Clear Sky Science · en
PARP-1 couples β-catenin/TCF4 signaling to epithelial–mesenchymal transition in endometriosis
Why this research matters for women’s health
Endometriosis is a painful condition in which tissue similar to the lining of the uterus grows in the wrong places, such as on the ovaries. These misplaced patches can invade nearby structures, cause scarring, and contribute to infertility, yet current treatments are often imperfect and symptoms frequently return. This study explores a molecular “switch” called PARP‑1 and asks whether it helps endometriosis lesions become more invasive—and whether existing drugs that block PARP‑1 might gently turn that switch down.
A closer look at invasive lesion behavior
Although endometriosis is classified as a benign disease, the ectopic (misplaced) tissue often behaves in a surprisingly aggressive way: it migrates, implants, and persists much like a slow‑moving cancer. One key process behind this behavior is called epithelial–mesenchymal transition, or EMT, in which orderly, sheet‑forming cells loosen their connections and adopt a more mobile, fibrous identity. The authors examined tissue from women with and without endometriosis and found that ovarian endometriotic lesions showed a clear EMT‑like pattern: the “glue” protein E‑cadherin was reduced, while markers of more mobile cells, such as N‑cadherin and vimentin, were increased. At the same time, levels of PARP‑1 and components of a growth‑control pathway known as Wnt/β‑catenin were highest in these lesions, suggesting that all of these changes move together.
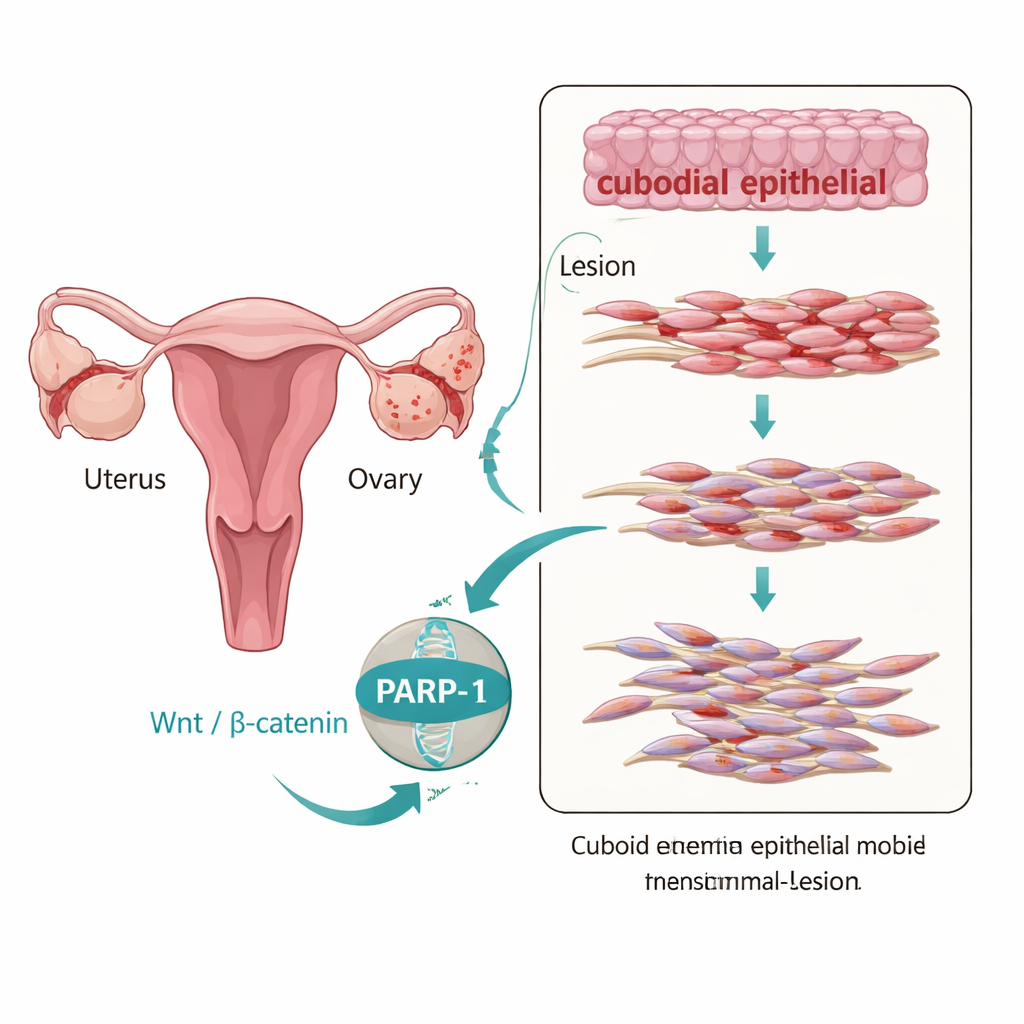
The molecular bridge between signals and cell change
To find out whether PARP‑1 is simply present or actively driving these changes, the researchers used an immortalized endometriotic epithelial cell line called 12Z. When they artificially raised PARP‑1 levels in these cells, EMT markers shifted toward a more invasive profile and the cells became more mobile and viable in lab tests that mimic wound healing and tissue invasion. When they reduced PARP‑1 using targeted RNA molecules, the pattern reversed: E‑cadherin increased, mesenchymal markers dropped, and the cells moved and grew less. Importantly, PARP‑1 physically associated with β‑catenin and TCF4, two key factors that translate Wnt signals into gene activity, pointing to PARP‑1 as a bridge that helps link outside cues to the EMT gene program inside the nucleus.
Dialing back the switch with a cancer drug
Because medicines that block PARP activity are already used in cancer care, the team next asked whether one such drug, olaparib, could soften EMT‑like traits and reduce lesion growth in a mouse model of endometriosis. Mice implanted with small fragments of uterine tissue developed ectopic lesions in the abdomen, mimicking aspects of human disease. When treated with olaparib for two weeks, these animals had markedly smaller total lesion volume and lighter uteri with adherent lesions compared with untreated controls. Tissue from treated mice also showed a more “epithelial” marker pattern—higher E‑cadherin and lower N‑cadherin, vimentin, β‑catenin, and TCF4—indicating that PARP inhibition had pushed cells away from the invasive EMT‑like state.
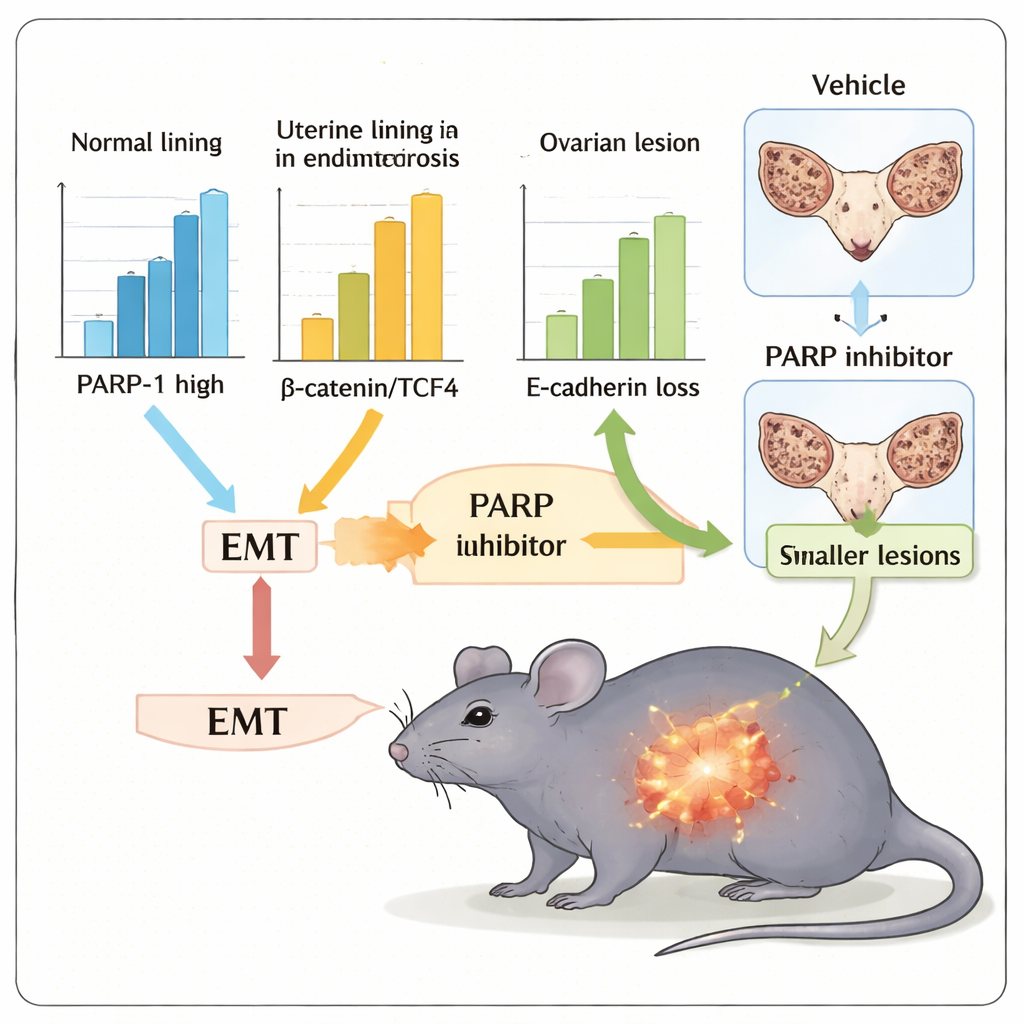
What this could mean for future care
Taken together, findings from patient samples, cell culture experiments, and the mouse model support a unifying idea: PARP‑1 acts as a drug‑targetable hub that couples Wnt/β‑catenin signaling to EMT‑like behavior in endometriosis. Rather than being the single master switch, PARP‑1 appears to be a key connector in a larger network of inflammation, oxidative stress, and hormone‑driven signals that shape how lesions grow and invade. Because PARP inhibitors already exist, this work raises the possibility of repurposing or adapting them—perhaps in combination with other pathway or immune‑directed drugs—to reduce lesion burden, scarring, and possibly the impact on fertility. Larger and longer‑term studies will be needed, but this research opens a concrete path toward more targeted, mechanism‑based therapies for a condition that affects millions of women worldwide.
Citation: Zhang, L., Li, X., Kong, L. et al. PARP-1 couples β-catenin/TCF4 signaling to epithelial–mesenchymal transition in endometriosis. Sci Rep 16, 6940 (2026). https://doi.org/10.1038/s41598-026-38335-8
Keywords: endometriosis, PARP-1, epithelial–mesenchymal transition, Wnt/β-catenin, PARP inhibitor therapy