Clear Sky Science · en
Remodelling hierarchical NiCo2O4@ZnS nanorods with multi-walled carbon nanotubes as a counter electrode for dye-sensitized solar cell applications
Why cheaper solar power materials matter
Platinum, the shiny metal used in jewelry and car exhaust systems, is also a workhorse in some solar cells—but it is rare and expensive. This study explores a clever way to replace platinum in dye-sensitized solar cells, a class of low-cost, semi-transparent solar devices, with a mixture of more common ingredients. By rethinking the tiny architecture on the back contact of the cell, the researchers manage to match and even slightly outdo a platinum-based device, pointing toward cheaper and more sustainable solar technologies.
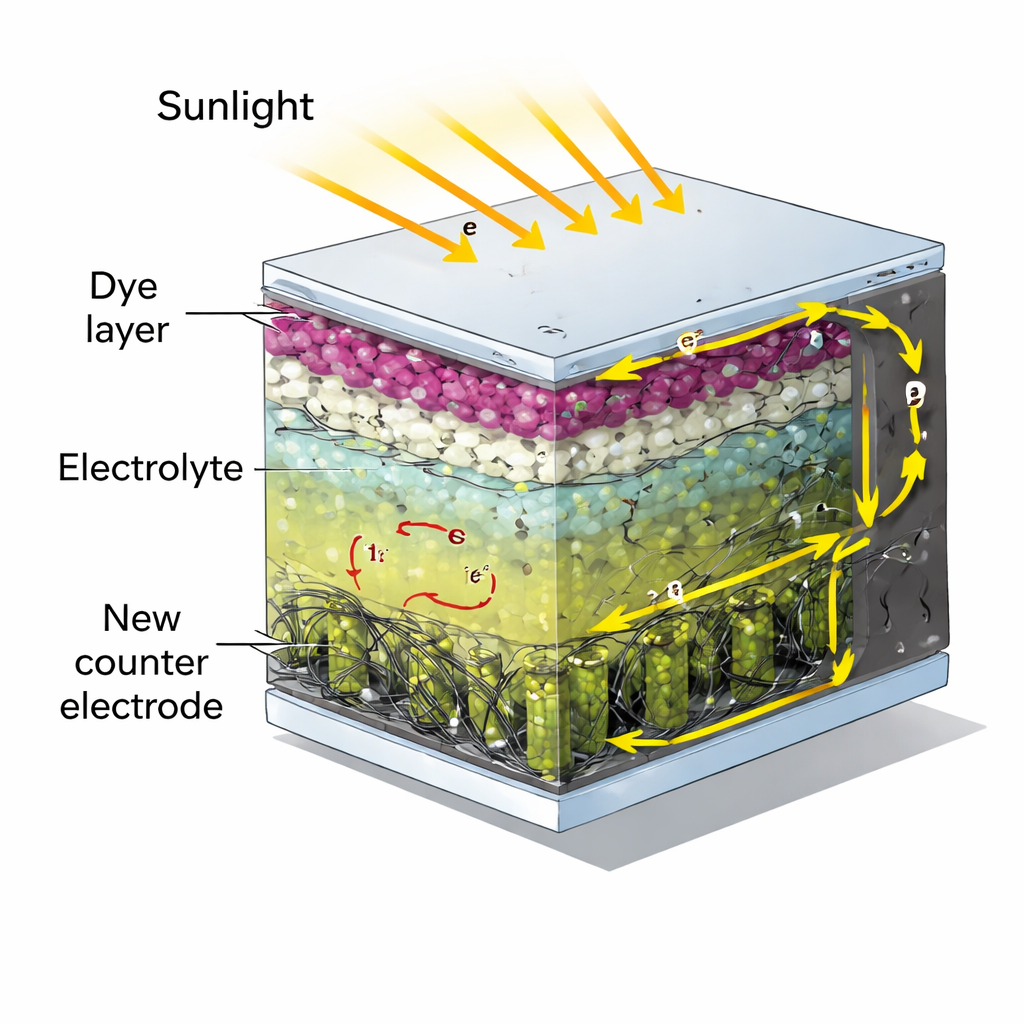
How this special type of solar cell works
Dye-sensitized solar cells operate a bit like artificial leaves. A colored dye on a porous white layer captures sunlight and injects electrons into an underlying semiconductor. These electrons then travel through an external circuit to do useful work before returning to the cell at a back contact called the counter electrode. Inside the cell, an iodine-based liquid shuttles charge between the dye and the counter electrode. The quality of this back contact strongly affects how efficiently the cell runs, because it must rapidly complete the final step of the electrical cycle: helping the iodine molecules swap electrons over and over again.
Building a new kind of back contact
Instead of a flat layer of platinum, the team built a three-part, sculpted material for the counter electrode. The backbone is made of nickel–cobalt oxide nanorods, which stand up like a microscopic forest and provide plenty of spots for chemical reactions. The surfaces of these rods are decorated with zinc sulfide particles that create extra reactive sites and tweak the local electronic environment where the redox chemistry takes place. Finally, a mesh of multi-walled carbon nanotubes threads through and around the rods, forming a highly conductive network that links the entire structure to the external circuit. All of this is assembled using solution-based steps at relatively low temperatures, which are compatible with scalable manufacturing.
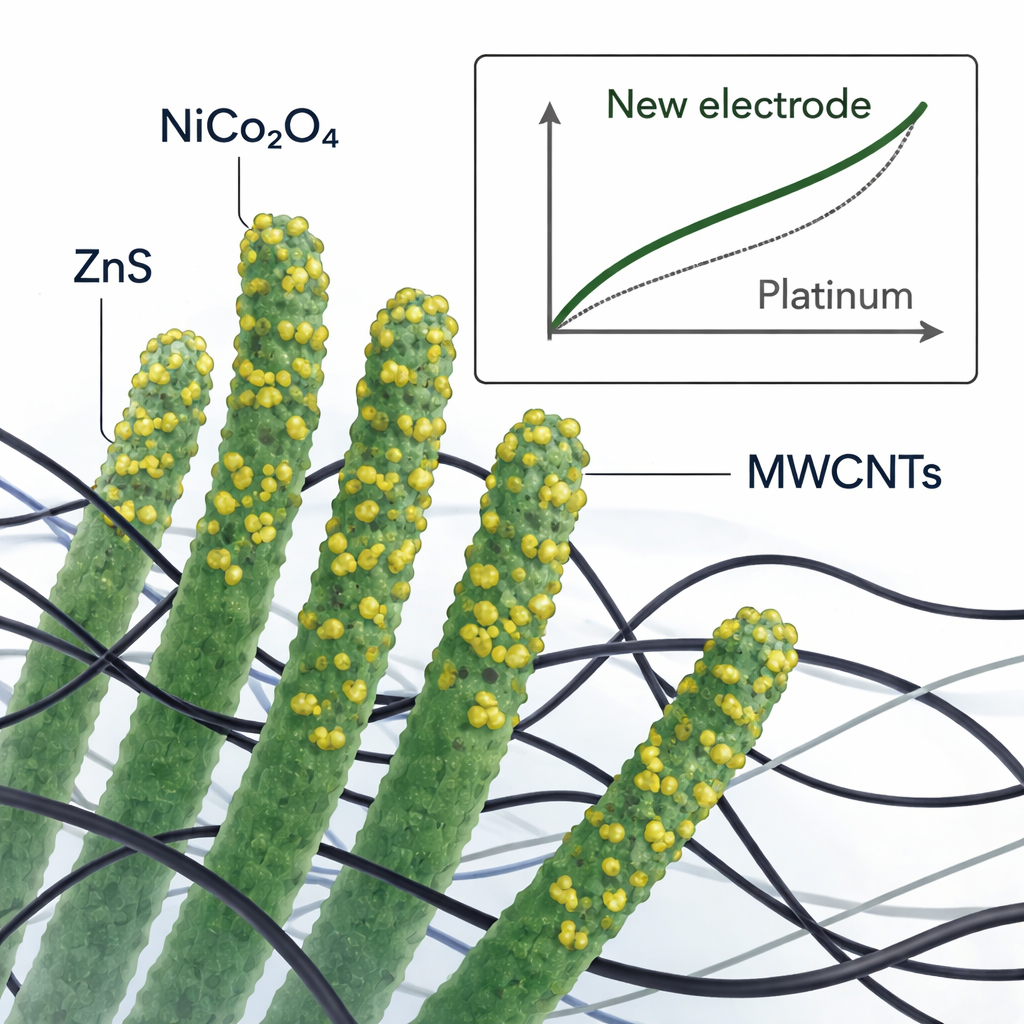
Peering into the structure at the nanoscale
To verify what they had built, the researchers used a suite of materials probes more familiar to a physics lab than a rooftop installer. X-ray diffraction confirmed that the nickel–cobalt oxide and zinc sulfide kept their well-ordered crystal structures when combined, and that the carbon nanotubes were successfully incorporated. Electron microscopes revealed long, straight nanorods coated with small zinc sulfide clusters, with worm-like nanotubes weaving between them. Chemical mapping showed that nickel, cobalt, zinc, sulfur, oxygen, and carbon were all present and well mixed, while surface-sensitive spectroscopy indicated a mix of oxidation states on nickel and cobalt—favorable for fast electron exchange with the iodine electrolyte.
From microscopic design to device performance
The team then tested how these intricate structures behaved both electrochemically and inside working solar cells. Electrochemical measurements showed that, as zinc sulfide and more carbon nanotubes were added, the material passed current more readily and required less extra voltage to drive the key iodine reactions. Impedance tests, which track how hard it is for charges to move across interfaces, revealed a marked drop in resistance for the optimized composite. When used as the counter electrode in a dye-sensitized solar cell, the best-performing mixture—containing 9 percent carbon nanotubes by weight—reached a power conversion efficiency of 10.03 percent under standard sunlight, slightly higher than an otherwise identical cell using platinum. It also showed better current output and a stronger "fill factor," a measure of how well the device maintains voltage under load.
Stability and practicality for real-world use
Thermogravimetric tests, which heat the material while tracking weight loss, indicated that the composite remained structurally robust in the temperature range relevant for solar-cell operation. Surface-area and pore measurements showed a mesoporous structure, with channels that allow the liquid electrolyte to penetrate and reach active sites without clogging the pathways for ion movement. Together, these traits—good electrical connectivity, ample reaction area, and maintained integrity—support reliable performance over time rather than a fragile laboratory curiosity.
What this means for future solar panels
For a non-specialist, the message is straightforward: by carefully layering common metal oxides, a sulfide coating, and carbon nanotubes at the nanometer scale, it is possible to replace pricey platinum in a key part of certain solar cells without sacrificing performance. The nickel–cobalt oxide provides the framework, zinc sulfide tunes the surface reactivity, and the nanotubes act as fast highways for electrons. This hierarchical design produces dye-sensitized solar cells that are efficient, potentially cheaper, and more sustainable, making them more attractive for applications such as building-integrated or flexible solar power where low cost and ease of fabrication are critical.
Citation: Nukunudompanich, M., Nachaithong, T., Phumuen, P. et al. Remodelling hierarchical NiCo2O4@ZnS nanorods with multi-walled carbon nanotubes as a counter electrode for dye-sensitized solar cell applications. Sci Rep 16, 6869 (2026). https://doi.org/10.1038/s41598-026-38255-7
Keywords: dye-sensitized solar cells, platinum-free electrodes, nickel cobalt oxide, carbon nanotubes, zinc sulfide