Clear Sky Science · en
A novel AI-coupled flow chamber method quantifying erythrocyte osmotic fragility
Why Red Blood Cell Fragility Matters
Every second, millions of your red blood cells squeeze through hair‑thin vessels while carrying oxygen. How easily these cells burst under stress—called their “fragility”—can reveal anemia, blood storage problems, and complications of severe infections like sepsis. This study introduces a modern, AI‑assisted microscope device that measures how quickly red blood cells fall apart in salty solutions, aiming to make this important test faster, more precise, and easier to run in both research labs and, one day, hospitals.
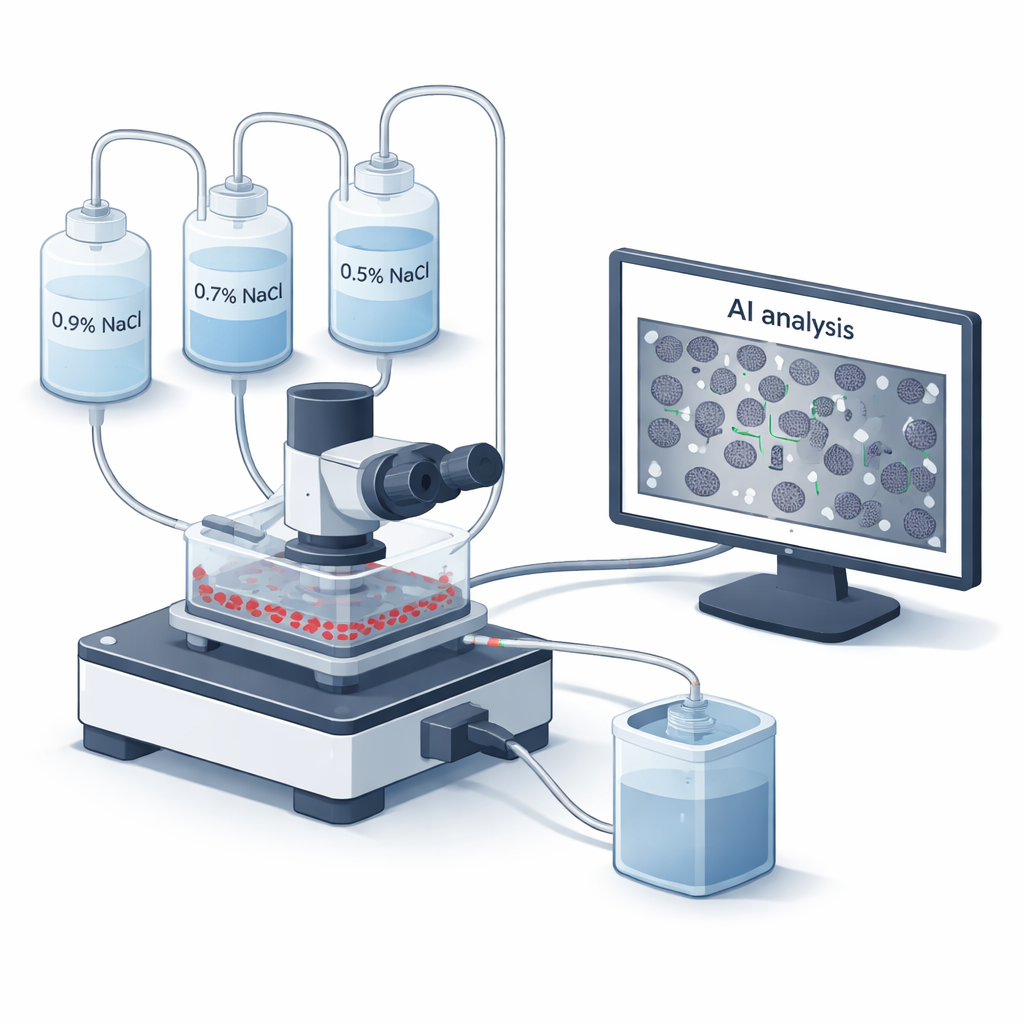
A New Way to Watch Blood Cells Under Stress
Doctors have used osmotic fragility tests since the 1940s to see how sturdy red blood cell membranes are. Traditionally, blood is mixed with a series of salt solutions, and a machine reads how much hemoglobin leaks out when cells burst. While reliable, this classic method is slow, manual, and blind to what individual cells actually look like as they fail. The team behind this paper built a new microfluidic “flow chamber” system, called BioExP, that spreads red blood cells as a thin monolayer on a treated glass slide. Different salt solutions are pumped across this layer while a microscope and camera record what happens in real time, and built‑in software uses artificial intelligence to count how many cells remain intact at each salt level.
Putting AI and Flow Control to the Test
To judge whether BioExP is trustworthy, the researchers compared it head‑to‑head with the old‑style spectrophotometer assay using blood from four healthy donors. They focused on a key number called MCF₅₀: the salt concentration at which half of the red cells have burst. First, they had to tune their new system by finding the best “satiation time”—how long to expose cells to a given low‑salt solution so that most vulnerable cells have lysed without overdoing it. By tracking how many cells disappeared at 0.4% salt over 15 minutes, they found that nearly 80% of all hemolysis happened in the first three minutes. This three‑minute window became their standard exposure time, balancing speed with reliable measurements.
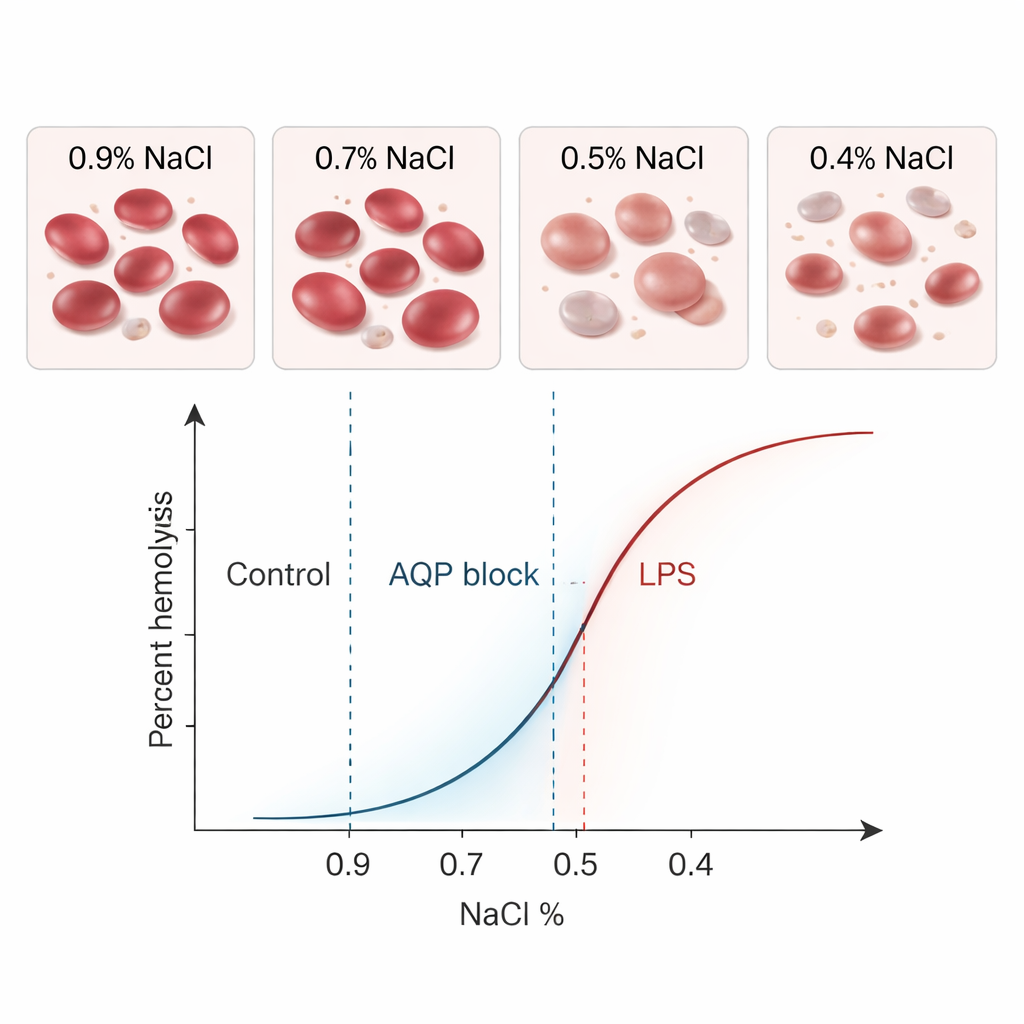
Matching the Classic Test and Probing Real Biology
When they ran the same healthy samples through both methods, BioExP and the classical test gave almost identical average MCF₅₀ values—0.41% salt—with similar variability. Statistical analyses showed no meaningful difference between methods, and most paired results fell within narrow limits of agreement. In other words, the new device was effectively “speaking the same language” as the established assay. The real power of BioExP, however, lies in its ability to test how various conditions change cell fragility. The researchers chose two biologically important modifiers: mercury chloride, which blocks water‑channel proteins called aquaporins in the cell membrane, and bacterial lipopolysaccharide (LPS), a toxin associated with sepsis.
How Toxins and Water Channels Change Cell Toughness
Blocking aquaporins made red blood cells harder to burst: the MCF₅₀ shifted to lower salt levels, meaning the cells tolerated more dilution before breaking. Both BioExP and the classical method detected this leftward shift clearly. LPS had the opposite effect. When red cells were incubated with a high dose of LPS in the absence of plasma, they became more fragile, bursting at higher salt concentrations. Under the microscope, many cells changed shape, becoming more spherical or spiky—visual signs that their membranes had been disturbed. Again, both methods recorded a strong, statistically significant rightward shift in the fragility curves. Across all these tests, the new flow‑chamber system consistently tracked the same biological trends as the older assay, just with slightly lower absolute values that were small and systematic.
What This Means for Patients and Labs
The BioExP platform cuts assay time from several hours to under an hour, needs only tiny blood volumes (potentially just a finger prick), and automates counting and curve‑fitting with AI rather than relying on many manual steps. Because it images the very same cells over time, it can connect how cells look with how easily they burst, and it can test many salt levels or drug conditions in a single run. The study shows that this modern approach can match the accuracy of the classic osmotic fragility test while adding speed, flexibility, and rich visual detail. If further validated in larger and more diverse groups of donors, such systems could become valuable tools for diagnosing hemolytic anemias, monitoring stored blood in banks, and exploring how infections or new drugs weaken or protect our red blood cells.
Citation: Fırat, I.S., Alaçayır, Ö., Creutz, T. et al. A novel AI-coupled flow chamber method quantifying erythrocyte osmotic fragility. Sci Rep 16, 7175 (2026). https://doi.org/10.1038/s41598-026-38322-z
Keywords: red blood cells, osmotic fragility, microfluidics, sepsis, artificial intelligence