Clear Sky Science · en
Facile Preparation of imidazole-functionalized nanofibers for Cobalt removal from spent lithium-ion batteries
Turning Old Batteries into Urban Mines
Modern life runs on lithium-ion batteries, from phones to electric cars. When these batteries wear out, they can leak toxic metals—but they are also rich in valuable elements such as cobalt. This study explores a new filter material made from ultra-thin plastic fibers decorated with tiny ring-shaped molecules. Together, they act like a smart sponge that can pull cobalt out of complicated waste liquids from used batteries, helping to make battery recycling cleaner and more efficient.
Why Cobalt Matters in a Battery-Powered World
As the number of electric vehicles and portable devices soars, so does the pile of spent lithium-ion batteries. If these batteries are simply discarded, metals like cobalt can contaminate soil and water. Yet those same metals are expensive and finite, making old batteries a kind of man-made ore, or “urban mine.” Recovering cobalt is especially important because it is scarce, costly, and central to many common battery designs. Current recycling methods can separate metals, but often need large amounts of chemicals, work slowly, or struggle to pick out cobalt from look-alike metals such as lithium, nickel, and manganese. There is a growing need for smarter materials that can both grab a lot of cobalt and ignore most of the other metals.
Building a Smart Filter from Hair-Thin Fibers
The researchers focused on nanofibers—plastic threads thousands of times thinner than a human hair—that form a porous mat. They began with a common polymer called polyacrylonitrile, which is easy to spin into fibrous sheets and already used in filters. On its own, this material barely interacts with cobalt. The team transformed it by adding special chemical groups in two simple steps. First, they attached flexible “arms” rich in nitrogen atoms. Then they fastened small ring-shaped units called imidazoles to those arms. These rings have nitrogen atoms that are particularly good at latching onto cobalt ions. Microscopy and spectroscopy confirmed that the fibers stayed intact, became rougher and more porous, and were coated evenly with nitrogen-rich sites that can act as docking stations for cobalt.
How the New Fibers Catch and Hold Cobalt
When the modified fiber mat was placed in water containing dissolved cobalt, it soaked up the metal quickly and held a large amount relative to its own weight. Detailed tests showed that the uptake followed a pattern consistent with a single layer of cobalt ions covering a set of equivalent binding spots on the fiber surface, with a maximum loading of about 95 milligrams of cobalt per gram of material. The process reached most of its capacity within a couple of hours and matched models in which chemical bonding—rather than simple sticking to the surface—controls the rate. Temperature and other measurements suggested that cobalt ions give up some of their water shell and form stable complexes with the nitrogen atoms on the imidazole rings and nearby chemical linkages, releasing water and gaining a more ordered attachment to the fiber.
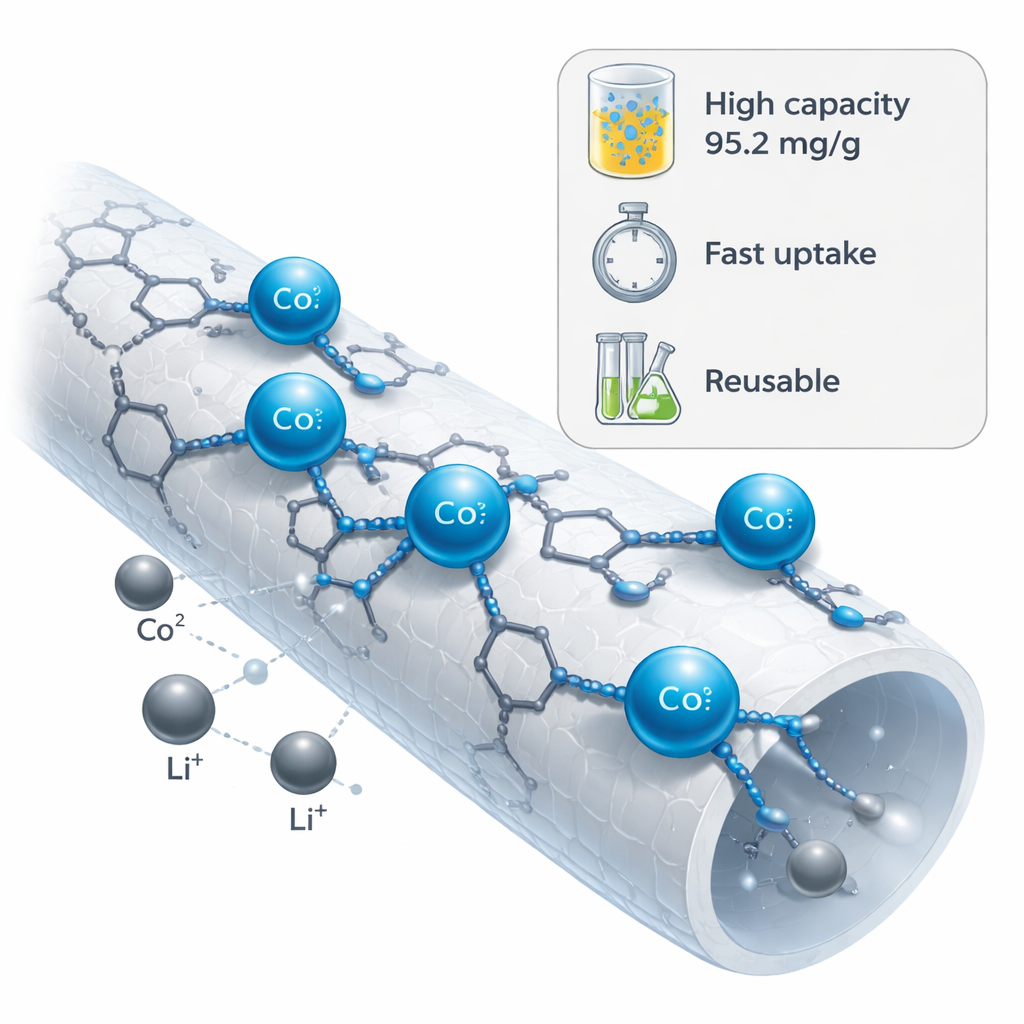
Picking Cobalt over Lithium and Lasting for Many Uses
A key challenge in real battery recycling is choosing cobalt out of a soup that also contains plenty of lithium. In side-by-side tests, the new nanofibers strongly preferred cobalt: they took up nearly twenty times more cobalt than lithium from mixtures similar to those obtained when batteries are dissolved in acid, and the calculated selectivity factor for cobalt over lithium was very high. After the fibers became loaded with cobalt, a mild acid wash was enough to strip the cobalt off and regenerate the material. Even after six rounds of use and cleaning, the fibers still kept more than four-fifths of their original capacity. This durability, combined with their high uptake and strong preference for cobalt, makes the material promising for repeated use in flow-through filters or packed columns.
A Step Toward Cleaner, Circular Battery Use
In everyday terms, the study shows a way to turn an ordinary plastic filter into a highly selective “cobalt magnet” using simple, scalable chemistry. By marrying the fast flow and large surface area of nanofiber mats with ring-shaped molecules that naturally like to grip cobalt, the researchers created a tool that can help recover this critical metal from complex battery waste streams. If adapted and tested with real factory-scale liquids, similar smart filters could support a more circular battery economy, where valuable metals are captured and reused rather than lost in landfills.
Citation: Sun, H., Shi, S., Li, Z. et al. Facile Preparation of imidazole-functionalized nanofibers for Cobalt removal from spent lithium-ion batteries. Sci Rep 16, 6884 (2026). https://doi.org/10.1038/s41598-026-38276-2
Keywords: battery recycling, cobalt recovery, nanofiber filters, wastewater treatment, critical metals