Clear Sky Science · en
Gut microbiota and resistome profiles of Swiss expatriates in Africa revealed by Nanopore metagenomics
Why your gut microbes change when you live abroad
Many people spend months or years living abroad, often in places where antibiotic‑resistant bacteria are common. This study asks a simple but important question: when Swiss expatriates live in African countries with a high burden of drug‑resistant infections, does the community of microbes and resistance genes in their intestines change in ways that might affect their health and the wider spread of antimicrobial resistance?
The invisible community inside us
Our intestines are home to trillions of microbes, mostly bacteria, that help digest food, train the immune system, and keep harmful germs at bay. Alongside these helpful microbes sits the “resistome” – the collection of genes that make bacteria resistant to antibiotics. Even healthy people carry many such genes. When individuals move to or travel through regions where multidrug‑resistant bacteria are common, they may quietly pick up new resistance genes and the mobile DNA elements, called plasmids, that spread them. Understanding how this happens is vital for controlling global antibiotic resistance.
Comparing expats in Europe and Africa
The researchers analyzed stool samples from 72 healthy Swiss expatriates returning to Switzerland: 39 had been living in African countries and 33 in other European countries. Instead of growing bacteria in the lab, they used a long‑read DNA sequencing technology called Nanopore shotgun metagenomics, which reads all the genetic material in a sample at once. This allowed them to map which bacteria were present (the microbiota) and which antibiotic resistance genes and plasmids they carried (the resistome and plasmidome). Each sample was sequenced twice for reliability, and sophisticated software was used to identify bacterial groups and resistance genes and to assemble longer genome fragments from the mixed DNA.
Surprising stability of gut microbes
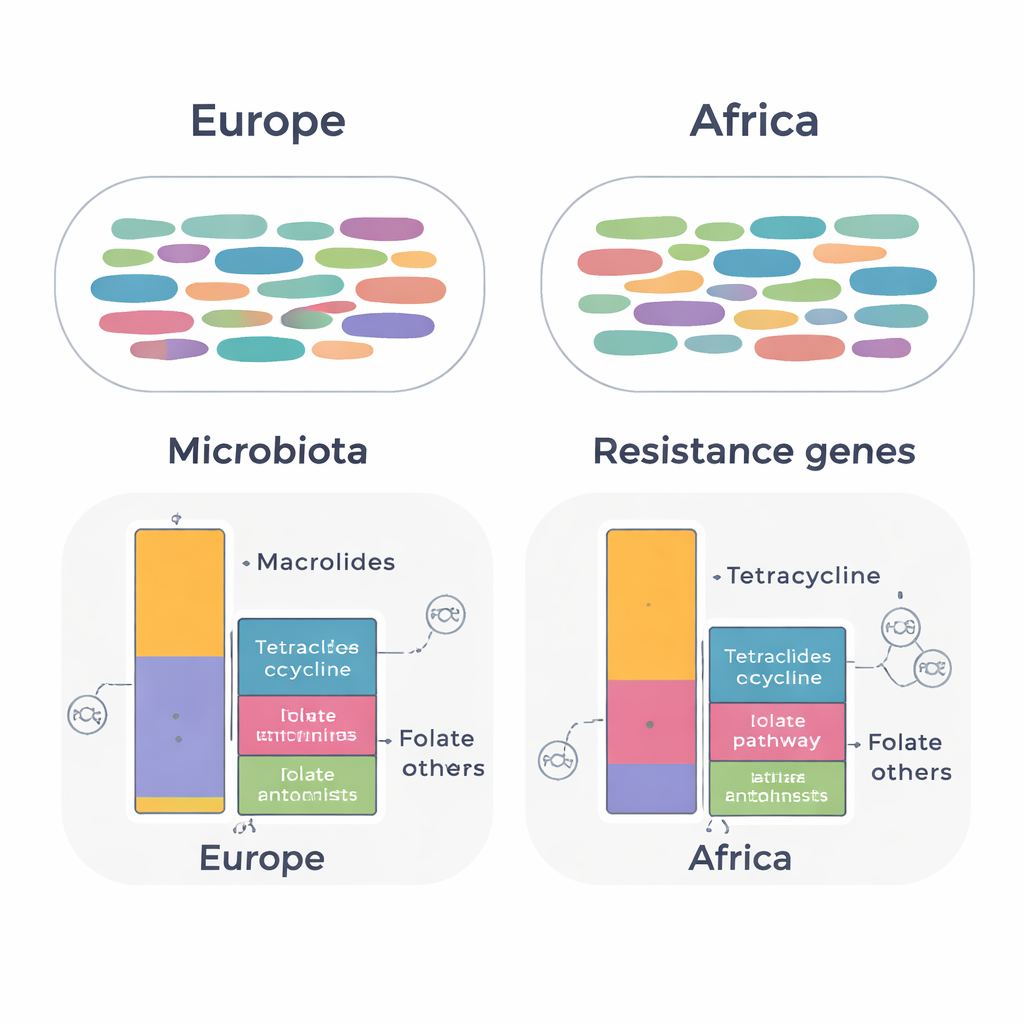
Despite more people in the Africa group being colonized with multidrug‑resistant gut bacteria in earlier culture‑based testing, the overall makeup of their gut microbiota looked remarkably similar to that of expats who had lived in Europe. Measures of diversity – how many different kinds of bacteria were present and how evenly they were represented – did not differ by continent, and statistical analyses showed no clear clustering of samples from Africa versus Europe. In both groups, familiar gut residents such as Blautia, Faecalibacterium, and Bacteroides dominated, suggesting that long‑term residence abroad does not necessarily overhaul the core bacterial community in healthy adults.
Resistance genes and mobile DNA tell a different story
When the team focused on resistance genes, more subtle but important differences emerged. Across all samples, they found 134 distinct resistance genes belonging to 14 classes of antibiotics. The total pattern of genes was broadly similar between continents, but expatriates who had lived in Africa carried higher levels of genes that protect bacteria against tetracyclines and drugs targeting the folate pathway (such as trimethoprim‑sulfamethoxazole). In contrast, expats from European countries showed higher levels of genes conferring resistance to macrolide antibiotics. Many of these genes were linked to common gut bacteria, including Ruminococcoides, Bifidobacterium, and Bacteroides. Clinically important genes, such as blaCTX-M-15, which can inactivate advanced cephalosporin antibiotics, were detected in Escherichia coli from both groups.
Plasmids as global shuttles for resistance
The study also tracked plasmids – small, often transferable DNA circles that shuttle resistance genes between bacteria and across environments. Using the long DNA reads, the researchers could sometimes see resistance genes and plasmid “replicon” markers on the same assembled DNA fragment, confirming they travelled together. They identified 46 different plasmid types, some unique to each continent and others shared. Notably, certain plasmids carried multiple resistance genes and resembled plasmids known from human, animal, food, and wastewater sources in different parts of the world. One plasmid type commonly linked to Enterococcus bacteria and found in chicken and wastewater was more frequent in stools from expats in Africa, underscoring how food, animals, and the environment may all contribute to what ends up in our gut.
What this means for everyday life and public health
For a lay reader, the key message is that simply living in a high‑risk region does not appear to radically reshape which gut bacteria you host, but it can tweak the mix of antibiotic resistance genes and the mobile DNA elements that spread them. These hidden changes, shaped in part by local antibiotic use patterns, may matter for future infections and for how resistance traits move between people, animals, and the environment. The work also shows that portable long‑read sequencing can act as an early warning tool, revealing how resistance genes and plasmids circulate in healthy travellers and expatriates before they cause disease.
Citation: Campos-Madueno, E.I., Aldeia, C. & Endimiani, A. Gut microbiota and resistome profiles of Swiss expatriates in Africa revealed by Nanopore metagenomics. Sci Rep 16, 7016 (2026). https://doi.org/10.1038/s41598-026-38302-3
Keywords: gut microbiome, antibiotic resistance, expatriates, plasmids, metagenomics