Clear Sky Science · en
Selective Lis1 inactivation disrupts migration and positioning of cortical somatostatin interneurons
Why tiny brain cells and a single gene matter
Inside our brains, billions of nerve cells must end up in exactly the right place for thoughts, emotions and memories to work properly. This study looks at a small but powerful group of brain cells called somatostatin interneurons and a gene named Lis1, already known to cause a severe "smooth brain" disorder when mutated. By asking what happens when Lis1 is switched off only in this tiny cell population, the researchers uncover how subtle errors in cell movement during development can reshape key brain regions involved in mood, memory and mental illness.
Key players in the brain’s brake system
Somatostatin interneurons act as part of the brain’s braking system. They release the inhibitory signal GABA and a peptide called somatostatin, mainly targeting the branches of excitatory pyramidal neurons. This helps keep brain activity in balance, which is essential for normal perception, decision‑making and emotional control. Many of these interneurons are born in a deep region of the embryonic brain called the medial ganglionic eminence. From there, they travel sideways over long distances into the cerebral cortex, including the cingulate cortex, a hub for emotion, pain, and memory. Because this journey is complex and tightly timed, genes that guide cell movement are critical. Lis1 is one such gene, known to regulate the cell’s internal scaffolding and motor machinery that move the nucleus and shape the cell during migration.
A targeted genetic “switch off” experiment
To find out what Lis1 does specifically in somatostatin interneurons, the authors engineered mice in which Lis1 is deleted only in cells that turn on the somatostatin gene. They also used a fluorescent red protein to permanently mark these cells and follow them as they developed. This design allowed the team to distinguish effects inside somatostatin cells themselves from changes in the rest of the brain. They then examined two cingulate regions—the anterior cingulate cortex and the retrosplenial cortex—around one month after birth, a time when cortical circuits are relatively mature but still plastic. Careful counting methods were used to estimate both the absolute number and the density of somatostatin interneurons in each cortical layer.
Fewer interneurons and a shifted cortical pattern
In young adult mice lacking Lis1 in somatostatin interneurons, both the anterior cingulate and the retrosplenial cortex contained far fewer of these cells than in control animals. The reduction was large and consistent across all examined layers, indicating a real loss rather than a simple redistribution. When the researchers looked more closely at how remaining cells were arranged, they found a change in the usual layer pattern. In healthy mice, somatostatin interneurons tend to be more concentrated in deeper layers of the cortex. In the mutants, this gradient was inverted, with relatively more cells in the upper layers and fewer in the deepest layer. Importantly, the overall size of the cingulate regions did not shrink, so the lower density truly reflects missing interneurons, not a compressed tissue volume.
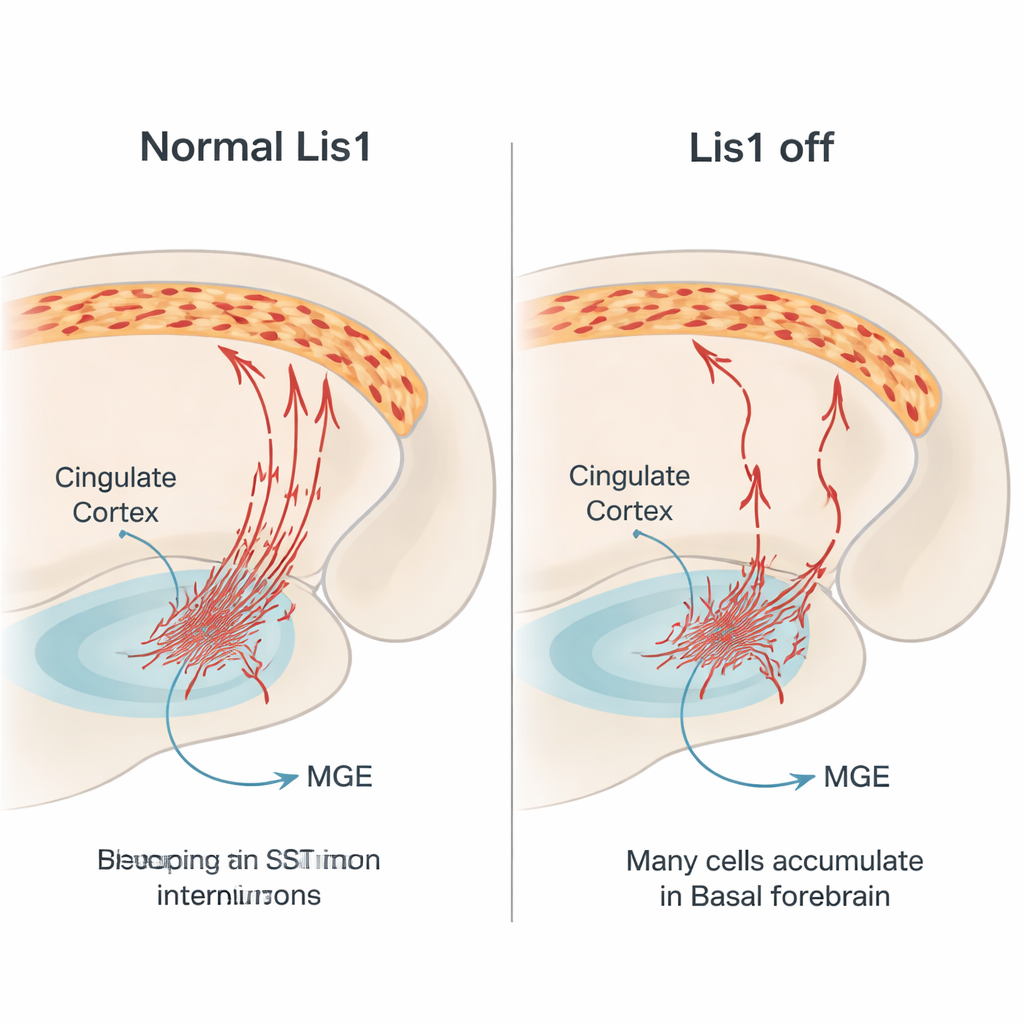
Developmental journey gone wrong
To understand how this loss arises, the team followed the red‑labeled somatostatin cells in embryos. They confirmed that Lis1 deletion starts after these cells have stopped dividing, ruling out a direct effect on their birth. Instead, the problem emerged during their long migration. At mid‑gestation stages, far fewer marked cells were found traveling through the normal pathways toward the cortex, and many appeared to stall in the basal forebrain, a ventral region they must cross. The migrating cells that did move showed altered shapes: shorter leading extensions, fewer branches and smaller, rounder cell bodies, all signs that their internal transport machinery was compromised. At later stages, the ventral region in mutants contained more cells showing markers of programmed cell death, suggesting that many stalled interneurons die before reaching the cortex.
What this means for brain health
Put simply, when Lis1 is switched off only in somatostatin interneurons, many of these cells start their journey normally but fail to reach their final destinations in the cingulate cortex. Some accumulate and die along the way, and those that arrive are fewer in number and unevenly positioned across cortical layers. Because somatostatin systems have been repeatedly linked to depression, schizophrenia, epilepsy and neurodegenerative diseases, this work highlights a direct, cell‑specific route by which Lis1 malfunction could contribute to disrupted brain rhythms and psychiatric symptoms. The study shows that even a modest‑sized neuron population, guided by a single gene, can be crucial for building the balanced circuits that underlie healthy thought and behavior.
Citation: Pombero, A., García-López, R., Geijo-Barrientos, E. et al. Selective Lis1 inactivation disrupts migration and positioning of cortical somatostatin interneurons. Sci Rep 16, 7330 (2026). https://doi.org/10.1038/s41598-026-38316-x
Keywords: somatostatin interneurons, Lis1 gene, neuronal migration, cingulate cortex, neurodevelopmental disorders