Clear Sky Science · en
DGCR8 regulates multiple processes of transcription coupled nucleotide excision repair
How Sunlight Talks to Our Genes
Every time we step into the sun, invisible ultraviolet (UV) rays strike our skin and damage the DNA in our cells. Most of the time, our cells quietly fix this damage before it causes trouble. This study uncovers a surprising player in that protection system: a protein called DGCR8, previously famous for helping cells make microRNAs. The researchers show that DGCR8 can switch jobs after UV exposure, coordinating several steps of a key DNA repair pathway that helps prevent cancer and other diseases.
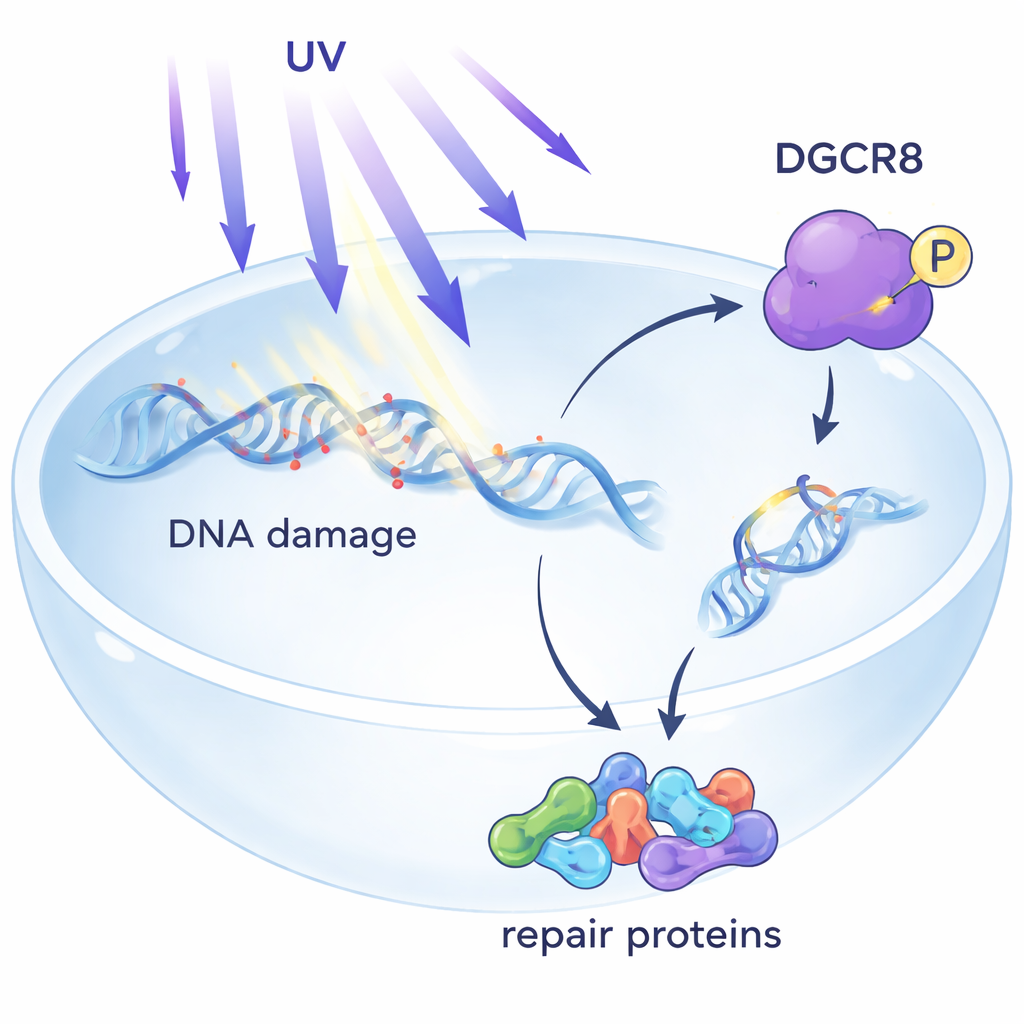
A Hidden Guardian Inside DNA Repair
UV light creates tiny lesions in DNA that can block the molecular machines that read genes. One of the body’s main defenses is transcription-coupled nucleotide excision repair, or TC-NER, which focuses on damage that stalls gene-reading enzymes. Until now, DGCR8 was mainly known for processing microRNAs, short RNA molecules that fine-tune gene activity. Earlier work hinted that DGCR8 also protects cells from UV, but how it did so was unclear. This paper shows that, upon UV exposure, DGCR8 physically associates with central TC-NER factors, acting as an organizer that brings repair components together right where gene reading is blocked.
A Molecular On–Off Switch for DGCR8
The key to DGCR8’s new role is a single chemical tag on one of its building blocks, the amino acid serine at position 153. When this site is phosphorylated—meaning a phosphate group is added—DGCR8 gains the ability to bind to core TC-NER players, including RNA polymerase II (the gene-reading enzyme) and repair factors called CSB, CSA, and UVSSA. Cells engineered with a version of DGCR8 that cannot be phosphorylated at this site (the S153A mutant) showed weak or lost interactions among these repair proteins, and the normal UV-triggered assembly of the repair complex failed. A “phosphomimic” version that behaves as if it is always phosphorylated had the opposite effect, strengthening key interactions even without UV. These results indicate that phosphorylation at Ser153 acts as an on–off switch that flips DGCR8 from its usual RNA-processing role into a DNA repair coordinator.
Untangling Dangerous DNA–RNA Knots
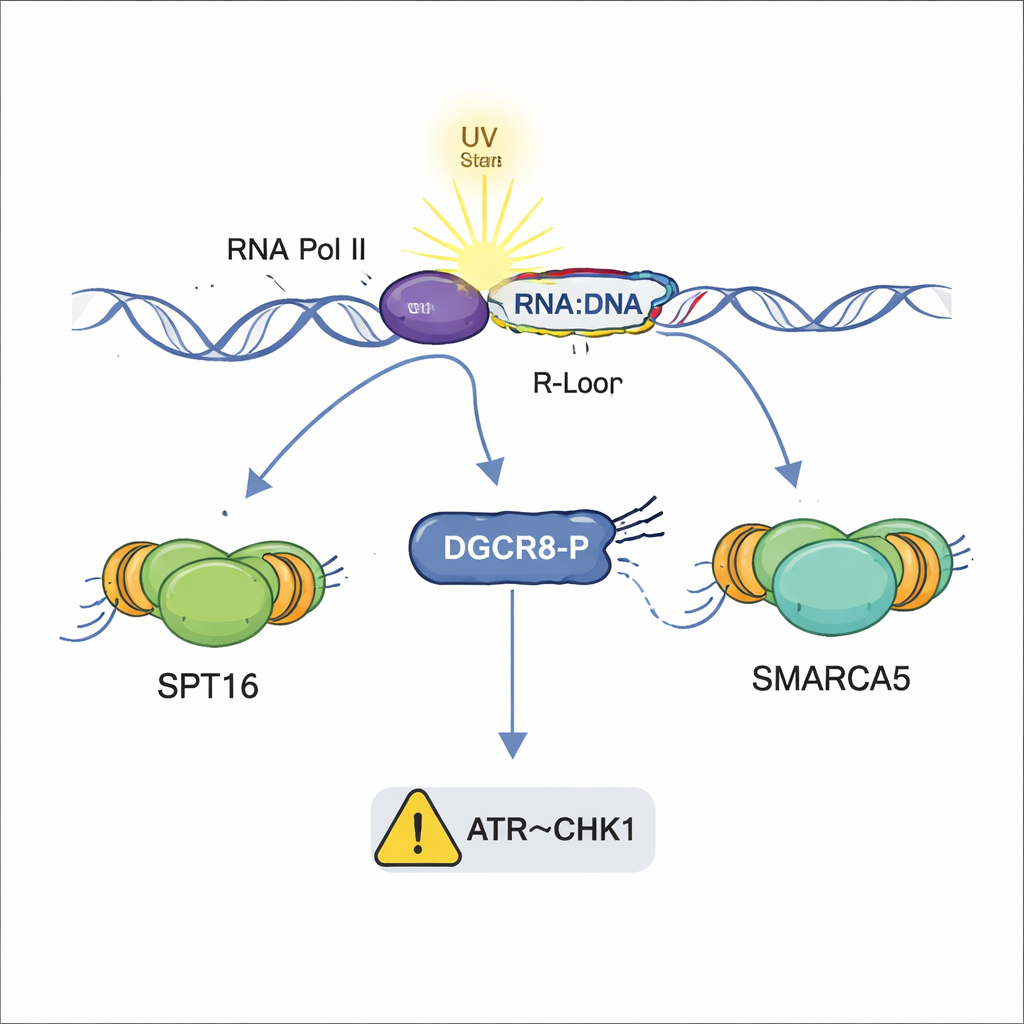
Beyond gathering repair proteins, DGCR8 also helps manage structures called R-loops—short stretches where newly made RNA reattaches to the DNA it came from, forming a DNA–RNA hybrid. In small amounts, R-loops can be useful, but when they accumulate after UV exposure, they can stall DNA copying and destabilize chromosomes. Using specialized antibodies and fluorescent probes, the authors found that cells lacking DGCR8, or carrying the non-phosphorylatable S153A form, build up far more R-loops after UV. Phosphorylated DGCR8 was seen at these hybrid structures, especially where they overlapped with UV-induced lesions, suggesting it helps prevent or clear harmful R-loops precisely at active genes under stress.
Keeping DNA Copying in Check
When DNA damage or R-loops interfere with copying the genome, cells normally slow replication through a safety circuit known as the ATR–CHK1 checkpoint. Fiber-tracking experiments showed that cells with normal DGCR8 slowed their replication tracks after UV exposure, reflecting an engaged checkpoint. In striking contrast, cells missing DGCR8 or carrying the S153A mutation continued replicating at nearly normal speed, even though they had accumulated DNA damage and R-loops. Further tests revealed that while ATR itself was activated, its key partner CHK1 was poorly switched on in these mutant cells. This indicates that phosphorylated DGCR8 helps relay the stress signal from damaged DNA and R-loops to CHK1, allowing the cell to pause and stabilize replication.
Why This Matters for Health and Disease
Taken together, the findings reveal DGCR8 as a multi-talented guardian that coordinates several layers of the UV damage response: assembling the TC-NER repair team, recruiting chromatin remodelers that open up DNA, restraining harmful DNA–RNA hybrids, and helping to trigger a replication slowdown through the ATR–CHK1 pathway. Crucially, these actions are separate from DGCR8’s traditional job in microRNA biogenesis and depend on phosphorylation of a single site. Because faults in DNA repair and R-loop control are linked to cancer and neurological diseases, and DGCR8 mutations have been found in some tumors, understanding this molecular switch may open new avenues for diagnosing or treating conditions driven by UV damage and genome instability.
Citation: Watanabe, T., Yoshinami, D., Yamasaki, H. et al. DGCR8 regulates multiple processes of transcription coupled nucleotide excision repair. Sci Rep 16, 7112 (2026). https://doi.org/10.1038/s41598-026-38338-5
Keywords: UV DNA damage, DGCR8, DNA repair, R-loops, genome stability