Clear Sky Science · en
Changes in circulating small non-coding RNAs after castration in a cohort of prostate cancer patients
Why tiny messengers in blood matter
When men with advanced prostate cancer undergo castration treatment, doctors focus mainly on lowering testosterone. But the testes release more than hormones. They also send out swarms of tiny RNA molecules into the bloodstream that may act as long‑distance messengers. This study asks a simple but far‑reaching question: what happens to those microscopic signals in the blood when testicular function is shut down?
A closer look at microscopic signals
Our blood contains countless fragments of genetic material known as small non‑coding RNAs. Unlike regular genes, they do not build proteins, but they can fine‑tune which genes are turned on or off. Some of these RNAs, including microRNAs and a lesser‑known group called piRNAs, are especially abundant in the testes, where they are crucial for sperm production. Because these molecules are surprisingly stable in blood, they can serve as fingerprints of what is happening inside organs we cannot easily sample.
Following patients through two types of castration
The researchers used samples from 57 men with advanced prostate cancer who had already taken part in a clinical trial. Half were treated with an injected drug that switches off the hormone signal from the brain to the testes (a GnRH agonist). The other half had a subcapsular orchiectomy, a surgery that removes the hormone‑producing tissue of the testes. Blood was drawn before treatment and again 12 and 24 weeks later. From these samples, the team isolated small RNAs and used high‑throughput sequencing to count more than 60,000 different RNA species.
Big changes in tiny molecules after treatment
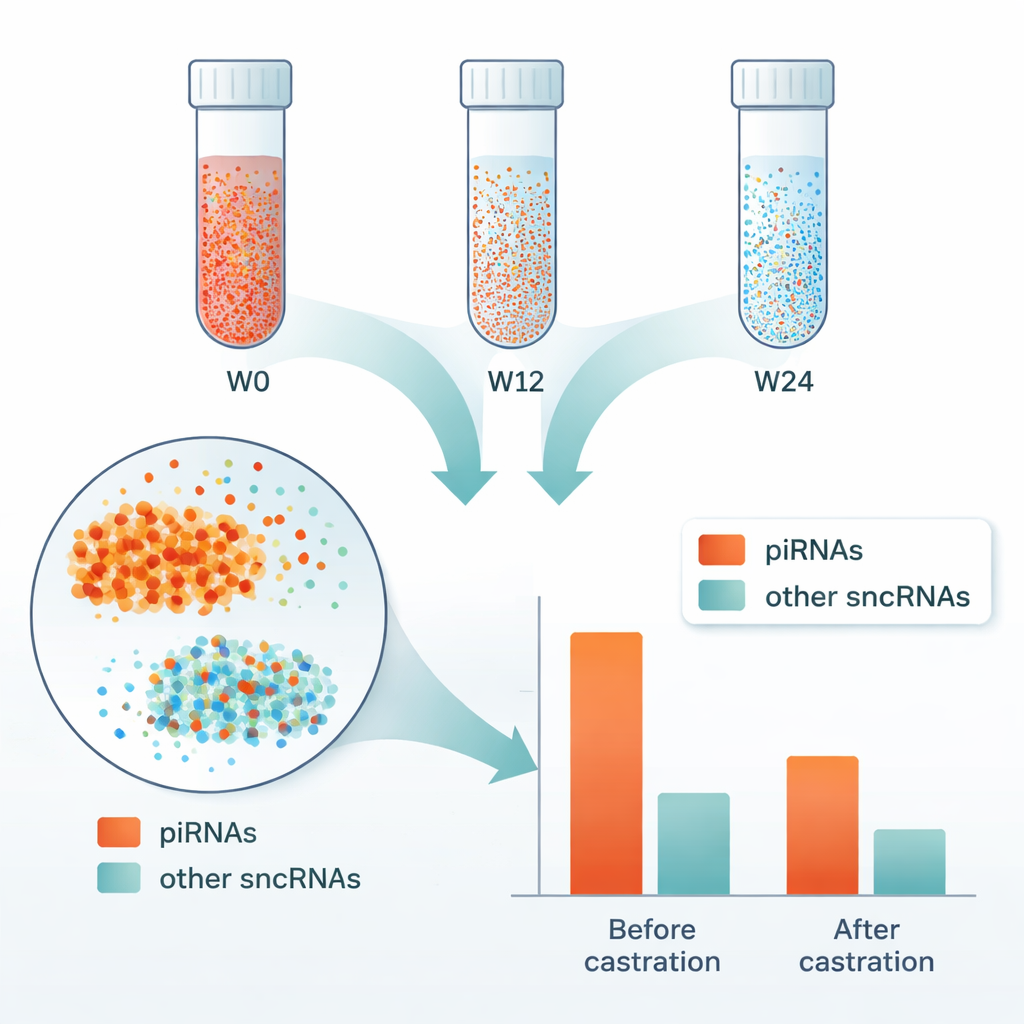
When the scientists compared RNA levels before and after castration, they saw striking shifts. In both the surgical and drug‑treated groups, dozens to hundreds of small RNAs changed, and the vast majority decreased over time. The most affected class were piRNAs, which are normally concentrated in germ cells in the testes. After 12 and 24 weeks, 83–86% of the altered RNAs were present at lower levels, and piRNAs made up almost half or more of these changes. This pattern strongly suggests that many of the circulating RNAs had come from the testes and were lost when testicular tissue was removed or shut down.
Tracing the likely origin of key RNAs
To focus on the strongest signals, the team looked for RNAs that changed consistently in both treatment arms and at both follow‑up visits. They identified 16 such molecules, including eight piRNAs and several other RNA types. Database searches showed that most of them are expressed in testis tissue, and a few also appear in the prostate. Two candidates, called miR‑153 and SNORD38A, were examined more closely. Laboratory tests on human tissues confirmed that these RNAs are present in the testis, and staining of testicular biopsies revealed that SNORD38A is especially abundant in early sperm‑forming cells. The drop in their blood levels after treatment is therefore most likely caused by the loss of testicular secretion.
What this means for patients and future research
Although both treatments aim to lower testosterone, they do so differently, and the RNA profiles reflected this. Some small RNAs differed between the surgery and drug groups, hinting that the exact way the testes are switched off could leave a distinct molecular signature. Importantly, the study cannot yet show that these RNAs act as true hormones that carry messages to distant organs. Still, the work demonstrates that castration reshapes the landscape of small RNAs in blood and highlights specific candidates with a likely testicular origin.
Take‑home message
For men undergoing castration as part of prostate cancer treatment, this research shows that the body loses more than testosterone. It also loses a cloud of tiny RNA molecules, particularly piRNAs made in the testes, that normally circulate in the bloodstream. Scientists do not yet know whether these lost signals have direct effects on other organs, but the findings offer a new window into how the testes communicate with the rest of the body and may eventually help in developing blood‑based markers of testicular function and treatment response.
Citation: Main, A.M., Sørensen, L.H., Winge, S.B. et al. Changes in circulating small non-coding RNAs after castration in a cohort of prostate cancer patients. Sci Rep 16, 7060 (2026). https://doi.org/10.1038/s41598-026-38334-9
Keywords: prostate cancer, testosterone suppression, small non-coding RNA, piRNA, endocrine biomarkers