Clear Sky Science · en
Spatiotemporal expression of endospore appendages and cryo-EM insights into Ena1C-mediated S-ENA anchoring in Bacillus paranthracis
Why stubborn bacterial spores matter
Bacteria in the Bacillus cereus family can turn into hardy spores that shrug off heat, chemicals and cleaning agents. These spores often cling to food-processing equipment and other surfaces, where they can cause food poisoning or contaminate products for long periods. This study looks at tiny hair-like fibers on these spores, called endospore appendages, and uncovers when they are built and how a key protein, Ena1C, locks them onto the spore surface. Understanding this process could help industry design better cleaning strategies and may also inspire new ways to engineer useful spores for agriculture or biotechnology.
Stiff hairs on sleeping bacteria
Under the microscope, spores from many Bacillus cereus group bacteria are covered in fine fibers that resemble bristles or pili. These endospore appendages are extremely tough and resist both enzymes and harsh chemicals, which has made them difficult to study. Earlier work using advanced cryo-electron microscopy had revealed that Bacillus paranthracis makes two main types of fibers: thick, staggered S-ENAs that make up about 90% of the hairs, and thinner, ladder-like L-ENAs that account for the rest. Genetic analyses had already linked specific ena genes to each type of fiber, but when and where these proteins appeared during spore formation, and how the S-ENAs were actually anchored to the spore, were still open questions.
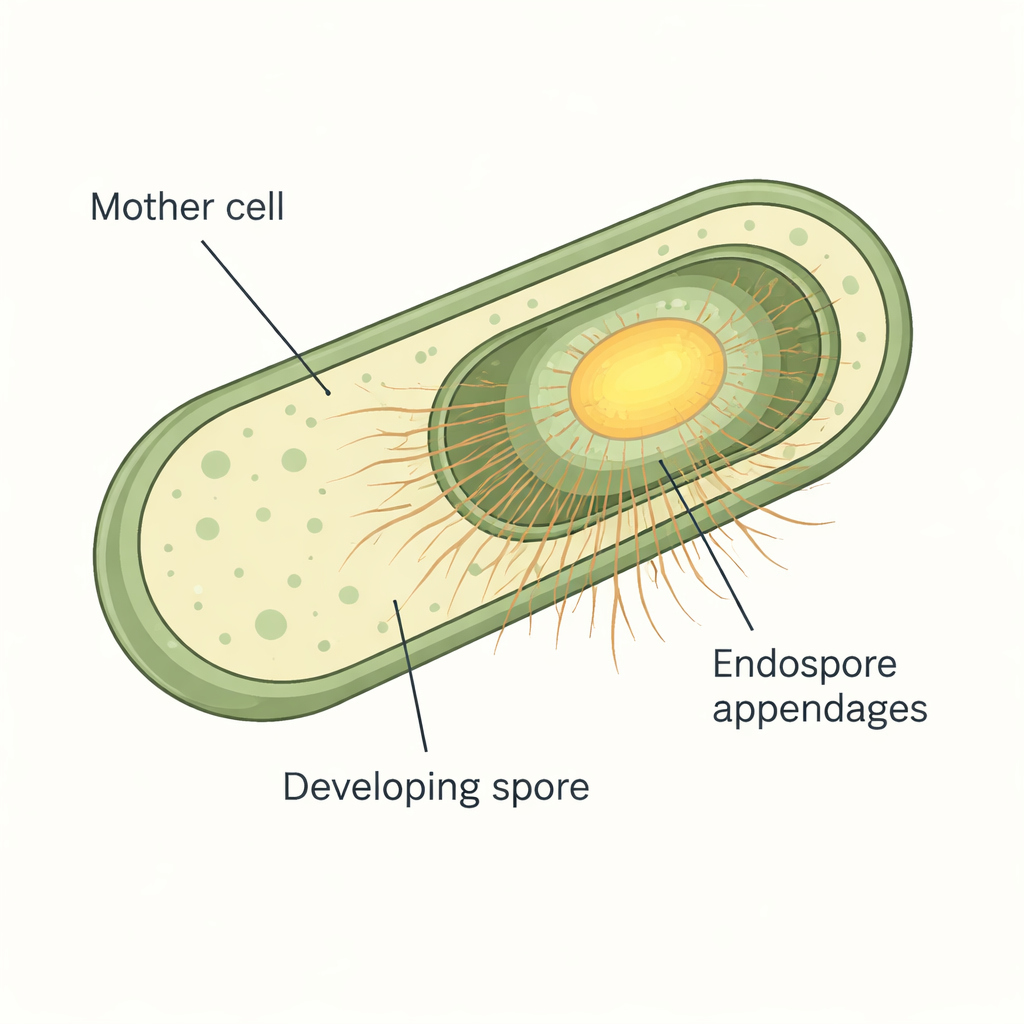
Watching fibers switch on in real time
To follow these building blocks as spores formed, the researchers fused the ENA proteins to bright fluorescent tags, allowing them to track each protein in living cells by time-lapse microscopy. They grew Bacillus paranthracis on specially prepared agar pads under the microscope and captured images every 10 to 12 minutes while cells progressed through sporulation. The team found that none of the ENA proteins were made while cells were still growing normally. Instead, production started only after the developing spores became "phase-bright," a visual sign that the spore core and protective layers had matured. ENA fluorescence then rose sharply late in sporulation and concentrated around the spore, especially at the interface between the mother cell and the forming spore, showing that these fibers are truly spore-specific decorations added near the end of the process.
Two fiber types on different schedules
The team also compared the timing of S-ENA and L-ENA production by tracking two proteins at once in the same cells. When S-ENA subunits (Ena1A or Ena1C) tagged in green were co-expressed with the L-ENA subunit Ena3A tagged in red, the green signal consistently appeared about an hour before the red. This staggered schedule matches what is known about spore architecture: S-ENAs emerge from the spore coat, which forms earlier, while L-ENAs are anchored to the exosporium, an outer sac added later. The findings suggest that the cell uses late-stage genetic switches to turn on ENA genes in a strict order, so that each fiber type is delivered to the correct spore layer at the right time.
A molecular “dock” that holds fibers in place
One of the most puzzling players was Ena1C, a protein required for S-ENA fibers to appear on spores, yet not part of the fiber stalk itself. By examining spores from bacteria lacking the ena1C gene, the researchers found that S-ENA fibers were still being assembled but floated free in the surrounding liquid instead of being attached to spores. When Ena1C was over-produced, spores carried many more S-ENAs, but each fiber was shorter, as though a limited pool of building blocks were being spread over extra attachment points. This pointed to Ena1C as a dedicated anchor that tethers S-ENA fibers to the spore coat, controlling both how many fibers attach and how long they grow.
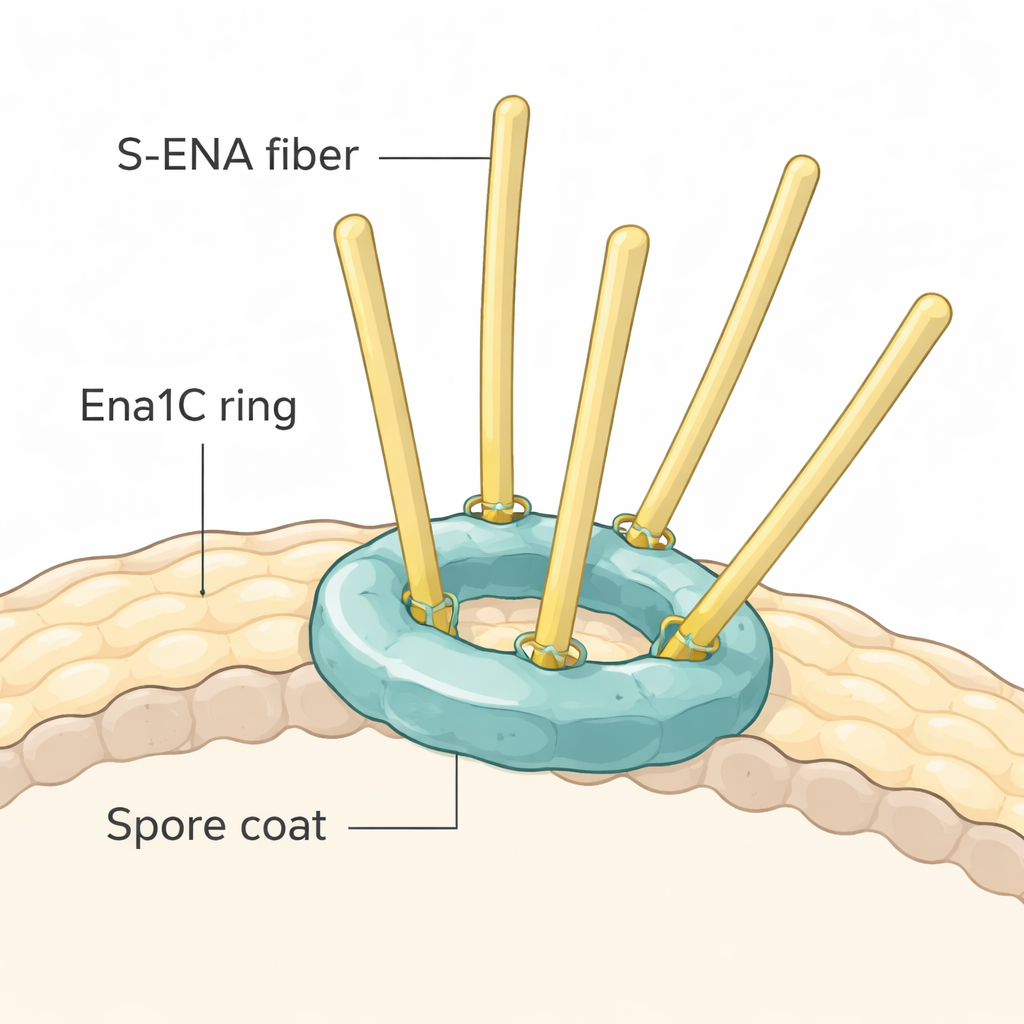
Ring-shaped anchors revealed by cryo-EM
To see how Ena1C might grip the fibers, the team purified the protein and imaged it using high-resolution cryo-electron microscopy. They discovered that Ena1C does not form long filaments. Instead, nine copies of the protein join to create a sturdy ring with a central opening, reinforced by multiple disulfide bonds—strong chemical links between sulfur-containing amino acids. Computer modeling and structural comparisons with known fiber proteins indicate that S-ENA stalks likely dock into the positively charged funnel at the center of this ring, where key cysteine residues on Ena1C can form disulfide bonds with matching sites on the fiber subunits. In this way, each Ena1C ring appears to serve as a molecular dock that clamps one or a few S-ENA fibers to the outer coat of the spore.
What this means for controlling stubborn spores
Taken together, the study shows that spore hairs in Bacillus paranthracis are produced only after the spore is mature, with S-ENA fibers built and anchored to the coat before L-ENAs are added to the exosporium. It also identifies Ena1C as a ring-shaped anchor that covalently links S-ENA fibers to the spore surface. For non-specialists, the key message is that the way spores stick to surfaces is not accidental: it is the result of a carefully timed construction project that adds robust, fiber-based Velcro to the outside of the spore. By targeting this anchoring system—either by disrupting Ena1C or altering ENA formation—future strategies may weaken spore adhesion, making industrial cleaning more effective, or conversely, allow engineers to design spores that deliberately bind to crops or materials in beneficial ways.
Citation: Zegeye, E.D., Sleutel, M., Jonsmoen, U.L. et al. Spatiotemporal expression of endospore appendages and cryo-EM insights into Ena1C-mediated S-ENA anchoring in Bacillus paranthracis. Sci Rep 16, 7122 (2026). https://doi.org/10.1038/s41598-026-38321-0
Keywords: bacterial spores, surface adhesion, cryo-electron microscopy, protein assembly, food safety