Clear Sky Science · en
Dexmedetomidine attenuates high-fat diet-induced metabolic dysfunction-associated steatotic liver disease in rats by regulating the autophagic pathway
Why this matters for everyday health
As waistlines expand worldwide, a silent problem is growing inside many people’s bodies: fat-laden livers that can progress to serious disease. This study explores whether a hospital sedative, dexmedetomidine, can also act as a surprising liver protector in rats fed a high-fat diet. By boosting the liver’s natural “self-cleaning” system, the drug appeared to limit fat buildup, oxidative damage, and cell death—pointing to a possible new way to tackle a very common modern illness.
Too much fat, a stressed liver
The liver is a central hub for handling fats, packaging them, burning them for energy, and sending them around the body. When that system is overwhelmed—often by long-term intake of calorie-dense, high-fat foods—fat droplets start to accumulate inside liver cells. This condition, now termed metabolic dysfunction-associated steatotic liver disease (MASLD), affects roughly a quarter of the global population and is tightly linked to obesity, diabetes, high blood pressure, and abnormal blood lipids. Over time, simple fat buildup can progress to inflammation, scarring, and even cirrhosis. A key idea in the field is the “two-hit” theory: first, excess fat piles up; second, stressed and damaged cell machinery sparks inflammation and further injury.
The cell’s cleanup crew: recycling fat from within
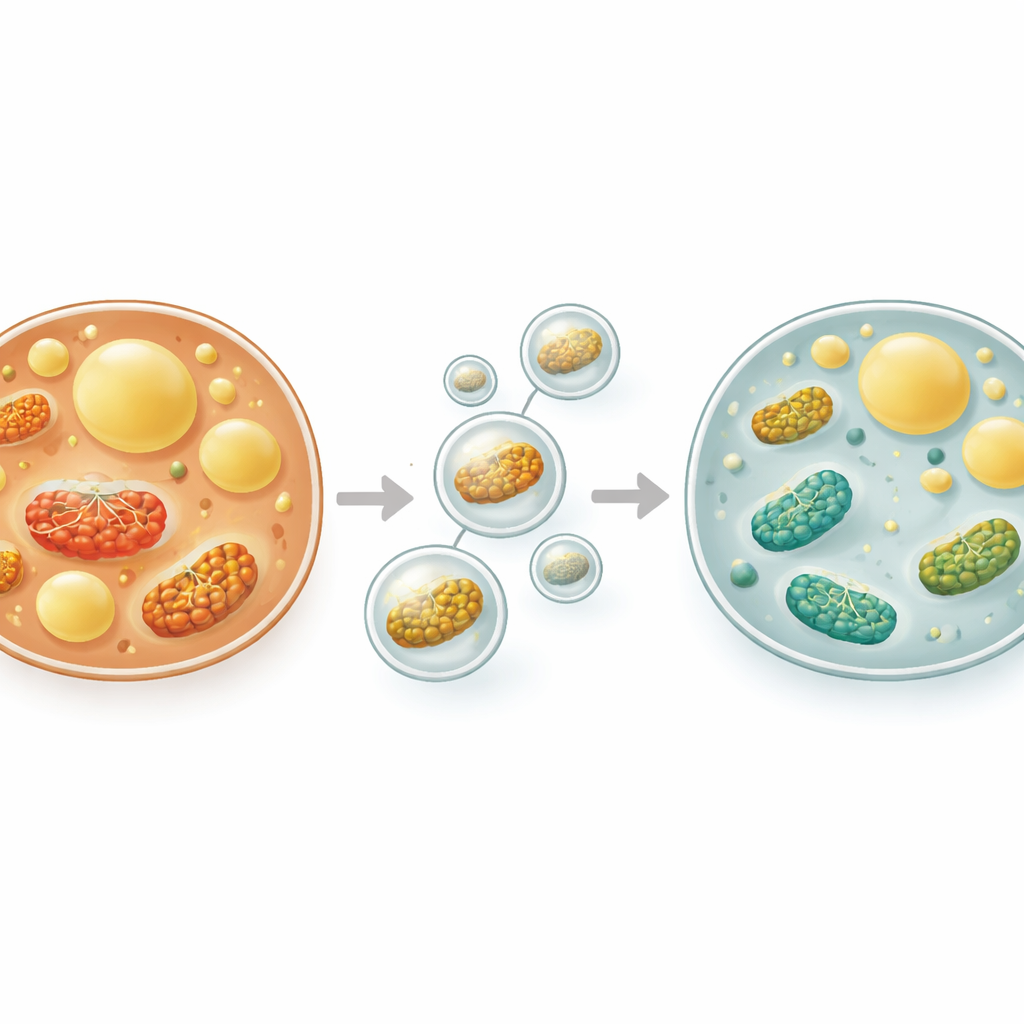
To counter these insults, liver cells rely on autophagy—a built-in recycling process in which worn-out components and surplus fat droplets are wrapped in tiny membranes and delivered to cellular “digestive” compartments. A specialized form, called lipophagy, specifically targets the fat droplets. When working well, lipophagy breaks down stored fat into smaller molecules that can be burned for energy, while removing damaged structures that generate harmful reactive oxygen species. When this inner cleanup crew slows down, fat and toxic by-products accumulate, driving MASLD forward. In this study, the researchers focused on several well-known molecular sentinels that track how active this system is, including proteins involved in starting autophagy and in marking cargo for disposal.
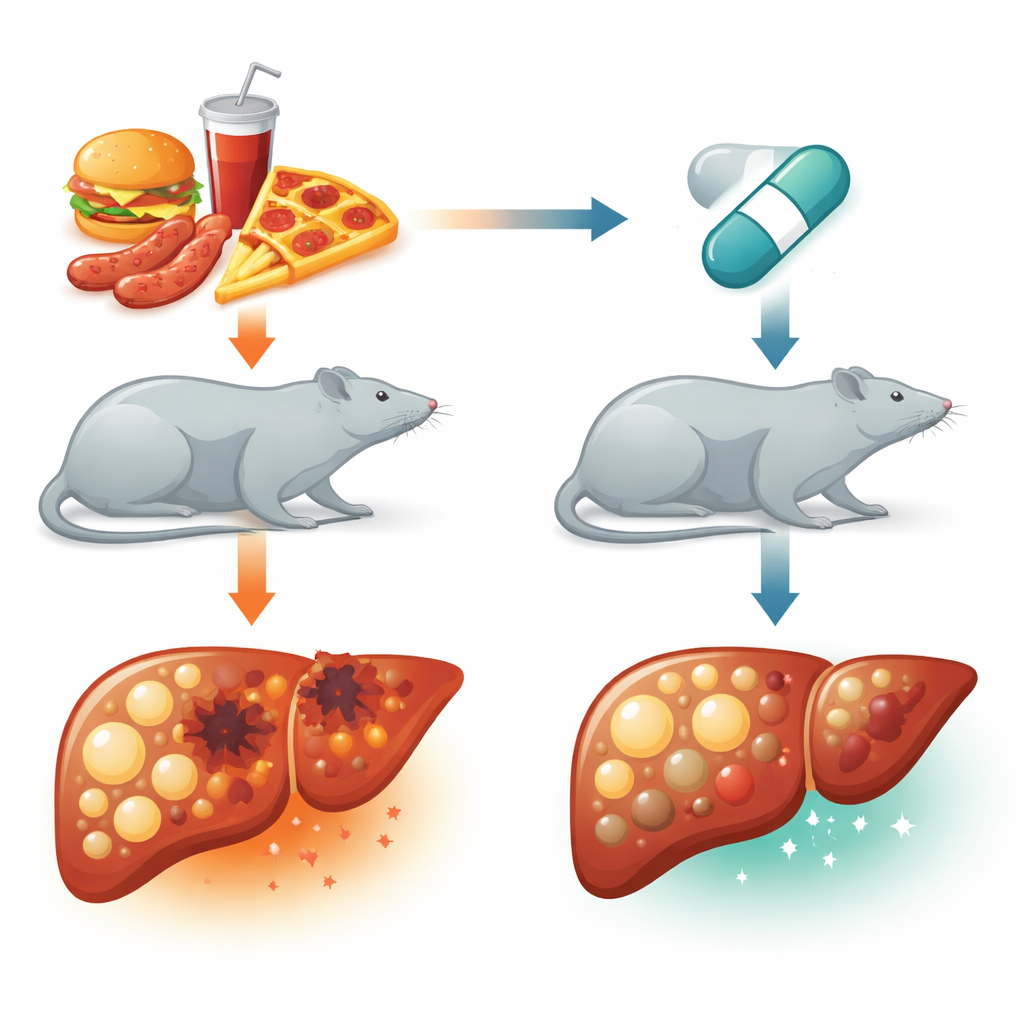
Testing a sedative as a liver shield in rats
The team used male rats and fed them a diet in which 60% of calories came from beef tallow for eight weeks, reliably triggering fatty liver and metabolic disruption. Two groups of these high-fat-fed rats also received daily injections of dexmedetomidine at low or higher doses, while control rats ate regular chow. At the end of the experiment, the scientists measured blood markers of liver damage, cholesterol and triglycerides, chemical signs of oxidative stress, and levels of key autophagy-related proteins in liver tissue. They also examined thin liver slices under the microscope and used staining methods to visualize proteins linked to cell survival or programmed cell death.
Cleaner livers, calmer cells
Rats on the high-fat diet alone developed severe fatty change in their livers: large fat droplets crowded the cells, blood tests for liver injury soared, and oxidative stress rose sharply while protective antioxidants fell. At the same time, molecular hallmarks of autophagy were strongly suppressed, and proteins that build up when this system stalls became abundant. Markers of cell suicide, such as caspase-3, increased, while protective proteins like Bcl-2 declined. In striking contrast, rats treated with dexmedetomidine showed dose-dependent improvements. Their livers contained far less visible fat, and blood levels of liver enzymes, total cholesterol, and triglycerides dropped toward normal. Inside liver cells, antioxidant defenses improved and damaging lipid peroxidation fell. Crucially, proteins that kick-start and carry out autophagy rebounded, while the “backlog” protein p62 fell, indicating that the cleanup machinery had not only been turned on but was functioning. At the same time, caspase-3 levels decreased and Bcl-2 rose, suggesting fewer cells were undergoing programmed death.
What these findings could mean
Taken together, the results suggest that dexmedetomidine helps the fatty, stressed liver by reactivating its internal recycling systems—especially the pathway that breaks down lipid droplets—and by reducing oxidative injury and cell loss. In this rat model, the drug nudged the liver away from a trajectory of worsening fat buildup and damage, toward a state that more closely resembles health. While these findings do not mean the sedative is ready as a treatment for people with MASLD, they highlight a promising strategy: supporting the liver’s own cleanup and repair tools to counteract the effects of high-fat diets and metabolic dysfunction.
Citation: Ezzat, S.M., Sharawy, M.H. & Suddek, G.M. Dexmedetomidine attenuates high-fat diet-induced metabolic dysfunction-associated steatotic liver disease in rats by regulating the autophagic pathway. Sci Rep 16, 8943 (2026). https://doi.org/10.1038/s41598-026-38250-y
Keywords: fatty liver disease, high-fat diet, autophagy, dexmedetomidine, oxidative stress