Clear Sky Science · en
DFT study of frontier orbitals and NLO properties of a phenanthroline and nitrophenol complex
Light, Molecules, and Future Tech
Everyday technologies—from smartphone screens to high‑speed internet—depend on materials that can control light and electric charge with great precision. This study explores a tiny paired system of two common organic molecules, 1,10‑phenanthroline and p‑nitrophenol, that lock together through a hydrogen bond and share electrical charge. By understanding how and why this “charge‑sharing partnership” forms and how it responds to light, scientists hope to design better components for sensors, optical switches, and next‑generation photonic devices.
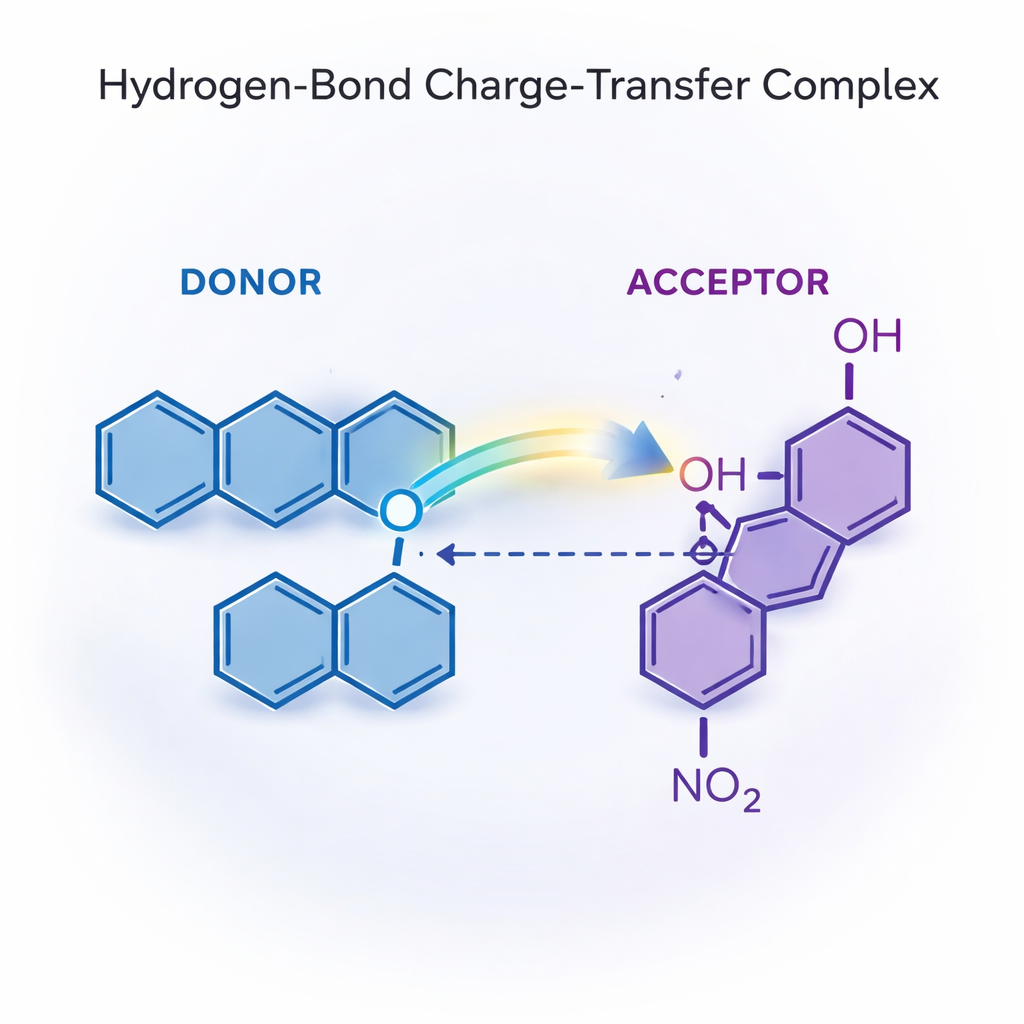
A Molecular Partnership Built on a Hydrogen Bond
The work focuses on a special kind of alliance called a hydrogen‑bond charge‑transfer complex. Here, one molecule plays the role of electron donor and the other acts as electron acceptor, with a hydrogen bond acting like a bridge between them. The authors show that when 1,10‑phenanthroline and p‑nitrophenol approach each other, the acidic hydrogen of p‑nitrophenol shifts toward nitrogen atoms on phenanthroline. This creates a strong, directional hydrogen bond and partial proton transfer, which in turn encourages electrons to move from one partner to the other. The result is a tightly held pair whose structure is distinctly different from that of the separate molecules.
Peering into Structure with Theory and Spectra
To reveal how this complex is put together, the researchers combine several experimental techniques with powerful quantum‑chemical calculations known as density functional theory. They model the optimal arrangement of the atoms, confirm that the predicted structure is stable, and examine key bond distances and angles that signal strong hydrogen bonding. Infrared spectroscopy tracks how specific bond vibrations shift when the complex forms, while nuclear magnetic resonance (NMR) shows how the local electronic environment of hydrogen and carbon atoms changes. Together, these measurements verify that a genuine hydrogen‑bond‑stabilized charge‑transfer complex has formed and that a proton has largely moved from p‑nitrophenol to phenanthroline.
How Electrons Move and Light Is Absorbed
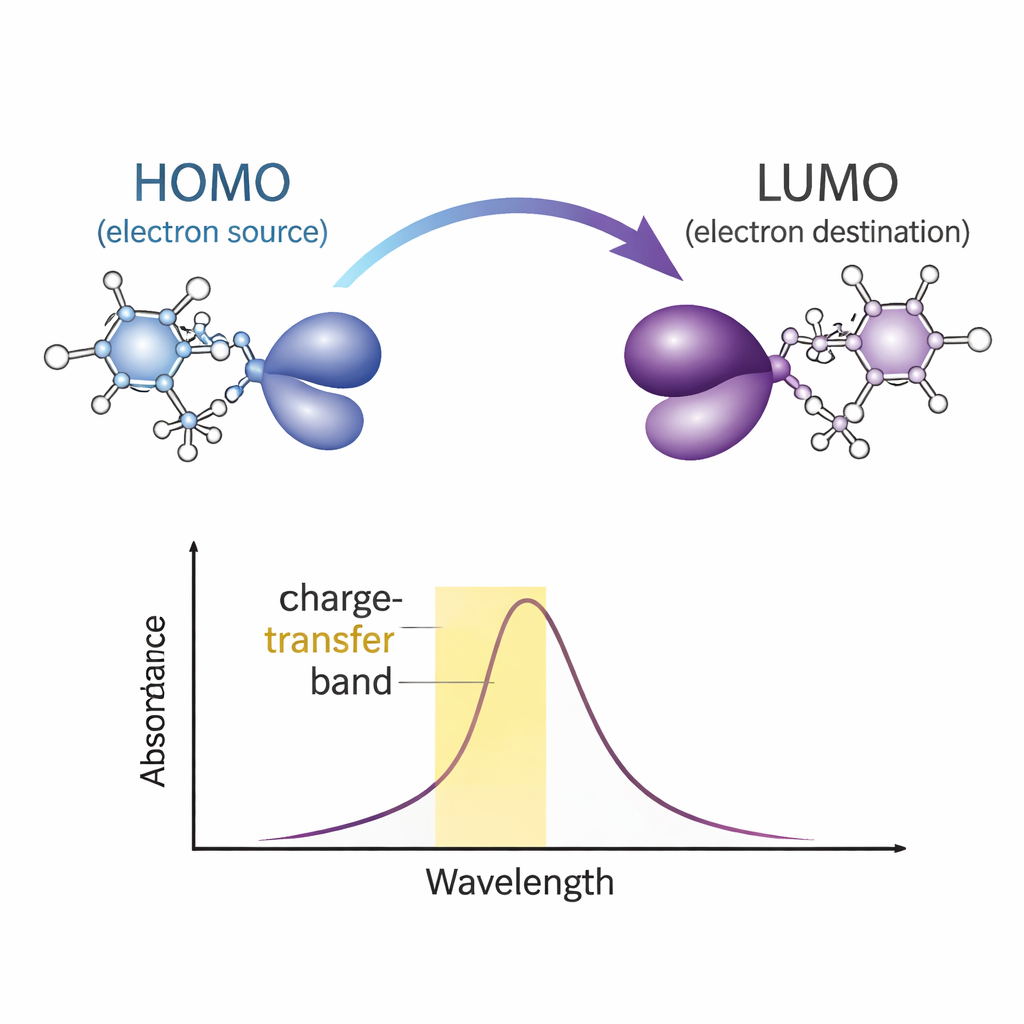
The team then asks how this pairing changes the way the system absorbs light and moves charge. Using both measured and computed ultraviolet–visible (UV–Vis) spectra, they identify a characteristic charge‑transfer band: a broad absorption peak that appears only when the two molecules form the complex. Frontier orbital analysis—looking at the highest occupied and lowest unoccupied electronic states—shows that the electron that is promoted by light effectively travels from one molecular fragment to the other across the hydrogen bond. The energy gap between these frontier orbitals indicates a complex that is electronically stable but primarily active under ultraviolet light, a useful trait for UV‑responsive materials.
Mapping Forces and Hidden Interactions
Beyond simple bonding pictures, the authors use detailed electron‑density analyses to see where charge actually gathers and how weak forces contribute to stability. Electrostatic potential maps highlight regions that are electron‑rich or electron‑poor, pinpointing the most reactive spots on each molecule and clarifying why the hydrogen bond forms where it does. Natural bond orbital calculations quantify how much electron density flows from donor to acceptor, confirming that phenanthroline donates charge while p‑nitrophenol receives it. Additional tools, such as reduced‑density‑gradient plots and atoms‑in‑molecules topology, visualize subtle non‑covalent attractions and repulsions—van der Waals contacts, hydrogen bonds, and π–π interactions—that help lock the complex in place.
From Molecular Detail to Optical Function
One especially promising outcome of this intricate picture is the prediction of strong non‑linear optical behavior: the complex is calculated to respond to intense light fields about twenty times more strongly than a standard reference material used in optics. In plain terms, this small hydrogen‑bonded pair can bend and mix light in ways that are valuable for optical switching, signal processing, and advanced photonic circuits. By showing exactly how hydrogen bonding and charge transfer reshape structure, charge distribution, and light absorption, the study provides a recipe for designing similar organic complexes with tunable electronic and optical properties—tiny molecular building blocks that could underpin future light‑based technologies.
Citation: Hadigheh Rezvan, V., Barani Pour, S., Dabbagh Hosseini Pour, M. et al. DFT study of frontier orbitals and NLO properties of a phenanthroline and nitrophenol complex. Sci Rep 16, 7754 (2026). https://doi.org/10.1038/s41598-026-38340-x
Keywords: charge transfer complex, hydrogen bonding, nonlinear optics, frontier orbitals, UV–Vis spectroscopy