Clear Sky Science · en
Heterogeneous graph neural networks reveal molecular mechanisms of folate deficiency in placental insufficiency through multiomics integration
Why This Matters for Mothers and Babies
Folate is best known as the vitamin in prenatal pills that helps prevent birth defects, but its role in a healthy pregnancy goes far deeper. This study looks at how a lack of folate can quietly damage the placenta—the organ that feeds and protects the growing baby. By combining cutting-edge biology with advanced artificial intelligence, the researchers show how subtle changes in genes, proteins, and small molecules work together to starve the placenta of what it needs, and explore how this knowledge could one day help doctors spot high‑risk pregnancies earlier.
When the Placenta Cannot Keep Up
Placental insufficiency occurs when the placenta cannot deliver enough oxygen and nutrients to the fetus, leading to complications such as poor growth, preeclampsia, and preterm birth. It affects roughly five to ten percent of pregnancies worldwide. Folate sits at the heart of many cell processes—building DNA, controlling which genes are turned on or off, and supporting healthy blood vessels. When folate levels are too low, these systems can misfire, but the precise chain of events inside placental tissue has been hard to trace. Traditional studies usually examine one type of biological data at a time, such as genes or metabolites, missing the bigger picture of how these layers interact.
Reading the Placenta with Many Lenses
To tackle this complexity, the team collected placental samples at delivery from 156 pregnancies with folate‑related placental insufficiency and 142 healthy controls. From each sample, they measured four different kinds of molecular information: DNA changes, gene activity, protein levels, and small metabolites. Instead of treating these measurements as long, separate lists, they linked them into a large biological map containing 6,704 molecules and over 16,000 known interactions. Genes, proteins, and metabolites were represented as different types of nodes connected by edges that reflected how they bind, regulate, or transform one another in real cells.
Teaching a Smart Network to Spot Trouble
The researchers then trained a specialized artificial intelligence model called a heterogeneous graph neural network on this molecular map. Unlike standard algorithms that see each measurement in isolation, this model “listens” to how signals spread across the entire network. It assigns attention weights to highlight the most informative molecules and connections for telling diseased placentas from healthy ones. On a held‑out test set, the model correctly classified 94.7% of placentas and achieved a near‑perfect area under the ROC curve of 0.978, clearly outperforming traditional machine‑learning methods and any analysis that used only a single type of data. This shows that the disease signal lies not just in individual markers, but in patterns shared across multiple biological layers.
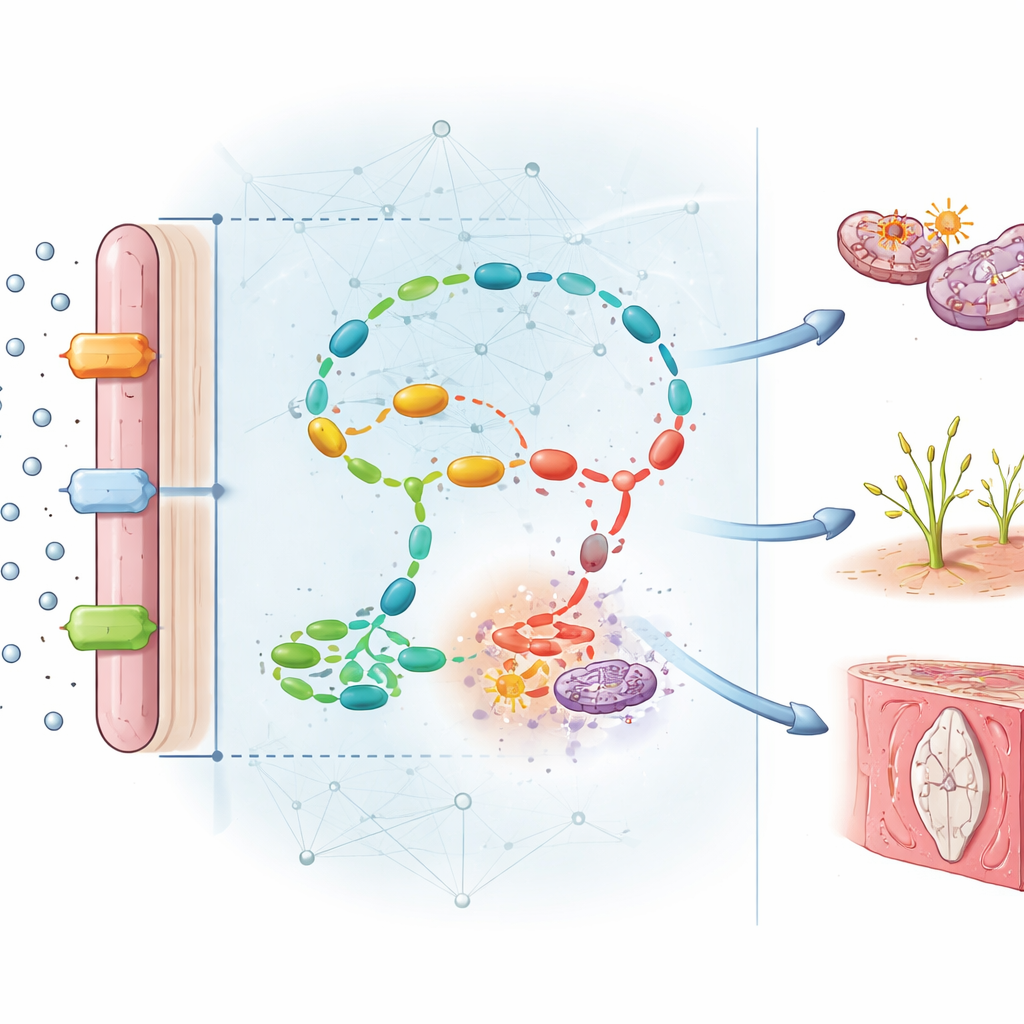
Uncovering the Hidden Chains of Damage
Beyond prediction, the model’s design allowed the authors to see which molecules carried the most weight in its decisions. Several stood out. Key folate‑handling components—such as the enzyme MTHFR and the folate transporter FOLR1—were strongly reduced in diseased placentas, while homocysteine, a toxic by‑product, accumulated more than sixfold. From the network, seven major functional modules emerged, linking disturbed folate chemistry to reduced DNA methylation, increased oxidative stress, faulty blood‑vessel growth, inflammation, and excessive cell death. In other words, folate deficiency does not harm the placenta in just one way; it sets off a web of connected failures that together limit blood flow and nutrient delivery to the fetus.
What This Could Mean for Future Care
For now, this work is mainly a powerful microscope rather than a bedside test. Because all samples were taken at delivery, the model cannot yet be used to predict risk during pregnancy, when intervention would matter most. Still, the molecular signatures it reveals—especially the combined changes in folate transport, homocysteine buildup, and damaged vessel‑growth pathways—offer a roadmap for searching blood‑based markers that might mirror what is happening in the placenta. With future studies that track mothers over time and compare their blood to their placental tissue, this approach could help build early, non‑invasive screening tools and more tailored nutritional or medical strategies to protect vulnerable pregnancies.
Citation: Xie, X., Li, Z., Xiao, Q. et al. Heterogeneous graph neural networks reveal molecular mechanisms of folate deficiency in placental insufficiency through multiomics integration. Sci Rep 16, 8417 (2026). https://doi.org/10.1038/s41598-026-38288-y
Keywords: placental insufficiency, folate deficiency, graph neural networks, multiomics, pregnancy complications