Clear Sky Science · en
Experimental and computational study of photocatalytic degradation of volatile organic compounds in bitumen using sulfur-doped phenol-rich bio-oils
Why Road Surfaces Matter for the Air We Breathe
Every time the sun beats down on a blacktop road, invisible fumes rise into the air. These fumes contain volatile organic compounds (VOCs), a large family of chemicals that can harm human health and worsen urban smog. This study explores a new, bio-based way to redesign asphalt so that it traps and even breaks down many of these fumes before they can escape, potentially making city air cleaner while also helping roads last longer.
Hidden Pollution from Everyday Asphalt
Asphalt pavements are heated during manufacture, transport, and paving, but they also emit VOCs for years afterward whenever they are warmed by sunlight. These vapors contain thousands of different molecules, some toxic and some that help form airborne particles and ground-level ozone. Ultraviolet (UV) radiation from the sun speeds up the breakdown and aging of asphalt, releasing even more VOCs and gradually weakening the road surface. Engineers have tried adding various chemical modifiers to slow this aging, but many of those additives themselves raise environmental or health concerns.
Turning Plant Waste and Sulfur into a Road Protector
The researchers focused on a greener alternative: bio-oils made from wood pellets that are rich in phenolic compounds—ring-shaped molecules similar to those found in plants. Separately, road engineers have long experimented with adding elemental sulfur, a cheap by-product of oil and gas refining, to improve asphalt’s strength. Under UV light, sulfur in asphalt can form highly reactive sulfur radicals, which are usually viewed as troublemakers because they accelerate aging. This work flips that script. The team proposes combining wood-derived phenolic bio-oils with sulfur to create a new kind of “radical sponge” inside asphalt that both strengthens the material and grabs VOC molecules, locking them into more stable forms instead of letting them drift into the air.
How the Molecular Trap Works
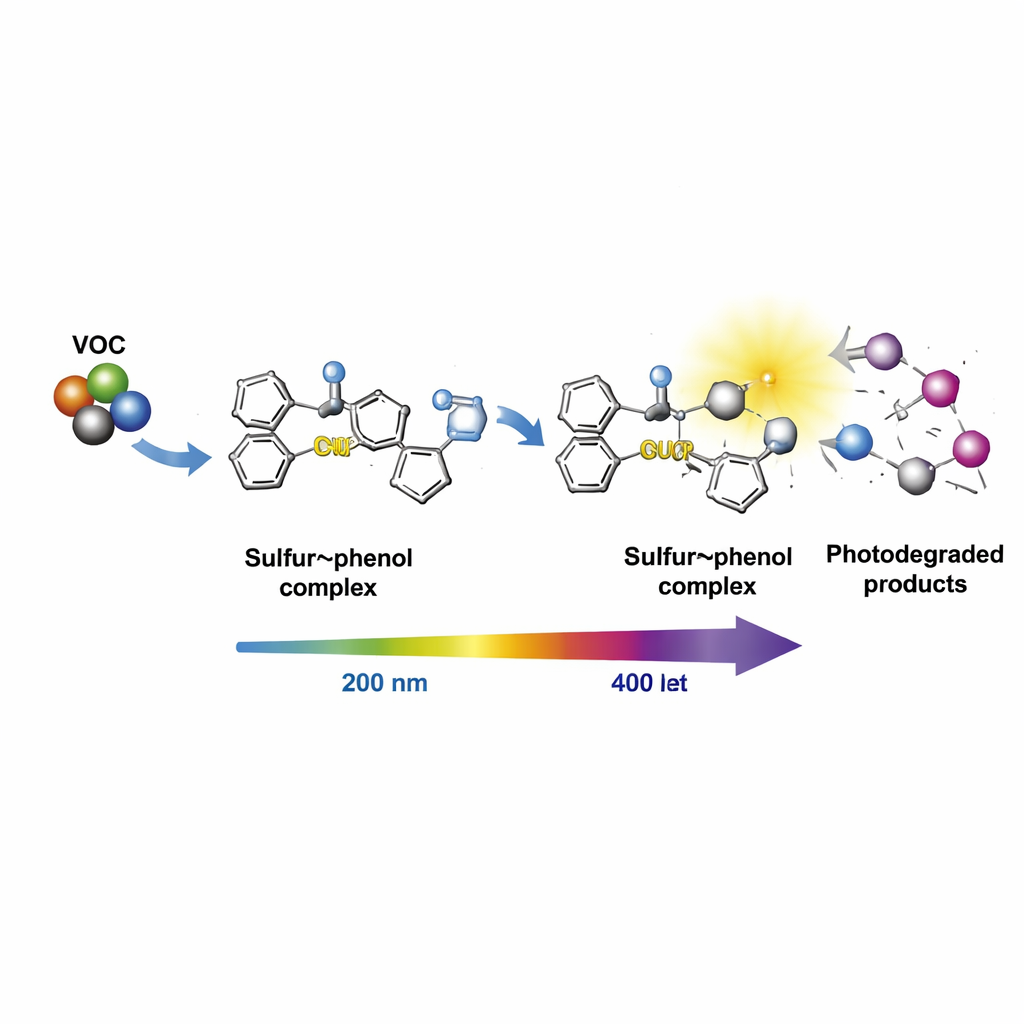
At the molecular level, UV light breaks open rings of elemental sulfur, producing a short chain of four sulfur atoms with an unpaired electron—a sulfur radical. This radical reacts with the phenolic rings in the bio-oil, forming a sulfur–phenol assembly that is still reactive but more stable. The study shows, through detailed computer modeling, that this assembly then readily hooks onto common VOC molecules via a carbon–sulfur (C–S) bond-forming step. The energy calculations indicate that these reactions are not only possible but favorable under realistic asphalt conditions, meaning the VOCs can be captured efficiently as traffic and sunlight heat the pavement.
Making Fumes Easier to Destroy with Light
The story does not end with capture. On their own, most VOCs absorb only very short-wavelength UV light, below about 200 nanometers, which is scarce at the Earth’s surface. That makes them stubbornly resistant to direct sunlight-driven breakdown. By forming bonded complexes with the sulfur–phenol assemblies, however, the VOCs’ light-absorption behavior changes dramatically. Both computer simulations and lab measurements of UV–visible spectra show that the main absorption peaks shift from around 200 nanometers to near 400 nanometers—a “red shift” into the near-UV and visible range where sunlight is abundant. The new complexes therefore soak up more of the light that actually reaches roads, making it easier for that light to trigger chemical reactions that break the VOCs down into heavier, less volatile species that stay locked in the asphalt.
Evidence from the Lab Bench
To confirm this mechanism beyond simulations, the researchers synthesized model sulfur–phenol–VOC compounds and examined them with several tools. UV–visible spectroscopy clearly showed the predicted red shift in light absorption after the VOCs reacted with the sulfur-doped bio-oils. Infrared spectroscopy detected specific bonds, including carbon–sulfur and sulfur–sulfur linkages, that signal successful coupling between the ingredients. Mass spectrometry revealed molecular weights and fragmentation patterns consistent with the proposed sulfur-rich structures. Together, these methods build a coherent picture: wood-derived phenolic oils and sulfur radicals can join forces to capture VOCs and turn them into light-sensitive targets for photocatalytic degradation.
What This Could Mean for Future Cities
For a non-specialist, the takeaway is straightforward: by carefully redesigning what goes into asphalt, it may be possible to turn roads from a steady source of harmful fumes into an active sink that traps and dismantles them. The sulfur-doped, phenol-rich bio-oils studied here are made from abundant industrial by-products—sulfur from refineries and bio-oils from waste wood—so they could be cost-effective as well as environmentally friendly. If scaled up, such modified asphalts could help reduce urban air pollution, slow the aging of pavements, and make better use of waste materials, offering a practical route toward cleaner and more resilient city infrastructure.
Citation: Almasi, M., Neyband, R.S. Experimental and computational study of photocatalytic degradation of volatile organic compounds in bitumen using sulfur-doped phenol-rich bio-oils. Sci Rep 16, 7779 (2026). https://doi.org/10.1038/s41598-026-38337-6
Keywords: asphalt emissions, volatile organic compounds, photocatalytic road materials, bio-based modifiers, urban air pollution