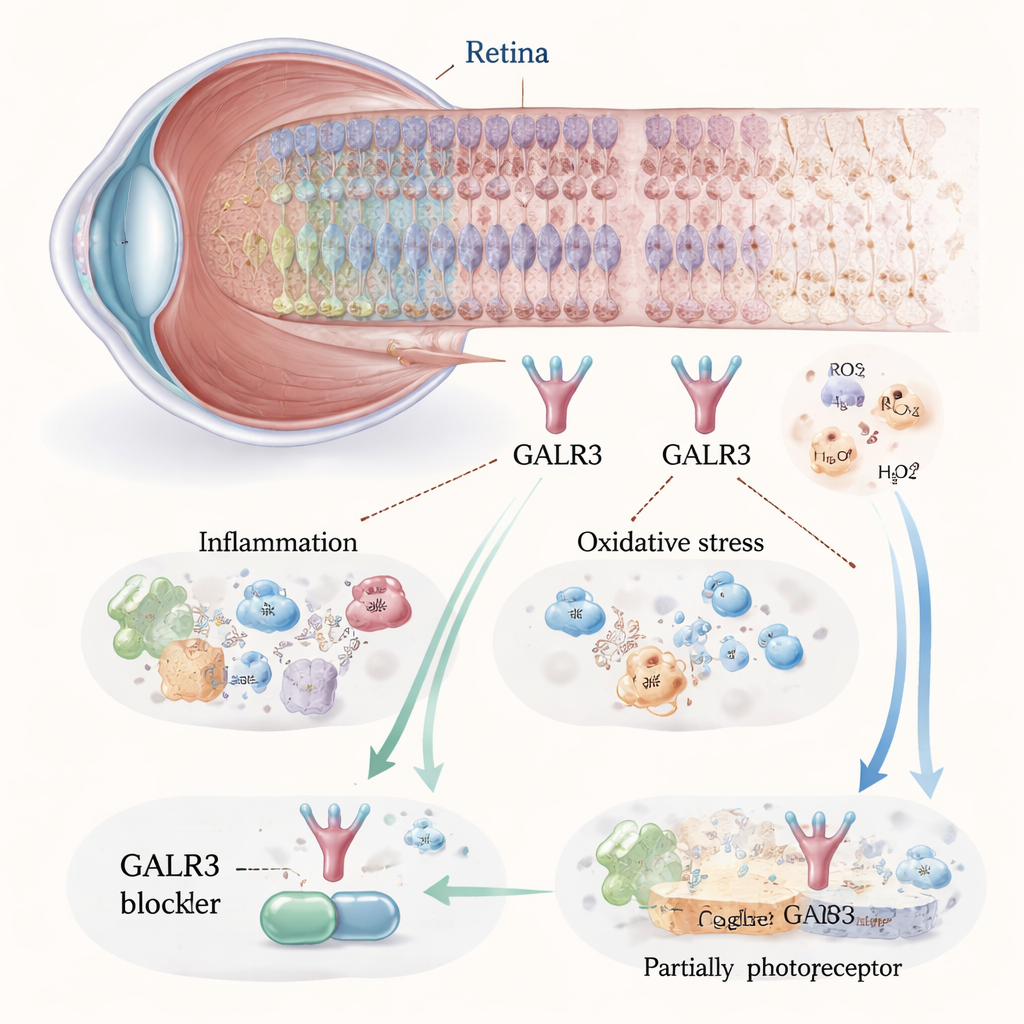
Slowing cell loss by blocking a harmful signal
To test whether GALR3 drives damage or protection, the researchers used two strategies in P23H mice: they bred animals completely lacking the Galr3 gene, and they treated others with a drug that specifically blocks the receptor, called SNAP-37,889. Over several weeks, they examined retinal structure with high-resolution imaging and measured electrical responses to light. In untreated P23H mice, the layer of nuclei that houses photoreceptors thinned by about half compared with normal mice by one month of age and continued to shrink. When GALR3 was blocked, this thinning was significantly reduced, and key visual proteins such as rhodopsin and cone opsin remained at higher levels. Electrical recordings from the retina also showed stronger light responses in treated or GALR3-deficient mice, meaning that the surviving cells were working better, not just present.
Turning down inflammation and boosting defenses
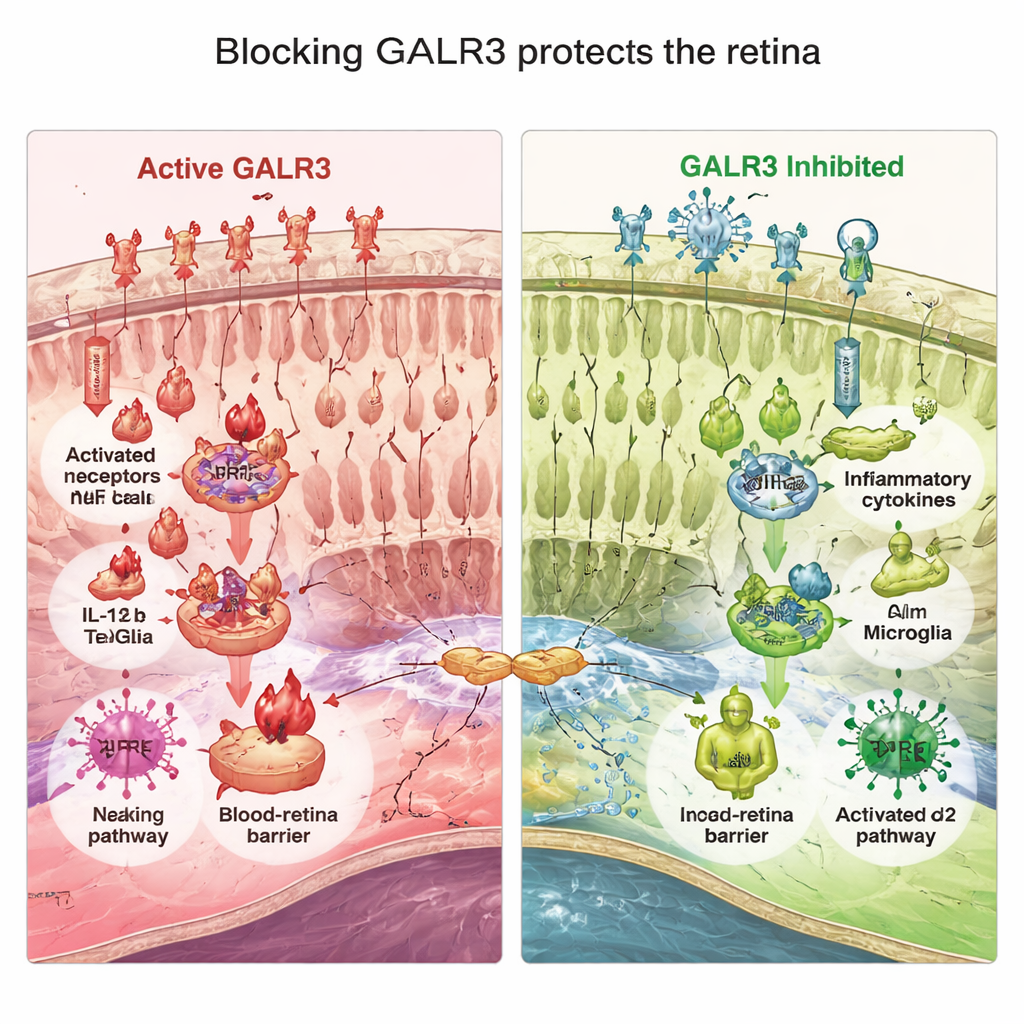
The team then asked why GALR3 inhibition helped. In retinitis pigmentosa, support cells in the retina—Müller glia and microglia—shift from caretakers into overactive responders that fuel chronic inflammation. In P23H mice, markers of this “alarm state,” including GFAP in glial cells and IBA-1 in microglia, were strongly increased, and a suite of inflammatory molecules and danger-sensing complexes, such as NF-κB and the NLRP3 inflammasome, were elevated. Blocking GALR3 calmed this response: glial activation was reduced, pro-inflammatory cytokines dropped, and anti-inflammatory signals linked to tissue repair, like IL‑10 and TGF‑β1, rose early in disease. In parallel, GALR3 inhibition strengthened the retina’s antioxidant machinery. It boosted the activity of NRF2, a master controller of antioxidant genes, and increased enzymes that neutralize reactive oxygen species, helping to limit further damage from metabolic stress.
Protecting the eye’s inner barrier
Healthy vision also depends on a tight “blood–retina barrier” that keeps unwanted molecules and immune cells from flooding into the delicate neural tissue. In P23H mice, this barrier began to leak: junctions between retinal pigment epithelium cells became ragged, and blood proteins seeped into the retina. Both genetic removal and drug blockade of GALR3 greatly reduced these defects, preserving the neat, tile-like pattern of the barrier and limiting leakage. In cell culture experiments, photoreceptor-like cells exposed to inflammatory signals from activated microglia ramped up their own inflammatory genes, but this effect was blunted when GALR3 in the photoreceptor cells was silenced. Together, these findings show that GALR3 sits at a crossroads linking microglial activation, oxidative stress, and barrier breakdown to photoreceptor death.
A mutation-agnostic path toward saving sight
For people with retinitis pigmentosa, especially those who may never receive gene-specific therapies, strategies that broadly slow retinal damage are highly valuable. This study identifies GALR3 as a key “volume knob” on harmful inflammation and oxidative stress in a widely used mouse model of the disease. Blocking GALR3 did not cure retinitis pigmentosa, but it consistently slowed photoreceptor loss, improved retinal responses to light, reduced toxic inflammation, strengthened natural antioxidant defenses, and helped maintain the eye’s internal barrier. Because GALR3 acts on stress and immune pathways rather than on a single mutation, drugs that target this receptor could, in principle, help patients with many different genetic causes of retinal degeneration.
Citation: Azam, M., Pashandi, Z., Liu, M. et al. Inhibition of galanin receptor 3 slows down retina degeneration in retinitis pigmentosa through modulation of inflammatory and oxidative stress response. Sci Rep 16, 7765 (2026). https://doi.org/10.1038/s41598-026-38345-6
Keywords: retinitis pigmentosa, retinal inflammation, photoreceptor survival, galanin receptor 3, oxidative stress