Clear Sky Science · en
Development and characterization of immortalized mouse intestinal epithelial cell lines
Why new gut cell models matter
The lining of the intestine is a busy border where food, helpful microbes, and dangerous germs all meet our bodies. To study how this barrier forms, heals, and fights infection, scientists need reliable cells they can grow in the lab. Today’s gold‑standard three‑dimensional “mini‑gut” cultures are powerful but expensive, delicate, and technically demanding, which limits who can use them and how often. This study introduces a more practical set of mouse gut cell lines designed to be easy to grow, genetically flexible, and still close enough to real tissue to answer many important biological and disease questions.
From mouse intestine to lab‑friendly cells
The researchers began with small segments of the mouse small intestine and colon, isolating tiny clusters of cells that include intestinal stem cells. These clusters, called spheroids, normally require a rich cocktail of growth factors and a gel‑like support material to survive. The team used a viral delivery system to introduce a gene called SV40 large T antigen, which allows cells to keep dividing indefinitely, creating “immortalized” lines. They then gradually weaned the cells off their complex media and gel support, adapting them to grow as simple flat layers on standard plastic dishes in common nutrient solutions. Clones were selected based on their appearance—either more sheet‑like and epithelial or more fibroblast‑like—and checked to be free of common contaminants such as mycoplasma. 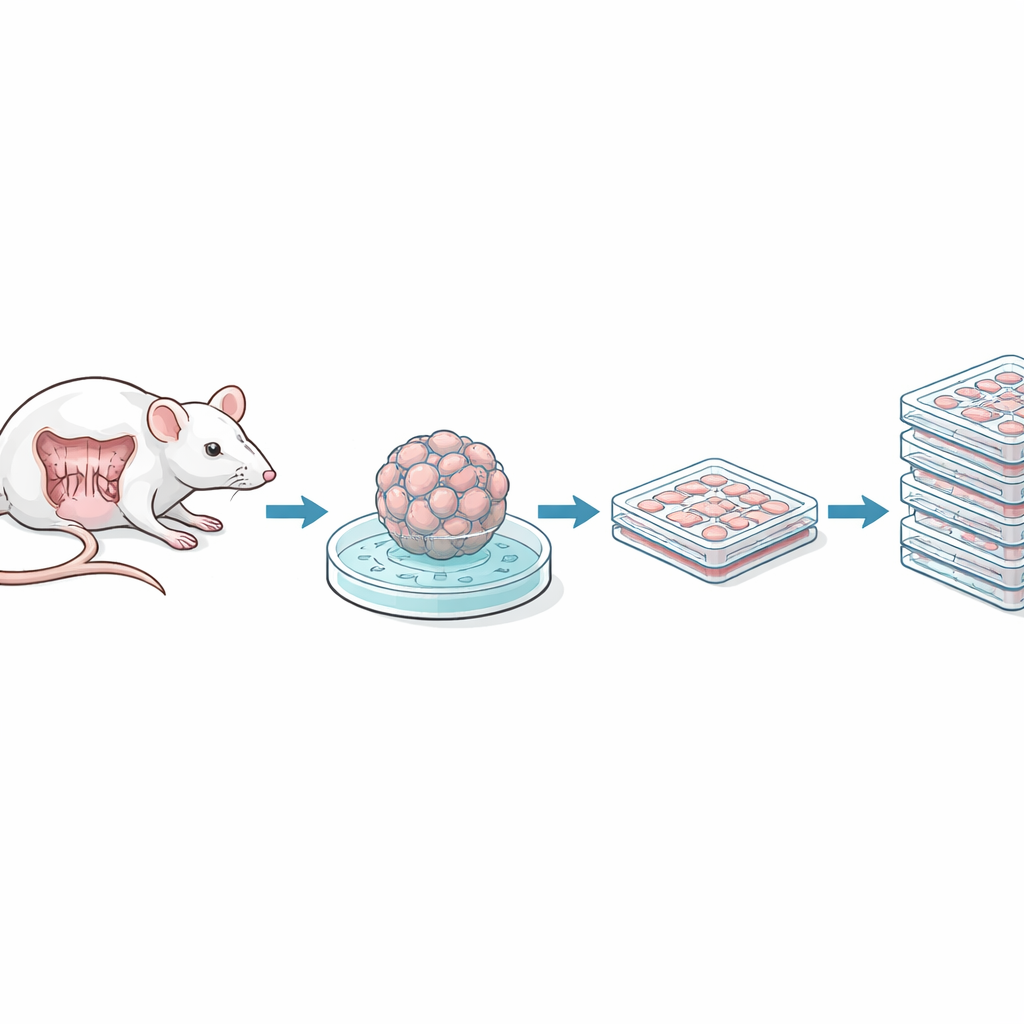
Checking identity and genetic health
Immortalizing cells can introduce unwanted genetic changes, so the team carefully verified what they had created. Using DNA fingerprinting methods, they showed that the new cell lines still closely matched the original C57BL/6 mouse tissue, and that no human DNA had crept in. Chromosome spreads revealed some extra or rearranged chromosomes, as is common in long‑lived cell lines, but overall the new lines were less heavily altered than widely used mouse colon cancer lines. When grown in a more demanding air–liquid interface setup that normally encourages full intestinal differentiation, the immortalized cells formed a basic single layer with a brush‑border‑like surface, although they did not fully mature into all the specialized cell types seen in native gut tissue. This suggests they most closely resemble actively dividing progenitor cells rather than fully differentiated cells.
Molecular personality: between epithelium and repair mode
To understand these cells at the gene expression level, the team compared their RNA profiles to those of the original spheroids and to normal intestinal cell types. The immortalized cells had turned down classic epithelial markers and turned up genes associated with a process called epithelial–mesenchymal transition, in which cells loosen their junctions and become more mobile—a state often seen during wound repair and in cancer. Yet they still expressed many junctional proteins and kept clear signatures of whether they came from the small intestine or colon. Overall, the lines resembled “transit amplifying” progenitor cells—rapidly dividing intermediates that lie between stem cells and mature gut cells—suggesting they model a regenerative, repair‑oriented epithelium.
Easy to reprogram and challenge with germs
A major advantage of these new lines is how readily they accept genetic and environmental manipulation. The researchers could introduce DNA into the cells using standard transfection reagents as well as lentiviral vectors, producing strong fluorescent reporter signals. They then tested how well the cells model host–microbe encounters. When infected with vesicular stomatitis virus, the cells supported viral replication but could be protected in a dose‑dependent way by pretreating them with interferon, a key antiviral signal. The same cells quickly switched on classic interferon‑stimulated genes, confirming an intact antiviral response. In separate experiments, the cells allowed invasion and survival of Salmonella bacteria and responded to bacterial components by switching on the NF‑κB pathway and secreting a range of inflammatory cytokines and chemokines, behaviors expected of an alert intestinal barrier. 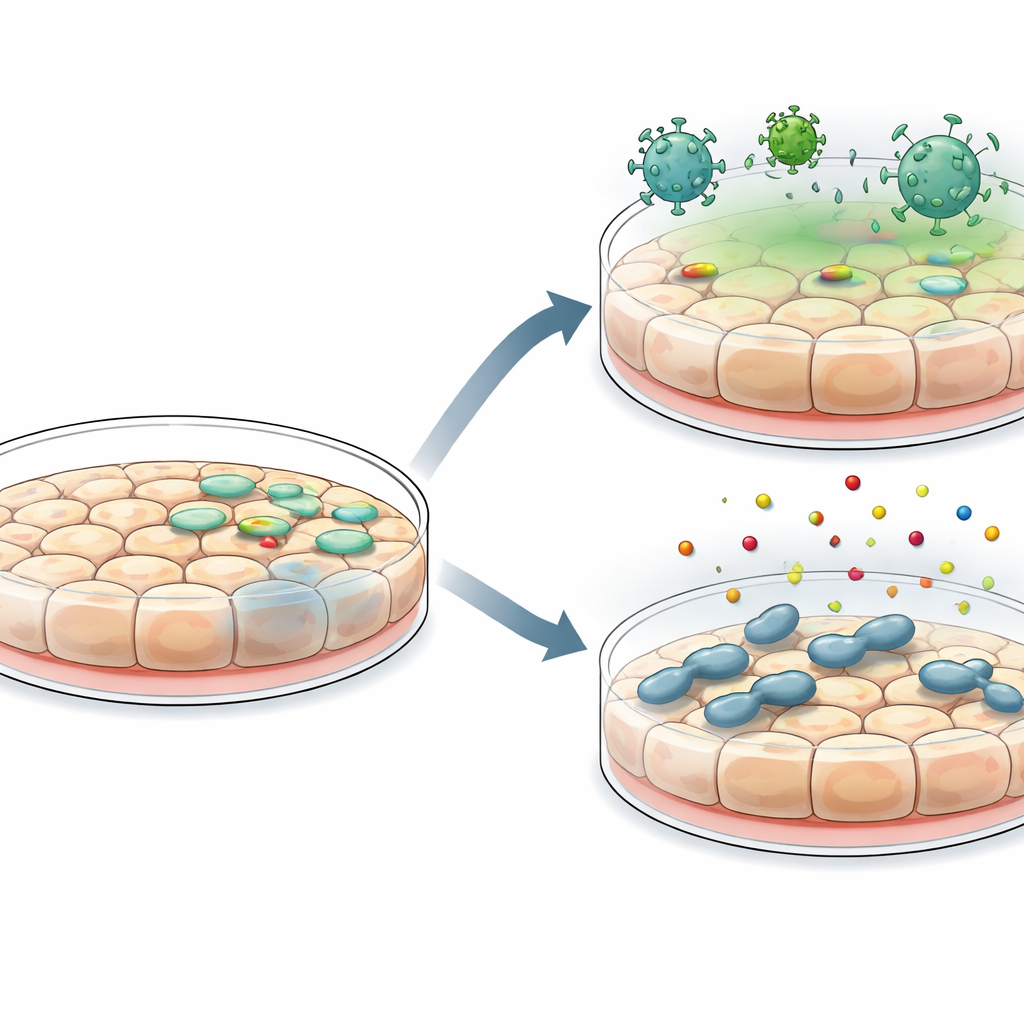
Lowering costs and opening doors
Because these immortalized lines grow on standard plastic in simple media, they are far cheaper and easier to handle than three‑dimensional mini‑guts. The authors estimate that running a 10,000‑compound screen would cost tens to hundreds of times less with their system than with primary spheroid or organoid cultures, and would require less time and specialized skill. While the cells do not perfectly mimic the full diversity and structure of the native intestine and show some traits of partial transformation, they fill an important niche: a mouse‑based, genetically tractable, and physiologically informed platform for high‑throughput discovery. In practice, these lines can serve as a first‑pass, scalable test bed, with promising findings later confirmed in more complex mini‑gut cultures or in animals.
What this means for gut research
In plain terms, the study delivers a new set of mouse gut cell lines that are easy to grow, easy to engineer, and responsive to viruses, bacteria, and immune signals in ways that echo real intestinal tissue. They sit between simple but poorly defined cancer lines and demanding but highly realistic organoid systems. By lowering both technical and financial barriers, these immortalized intestinal epithelial cells are poised to speed up studies of gut development, infection, inflammation, and drug responses, helping researchers move more quickly from basic questions to insights that can be tested in higher‑fidelity models and, eventually, in living organisms.
Citation: Zhou, J.Y., Espenschied, S.T., Lu, Q. et al. Development and characterization of immortalized mouse intestinal epithelial cell lines. Sci Rep 16, 8297 (2026). https://doi.org/10.1038/s41598-026-38295-z
Keywords: intestinal epithelial cells, immortalized cell lines, mouse gut models, host–pathogen interactions, innate immunity